Abstract
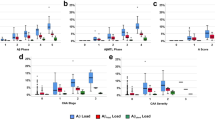
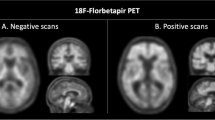
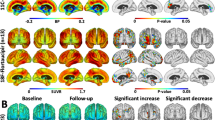
Pittsburgh compound B (PiB) radiotracer for positron emission tomography (PET) imaging can bind to different types of amyloid-β plaques and blood vessels (cerebral amyloid angiopathy). However, the relative contributions of different plaque subtypes (diffuse versus cored/compact) to in vivo PiB PET signal on a region-by-region basis are incompletely understood. Of particular interest is whether the same staging schemes for summarizing amyloid-β burden are appropriate for both late-onset and autosomal dominant forms of Alzheimer disease (LOAD and ADAD). Here, we compared antemortem PiB PET with follow-up postmortem estimation of amyloid-β burden using stereologic methods to estimate the relative area fraction of diffuse and cored/compact amyloid-β plaques across 16 brain regions in 15 individuals with ADAD and 14 individuals with LOAD. In ADAD, we found that PiB PET correlated with diffuse plaques in the frontal, parietal, temporal, and striatal regions commonly used to summarize amyloid-β burden in PiB PET, and correlated with both diffuse and cored/compact plaques in the occipital lobe and parahippocampal gyrus. In LOAD, we found that PiB PET correlated with both diffuse and cored/compact plaques in the anterior cingulate, frontal lobe (middle frontal gyrus), and parietal lobe, and showed additional correlations with diffuse plaque in the amygdala and occipital lobe, and with cored/compact plaque in the temporal lobe. Thus, commonly used PiB PET summary regions predominantly reflect diffuse plaque burden in ADAD and a mixture of diffuse and cored/compact plaque burden in LOAD. In direct comparisons of ADAD and LOAD, postmortem stereology identified much greater mean amyloid-β plaque burdens in ADAD versus LOAD across almost all brain regions studied. However, standard PiB PET did not recapitulate these stereologic findings, likely due to non-trivial amyloid-β plaque burdens in ADAD within the cerebellum and brainstem—commonly used reference regions in PiB PET. Our findings suggest that PiB PET summary regions correlate with amyloid-β plaque burden in both ADAD and LOAD; however, they might not be reliable in direct comparisons of regional amyloid-β plaque burden between the two forms of AD.



Similar content being viewed by others
References
Bateman RJ, Aisen PS, De Strooper B, Fox NC, Lemere CA, Ringman JM et al (2011) Autosomal-dominant Alzheimer’s disease: a review and proposal for the prevention of Alzheimer’s disease. Alz Res Therapy 3, 1. https://doi.org/10.1186/alzrt59
Benzinger TLS, Blazey T, Jack CR, Koeppe RA, Su Y, Xiong C et al (2013) Regional variability of imaging biomarkers in autosomal dominant Alzheimer’s disease. Proc Natl Acad Sci USA 110:E4502–E4509. https://doi.org/10.1073/pnas.1317918110
Van Berckel BNM, Ossenkoppele R, Tolboom N, Yaqub M, Foster-Dingley JC, Windhorst AD et al (2013) Longitudinal amyloid imaging using 11C-PiB: Methodologic considerations. J Nucl Med 54:1570–1576. https://doi.org/10.2967/jnumed.112.113654
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259. https://doi.org/10.1007/BF00308809
Burkoth TS, Benzinger TLS, Urban V, Lynn DG, Meredith SC, Thiyagarajan P (1999) Self-assembly of Aβ((10–35))-PEG block copolymer fibrils [13]. J Am Chem Soc 121:7429–7430
van Buuren S, Groothuis-Oudshoorn K (2011) Mice: multivariate imputation by chained equations in R. J Stat Softw 45:1–67. https://doi.org/10.18637/jss.v045.i03
Cairns NJ, Perrin RJ, Franklin EE, Carter D, Vincent B, Xie M et al (2015) Neuropathologic assessment of participants in two multi-center longitudinal observational studies: the Alzheimer Disease Neuroimaging Initiative (ADNI) and the Dominantly Inherited Alzheimer Network (DIAN). Neuropathology 35:390–400. https://doi.org/10.1111/neup.12205
Caro LG (1964) Chapter 16 high-resolution autoradiography. Methods Cell Biol 1:327–363. https://doi.org/10.1016/S0091-679X(08)62098-1
Charidimou A, Farid K, Baron JC (2017) Amyloid-PET in sporadic cerebral amyloid angiopathy. Neurology 89:1490–1498. https://doi.org/10.1212/WNL.0000000000004539
Chen CD, Holden TR, Gordon BA, Franklin EE, Li Y, Coble DW et al (2020) Ante- and postmortem tau in autosomal dominant and late-onset Alzheimer’s disease. Ann Clin Transl Neurol 7:2475–2480. https://doi.org/10.1002/acn3.51237
Clark CM, Pontecorvo MJ, Beach TG, Bedell BJ, Coleman RE, Doraiswamy PM et al (2012) Cerebral PET with florbetapir compared with neuropathology at autopsy for detection of neuritic amyloid-β plaques: a prospective cohort study. Lancet Neurol 11:669–678. https://doi.org/10.1016/S1474-4422(12)70142-4
Clark CM, Schneider JA, Bedell BJ, Beach TG, Bilker WB, Mintun MA et al (2011) Use of Florbetapir-PET for imaging β-amyloid pathology. JAMA 305(3):275–283. https://doi.org/10.1001/JAMA.2010.2008
Corrada MM, Sonnen JA, Kim RC, Kawas CH (2016) Microinfarcts are common and strongly related to dementia in the oldest-old: The 90+ study. Alzheimer’s Dement 12:900–908. https://doi.org/10.1016/j.jalz.2016.04.006
Curtis C, Gamez JE, Singh U, Sadowsky CH, Villena T, Sabbagh MN et al (2015) Phase 3 trial of flutemetamol labeled with radioactive fluorine 18 imaging and neuritic plaque density. JAMA Neurol 72:287–294. https://doi.org/10.1001/jamaneurol.2014.4144
Davies CA, Mann DMA (1993) Is the “preamyloid” of diffuse plaques in Alzheimer’s disease really nonfibrillar? Am J Pathol 143:1594–1605
Dickson TC, Vickers JC (2001) The morphological phenotype of β-amyloid plaques and associated neuritic changes in Alzheimer’s disease. Neuroscience 105:99–107. https://doi.org/10.1016/S0306-4522(01)00169-5
Ding T, Wu T, Mazidi H, Zhang O, Lew MD (2020) Single-molecule orientation localization microscopy for resolving structural heterogeneities between amyloid fibrils. Optica 7:602. https://doi.org/10.1364/optica.388157
Driscoll I, Troncoso JC, Rudow G, Sojkova J, Pletnikova O, Zhou Y et al (2012) Correspondence between in vivo 11C-PiB-PET amyloid imaging and postmortem, region-matched assessment of plaques. Acta Neuropathol 124:823–831. https://doi.org/10.1007/s00401-012-1025-1
Dé Rique Bard F, Barbour R, Cannon C, Carretto R, Fox M, Games D et al (2003) Epitope and isotype specificities of antibodies to-amyloid peptide for protection against Alzheimer’s disease-like neuropathology. Proceedings of the National Academy of Sciences 100(4):2023–2028. https://doi.org/10.1073/pnas.0436286100
Efron B (2007) Size, power and false discovery rates. Ann Stat 35:1351–1377. https://doi.org/10.1214/009053606000001460
Di Fede G, Catania M, Maderna E, Ghidoni R, Benussi L, Tonoli E et al (2018) Molecular subtypes of Alzheimer’s disease. Sci Rep. https://doi.org/10.1038/s41598-018-21641-1
Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C et al (2002) Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33:341–355. https://doi.org/10.1016/S0896-6273(02)00569-X
Frucht SJ, Koo EH (1993) β-amyloid protein is higher in alzheimer’s disease brains: description of aquantitative biochemical assay. J Neuropathol Exp Neurol 52:640–647. https://doi.org/10.1097/00005072-199311000-00011
Gordon BA, Blazey TM, Su Y, Hari-Raj A, Dincer A, Flores S et al (2018) Spatial patterns of neuroimaging biomarker change in individuals from families with autosomal dominant Alzheimer’s disease: a longitudinal study. Lancet Neurol 17:241–250. https://doi.org/10.1016/S1474-4422(18)30028-0
Gravina SA, Ho L, Eckman CB, Long KE, Otvos L, Younkin LH et al (1995) Amyloid β protein (Aβ) in Alzheimer’s disease brain. Biochemical and immunocytochemical analysis with antibodies specific for forms ending at Aβ40 or Aβ42(43). J Biol Chem 270:7013–7016. https://doi.org/10.1074/jbc.270.13.7013
Grothe MJ, Barthel H, Sepulcre J, Dyrba M, Sabri O, Teipel SJ (2017) In vivo staging of regional amyloid deposition. Neurology 89:2031–2038. https://doi.org/10.1212/WNL.0000000000004643
Hanseeuw BJ, Betensky RA, Mormino EC, Schultz AP, Sepulcre J, Becker JA et al (2018) PET staging of amyloidosis using striatum. Alzheimer’s Dement 14:1281–1292. https://doi.org/10.1016/j.jalz.2018.04.011
Hyman BT, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Carrillo MC et al (2012) National Institute on Aging-Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease. Alzheimer’s Dement 8:1–13. https://doi.org/10.1016/j.jalz.2011.10.007
Ikonomovic MD, Buckley CJ, Heurling K, Sherwin P, Jones PA, Zanette M et al (2016) Post-mortem histopathology underlying β-amyloid PET imaging following flutemetamol F 18 injection. Acta Neuropathol Commun 4:130. https://doi.org/10.1186/s40478-016-0399-z
Ikonomovic MD, Klunk WE, Abrahamson EE, Mathis CA, Price JC, Tsopelas ND et al (2008) Post-mortem correlates of in vivo PiB-PET amyloid imaging in a typical case of Alzheimer’s disease. Brain 131:1630–1645. https://doi.org/10.1093/brain/awn016
Jack CR, Knopman DS, Jagust WJ, Petersen RC, Weiner MW, Aisen PS et al (2013) Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol 12:207–216
Jack CR, Knopman DS, Jagust WJ, Shaw LM, Aisen PS, Weiner MW et al (2010) Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol 9:119–128
James G, Witten D, Hastie T, Tibshirani R (2017) An introduction to statistical learning. Springer, New York
Johnson KA, Gregas M, Becker JA, Kinnecom C, Salat DH, Moran EK et al (2007) Imaging of amyloid burden and distribution in cerebral amyloid angiopathy. Ann Neurol 62:229–234. https://doi.org/10.1002/ana.21164
La Joie R, Ayakta N, Seeley WW, Borys E, Boxer AL, DeCarli C et al (2019) Multisite study of the relationships between antemortem [11 C]PIB-PET Centiloid values and postmortem measures of Alzheimer’s disease neuropathology. Alzheimer’s Dement 15:205–216. https://doi.org/10.1016/j.jalz.2018.09.001
Kida E, Wisniewski KE, Wisniewski HM (1995) Early amyloid -β deposits show different immunoreactivity to the amino- and carboxy-terminal regions of β-peptide in Alzheimer’s disease and Down’s syndrome brain. Neurosci Lett 193:105–108. https://doi.org/10.1016/0304-3940(95)11678-P
Klunk WE, Engler H, Nordberg A, Wang Y, Blomqvist G, Holt DP et al (2004) Imaging brain amyloid in Alzheimer’s Disease with Pittsburgh Compound-B. Ann Neurol 55:306–319. https://doi.org/10.1002/ana.20009
Klunk WE, Koeppe RA, Price JC, Benzinger TL, Devous MD, Jagust WJ et al (2015) The Centiloid project: standardizing quantitative amyloid plaque estimation by PET. Alzheimer’s Dement 11:1-15.e4. https://doi.org/10.1016/j.jalz.2014.07.003
Klunk WE, Lopresti BJ, Ikonomovic MD, Lefterov IM, Koldamova RP, Abrahamson EE et al (2005) Binding of the positron emission tomography tracer Pittsburgh Compound-B reflects the amount of amyloid-β in Alzheimer’s Disease brain but not in transgenic mouse brain. J Neurosci 25:10598–10606. https://doi.org/10.1523/JNEUROSCI.2990-05.2005
Klunk WE, Price JC, Mathis CA, Tsopelas ND, Lopresti BJ, Ziolko SK et al (2007) Amyloid deposition begins in the striatum of presenilin-1 mutation carriers from two unrelated pedigrees. J Neurosci 27:6174–6184. https://doi.org/10.1523/JNEUROSCI.0730-07.2007
Knowles RB, Wyart C, Buldyrev SV, Cruz L, Urbanc B, Hasselmo ME et al (1999) Plaque-induced neurite abnormalities: implications for disruption of neural networks in Alzheimer’s disease. Proc Natl Acad Sci USA 96:5274–5279. https://doi.org/10.1073/pnas.96.9.5274
Koivunen J, Verkkoniemi A, Aalto S, Paetau A, Ahonen JP, Viitanen M et al (2008) PET amyloid ligand [11C]PIB uptake shows predominantly striatal increase in variant Alzheimer’s disease. Brain 131:1845–1853. https://doi.org/10.1093/brain/awn107
Lalowski M, Golabek A, Lemere CA, Selkoe DJ, Wisniewski HM, Beavis RC et al (1996) The “Nonamyloidogenic” p3 fragment (amyloid β17-42) is a major constituent of down’s syndrome cerebellar preamyloid. J Biol Chem 271:33623–33631. https://doi.org/10.1074/jbc.271.52.33623
Larner AJ, Doran M (2006) Clinical phenotypic heterogeneity of Alzheimer’s disease associated with mutations of the presenilin-1 gene. J Neurol 253:139–158
Lemere CA, Blusztajn JK, Yamaguchi H, Wisniewski T, Saido TC, Selkoe DJ (1996) Sequence of deposition of heterogeneous amyloid β-peptides and APO E in down syndrome: implications for initial events in amyloid plaque formation. Neurobiol Dis 3:16–32. https://doi.org/10.1006/nbdi.1996.0003
Lockhart A, Lamb JR, Osredkar T, Sue LI, Joyce JN, Ye L et al (2007) PIB is a non-specific imaging marker of amyloid-beta (Aβ) peptide-related cerebral amyloidosis. Brain 130:2607–2615. https://doi.org/10.1093/brain/awm191
Lowe VJ, Lundt ES, Senjem ML, Schwarz CG, Min HK, Przybelski SA et al (2018) White matter reference region in PET studies of 11C-Pittsburgh compound B uptake: effects of age and amyloid-β deposition. J Nucl Med 59:1583–1589. https://doi.org/10.2967/jnumed.117.204271
Ly JV, Donnan GA, Villemagne VL, Zavala JA, Ma H, O’Keefe G et al (2010) 11C-PIB binding is increased in patients with cerebral amyloid angiopathy-related hemorrhage. Neurology 74:487–493. https://doi.org/10.1212/WNL.0b013e3181cef7e3
Maarouf CL, Daugs ID, Spina S, Vidal R, Kokjohn TA, Patton RL et al (2008) Histopathological and molecular heterogeneity among individuals with dementia associated with Presenilin mutations. Mol Neurodegener 3:20. https://doi.org/10.1186/1750-1326-3-20
Mirra SS, Heyman A, McKeel D, Sumi SM, Crain BJ, Brownlee LM et al (1991) The consortium to establish a registry for Alzheimer’s disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer’s disease. Neurology 41:479–486. https://doi.org/10.1212/wnl.41.4.479
Montine TJ, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Dickson DW et al (2012) National institute on aging-Alzheimer’s association guidelines for the neuropathologic assessment of Alzheimer’s disease: a practical approach. Acta Neuropathol 123:1–11. https://doi.org/10.1007/s00401-011-0910-3
Morris JC, Aisen PS, Bateman RJ, Benzinger TL, Cairns NJ, Fagan AM et al (2012) Developing an international network for Alzheimer’s research: the dominantly inherited alzheimer network. Clin Investig (Lond) 2:975–984. https://doi.org/10.4155/cli.12.93
Murray ME, Lowe VJ, Graff-Radford NR, Liesinger AM, Cannon A, Przybelski SA et al (2015) Clinicopathologic and 11C-Pittsburgh compound B implications of Thal amyloid phase across the Alzheimer’s disease spectrum. Brain 138:1370–1381. https://doi.org/10.1093/brain/awv050
Nelson PT, Alafuzoff I, Bigio EH, Bouras C, Braak H, Cairns NJ et al (2012) Correlation of Alzheimer disease neuropathologic changes with cognitive status: a review of the literature. J Neuropathol Exp Neurol 71:362–381
Nelson PT, Dickson DW, Trojanowski JQ, Jack CR, Boyle PA, Arfanakis K et al (2019) Limbic-predominant age-related TDP-43 encephalopathy (LATE): consensus working group report. Brain 142:1503–1527
Olichney JM, Hansen LA, Hofstetter CR, Lee JH, Katzman R, Thal LJ (2000) Association between severe cerebral amyloid angiopathy and cerebrovascular lesions in Alzheimer disease is not a spurious one attributable to apolipoprotein E4. Arch Neurol 57:869–874. https://doi.org/10.1001/archneur.57.6.869
Portelius E, Andreasson U, Ringman JM, Buerger K, Daborg J, Buchhave P et al (2010) Distinct cerebrospinal fluid amyloid peptide signatures in sporadic and PSEN1 A431E-associated familial Alzheimer’s disease. Mol Neurodegener 5:2. https://doi.org/10.1186/1750-1326-5-2
Price JL, McKeel DW, Buckles VD, Roe CM, Xiong C, Grundman M et al (2009) Neuropathology of nondemented aging: presumptive evidence for preclinical Alzheimer disease. Neurobiol Aging 30:1026–1036. https://doi.org/10.1016/j.neurobiolaging.2009.04.002
Remes AM, Laru L, Tuominen H, Aalto S, Kemppainen N, Mononen H et al (2008) Carbon 11-labeled Pittsburgh Compound B positron emission tomographic amyloid imaging in patients with APP locus duplication. Arch Neurol 65:540–544. https://doi.org/10.1001/archneur.65.4.540
Ringman JM, Monsell S, Ng DW, Zhou Y, Nguyen A, Coppola G et al (2016) Neuropathology of autosomal dominant Alzheimer disease in the National Alzheimer Coordinating Center Database. J Neuropathol Exp Neurol 75:284. https://doi.org/10.1093/JNEN/NLV028
Roberts BR, Lind M, Wagen AZ, Rembach A, Frugier T, Li QX et al (2017) Biochemically-defined pools of amyloid-β in sporadic Alzheimer’s disease: Correlation with amyloid PET. Brain 140:1486–1498. https://doi.org/10.1093/brain/awx057
Ryman DC, Acosta-Baena N, Aisen PS, Bird T, Danek A, Fox NC et al (2014) Symptom onset in autosomal dominant Alzheimer disease: a systematic review and meta-analysis. Neurology 83:253–260. https://doi.org/10.1212/WNL.0000000000000596
Sabri O, Sabbagh MN, Seibyl J, Barthel H, Akatsu H, Ouchi Y et al (2015) Florbetaben PET imaging to detect amyloid beta plaques in Alzheimer’s disease: Phase 3 study. Alzheimer’s Dement 11:964–974. https://doi.org/10.1016/j.jalz.2015.02.004
Schöll M, Wall A, Thordardottir S, Ferreira D, Bogdanovic N, Långström B et al (2012) Low PiB PET retention in presence of pathologic CSF biomarkers in Arctic APP mutation carriers. Neurology 79:229–236. https://doi.org/10.1212/WNL.0b013e31825fdf18
Sepulveda-Falla D, Chavez-Gutierrez L, Portelius E, Vélez JI, Barrera-Ocampo A, Dinkel F et al (2018) A multifactorial model of pathology for age of onset variability in Familial Alzheimer’s disease. Sci Transl Med revised ve. https://doi.org/10.1007/s00401-020-02249-0
Su Y, Blazey TM, Snyder AZ, Raichle ME, Marcus DS, Ances BM et al (2015) Partial volume correction in quantitative amyloid imaging. Neuroimage 107:55–64. https://doi.org/10.1016/j.neuroimage.2014.11.058
Su Y, D’Angelo GM, Vlassenko AG, Zhou G, Snyder AZ, Marcus DS et al (2013) Quantitative analysis of PiB-PET with FreeSurfer ROIs. PLoS ONE 8:e73377. https://doi.org/10.1371/journal.pone.0073377
Su Y, Flores S, Hornbeck RC, Speidel B, Vlassenko AG, Gordon BA et al (2018) Utilizing the Centiloid scale in cross-sectional and longitudinal PiB PET studies. NeuroImage Clin 19:406–416. https://doi.org/10.1016/j.nicl.2018.04.022
Tang M, Ryman DC, McDade E, Jasielec MS, Buckles VD, Cairns NJ et al (2016) Neurological manifestations of autosomal dominant familial Alzheimer’s disease: a comparison of the published literature with the Dominantly Inherited Alzheimer Network observational study (DIAN-OBS). Lancet Neurol 15:1317–1325. https://doi.org/10.1016/S1474-4422(16)30229-0
Thal DR, Beach TG, Zanette M, Lilja J, Heurling K, Chakrabarty A et al (2018) Estimation of amyloid distribution by [18F]flutemetamol PET predicts the neuropathological phase of amyloid β-protein deposition. Acta Neuropathol 136:557–567. https://doi.org/10.1007/s00401-018-1897-9
Thal DR, Ronisz A, Tousseyn T, Rijal Upadhaya A, Balakrishnan K, Vandenberghe R et al (2019) Different aspects of Alzheimer’s disease-related amyloid β-peptide pathology and their relationship to amyloid positron emission tomography imaging and dementia. Acta Neuropathol Commun 7:178. https://doi.org/10.1186/s40478-019-0837-9
Thal DR, Rüb U, Orantes M, Braak H (2002) Phases of Aβ-deposition in the human brain and its relevance for the development of AD. Neurology 58:1791–1800. https://doi.org/10.1212/WNL.58.12.1791
Thal DR, Sassin I, Schultz C, Haass C, Braak E, Braak H (1999) Fleecy amyloid deposits in the internal layers of the human entorhinal cortex are comprised of N-terminal truncated fragments of aβ. J Neuropathol Exp Neurol 58:210–216. https://doi.org/10.1097/00005072-199902000-00010
Theuns J, Marjaux E, Vandenbulcke M, Van Laere K, Kumar-Singh S, Bormans G et al (2006) Alzheimer dementia caused by a novel mutation located in the APP C-terminal intracytosolic fragment. Hum Mutat 27:888–896. https://doi.org/10.1002/humu.20402
Thiyagarajan P, Burkoth TS, Urban V, Seifert S, Benzinger TLS, Morgan DM et al (2000) pH dependent self assembly of β-amyloid(10–35) and β-amyloid(10–35)-PEG3000. J Appl Cryst 33:535–539. https://doi.org/10.1107/S0021889899014387
Tomiyama T, Nagata T, Shimada H, Teraoka R, Fukushima A, Kanemitsu H et al (2008) A new amyloid β variant favoring oligomerization in Alzheimer’s-type dementia. Ann Neurol 63:377–387. https://doi.org/10.1002/ana.21321
Van Essen DC, Jbabdi S, Sotiropoulos SN, Chen C, Dikranian K, Coalson T et al (2013) Mapping connections in humans and non-human primates: aspirations and challenges for diffusion imaging. In: Diffusion MRI: from quantitative measurement to in vivo neuroanatomy, 2nd edn. Elsevier Inc., Amsterdam, pp 337–358. https://doi.org/10.1016/B978-0-12-396460-1.00016-0
Verwey NA, Hoozemans JJM, Korth C, Van Royen MR, Prikulis I, Wouters D et al (2013) Immunohistochemical characterization of novel monoclonal antibodies against the N-terminus of amyloid β-peptide. Amyloid 20:179–187. https://doi.org/10.3109/13506129.2013.797389
Villemagne VL, Burnham S, Bourgeat P, Brown B, Ellis KA, Salvado O et al (2013) Amyloid β deposition, neurodegeneration, and cognitive decline in sporadic Alzheimer’s disease: a prospective cohort study. Lancet Neurol 12:357–367. https://doi.org/10.1016/S1474-4422(13)70044-9
Vonsattel JPG, Myers RH, Tessa Hedley-Whyte E, Ropper AH, Bird ED, Richardson EP (1991) Cerebral amyloid angiopathy without and with cerebral hemorrhages: a comparative histological study. Ann Neurol 30:637–649. https://doi.org/10.1002/ana.410300503
Wong DF, Rosenberg PB, Zhou Y, Kumar A, Raymont V, Ravert HT et al (2010) In vivo imaging of amyloid deposition in Alzheimer disease using the radioligand 18F-AV-45 (flobetapir F 18). J Nucl Med 51:913–920. https://doi.org/10.2967/jnumed.109.069088
Funding
Data collection and sharing for this project was supported by the Knight Alzheimer Disease Research Center (Knight ADRC, P30AG066444, P01AG026276, and P01AG03991) and the Dominantly Inherited Alzheimer Network (DIAN, U19AG032438). This manuscript has been reviewed by DIAN Study investigators for scientific content and consistency of data interpretation with previous DIAN Study publications. We acknowledge the altruism of the participants and their families and contributions of the Knight ADRC and DIAN research and support staff at each of the participating sites for their contributions to this study. For the provision of brain tissue and staining, we acknowledge the Knight ADRC Neuropathology Core and the DIAN Neuropathology Core (P01AG003991). We thank the staff of the Betty Martz Laboratory for Neurodegenerative Disease for their excellent technical support. The authors also acknowledge support from the Neuroimaging Informatics and Analysis Center (P30NS098577). C.D.C. acknowledges support from the NSF GRFP (DGE-1745038). N.J-M. acknowledges support from the Alzheimer’s Association International Research Grant Program (AARFD-20-681815). B.A.G. acknowledges support from the NIH (K01AG053474). J.C.M. acknowledges support from the NIH (UF1AG032438).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
J.C. has served on a medical advisory board for Otsuka Pharmaceuticals. GE Healthcare holds a license agreement with the University of Pittsburgh based on the PiB-PET technology described in this manuscript. W.E.K. is a co-inventor of PiB and, as such, has a financial interest in this license agreement. GE Healthcare provided no grant support for this study and had no role in the design or interpretation of results or preparation of this manuscript. All other authors have no conflicts of interest with PiB-PET and had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. J.C.M. is funded by NIH grants # P30 AG066444; P01AG003991; P01AG026276; U19 AG032438; and U19 AG024904. Neither J.C.M. nor his family owns stock or has equity interest (outside of mutual funds or other externally directed accounts) in any pharmaceutical or biotechnology company. T.L.S.B. has investigator-initiated research funding from the NIH, the Alzheimer’s Association, the Barnes-Jewish Hospital Foundation, and Avid Radiopharmaceuticals (a wholly owned subsidiary of Eli Lilly); participates as a site investigator in clinical trials sponsored by Avid Radiopharmaceuticals, Eli Lilly, Biogen, Eisai, Jaansen, and Roche; serves as an unpaid consultant to Eisai and Siemens; and is on the Speaker’s Bureau for Biogen.
Data and materials availability
Data are available by request to the Knight ADRC (knightadrc.wustl.edu/research/resourcerequest.htm) and the DIAN-Obs (dian.wustl.edu/our-research/observational-study/dian-observational-study-investigator-resources/data-request-terms-and-instructions/).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, C.D., Joseph-Mathurin, N., Sinha, N. et al. Comparing amyloid-β plaque burden with antemortem PiB PET in autosomal dominant and late-onset Alzheimer disease. Acta Neuropathol 142, 689–706 (2021). https://doi.org/10.1007/s00401-021-02342-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-021-02342-y