Abstract
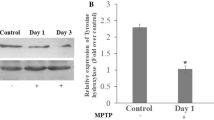
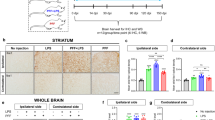
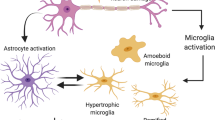
Parkinson's disease (PD) is a common neurodegenerative disorder characterized by a selective loss of dopaminergic neurons in the substantia nigra (SN). It has been suggested that microglial inflammation augments the progression of PD. Neuromelanin (NM), a complex polymer pigment found in catecholaminergic neurons, has sparked interest because of the suggestion that NM is involved in cell death in Parkinson's disease, possibly via microglia activation. To further investigate the possible role of NM in the pathogenesis of PD, we conducted in vivo experiments to find out whether microglial cells become activated after injection of human neuromelanin (NM) into (1) the cerebral cortex or (2) the substantia nigra to monitor in this PD-relevant model both microglial activation and possible neurodegeneration. In this study, adult male Wistar rats received an intracerebral injection of either NM, bacterial lipopolysaccharide (LPS, positive control), phosphate-buffered saline (PBS, negative control) or colloidal gold suspension (negative particular control). After different survival times (1, 8 or 12 weeks), brain slices from the cerebral cortex or substantia nigra (SN, 1 week) were stained with Iba-1 and/or GFAP antibody to monitor microglial and astrocytic reaction, and with tyrosine hydroxylase (TH) to monitor dopaminergic cell survival (SN group only). The injection of LPS induced a strong inflammatory response in the cortex as well in the substantia nigra. Similar results could be obtained after NM injection, while the injection of PBS or gold suspension showed only moderate or no glial activation. However, the inflammatory response declined during the time course. In the SN group, there was, apart from strong microglia activation, a significant dopaminergic cell loss after 1 week of survival time. Our findings clearly indicate that extracellular NM could be one of the key molecules leading to microglial activation and neuronal cell death in the substantia nigra. This may be highly relevant to the elucidation of therapeutic strategies in PD.




Similar content being viewed by others
References
Banati RB, Daniel SE, Blunt SB (1998) Glial pathology but absence of apoptotic nigral neurons in long- standing Parkinson's disease. Mov Disord 13:221–227
Banati RB, Gehrmann J, Schubert P, Kreutzberg GW (1993) Cytotoxicity of microglia. Glia 7:111–118
Beach TG, Sue LI, Walker DG, Lue LF, Connor DJ, Caviness JN, Sabbagh MN, Adler CH (2007) Marked microglial reaction in normal aging human substantia nigra: correlation with extraneuronal neuromelanin pigment deposits. Acta Neuropathol 114:419–424
Block ML, Hong JS (2005) Microglia and inflammation-mediated neurodegeneration: multiple triggers with a common mechanism. Prog Neurobiol 76:77–98
Braak H, Braak E, Yilmazer D, Schultz C, de Vos RA, Jansen EN (1995) Nigral and extranigral pathology in Parkinson's disease. J Neural Transm Suppl 46:15–31
Castano A, Herrera AJ, Cano J, Machado A (1998) Lipopolysaccharide intranigral injection induces inflammatory reaction and damage in nigrostriatal dopaminergic system. J Neurochem 70:1584–1592
Croisier E, Graeber MB (2006) Glial degeneration and reactive gliosis in alpha-synucleinopathies: the emerging concept of primary gliodegeneration. Acta Neuropathol 112:517–530
De Pablos RM, Herrera AJ, Villaran RF, Cano J, Machado A (2005) Dopamine-dependent neurotoxicity of lipopolysaccharide in substantia nigra. FASEB J 19:407–409
Faucheux BA, Martin ME, Beaumont C, Hauw JJ, Agid Y, Hirsch EC (2003) Neuromelanin associated redox-active iron is increased in the substantia nigra of patients with Parkinson's disease. J Neurochem 86:1142–1148
Gibb WR (1992) Melanin, tyrosine hydroxylase, calbindin and substance P in the human midbrain and substantia nigra in relation to nigrostriatal projections and differential neuronal susceptibility in Parkinson's disease. Brain Res 581:283–291
Giulian D, Corpuz M (1993) Microglial secretion products and their impact on the nervous system. Adv Neurol 59:315–320
Ghosh A, Roy A, Liu X, Kordower JH, Mufson EJ, Hartley DM, Ghosh S, Mosley RL, Gendelman HE, Pahan K (2007) Selective inhibition of NF-κB activation prevents dopaminergic neuronal loss in a mouse model of Parkinson's disease. Proc Natl Acad Sci USA 104:18754–18759
Hagg T, Varon S (1993) Ciliary neurotrophic factor prevents degeneration of adult rat substantia nigra dopaminergic neurons in vivo. Proc Natl Acad Sci U S A 90:6315–6319
Imamura K, Hishikawa N, Sawada M, Nagatsu T, Yoshida M, Hashizume Y (2003) Distribution of major histocompatibility complex class II-positive microglia and cytokine profile of Parkinson's disease brains. Acta Neuropathol 106:518–526
Ishikawa A, Takahashi H (1998) Clinical and neuropathological aspects of autosomal recessive juvenile parkinsonism. J Neurol 245:4–9
Kastner A, Hirsch EC, Lejeune O, Javoy-Agid F, Rascol O, Agid Y (1992) Is the vulnerability of neurons in the substantia nigra of patients with Parkinson's disease related to their neuromelanin content? J Neurochem 59:1080–1089
Langston JW, Forno LS, Tetrud J, Reeves AG, Kaplan JA, Karluk D (1999) Evidence of active nerve cell degeneration in the substantia nigra of humans years after 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine exposure. Ann Neurol 46:598–605
Lawson LJ, Perry VH, Dri P, Gordon S (1990) Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain. Neuroscience 39:151–170
Lindquist NG, Larsson BS, Lyden-Sokolowski A (1988) Autoradiography of [14C]paraquat or [14C]diquat in frogs and mice: accumulation in neuromelanin. Neurosci Lett 93:1–6
McGeer PL, Itagaki S, Boyes BE, McGeer EG (1988) Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson's and Alzheimer’s disease brains. Neurology 38:1285–1291
McGeer PL, McGeer EG (1995) The inflammatory response system of brain: implications for therapy of Alzheimer and other neurodegenerative diseases. Brain Res Brain Res Rev 21:195–218
McGeer PL, Yasojima K, McGeer EG (2001) Inflammation in Parkinon´s disease. Adv Neurol 86:83–89
Pal PK, Samii A, Calne DB (1999) Manganese neurotoxicity: a review of clinical features, imaging and pathology. Neurotoxicology 20:227–238
Paxinos G, Watson C (1982) The rat brain in stereotaxic coordinates. Academic, Sydney
Shamoto-Nagai M, Maruyama W, Yi H, Akao Y, Tribl F, Gerlach M, Osawa T, Riederer P, Naoi M, (2005) Neuromelanin induces oxidative stress in mitochondria through release of iron: mechanism behind the inhibition of 26S proteasome. J Neural Transm 113:633–644
Shoham S, Youdim MB (2000) Iron involvement in neural damage and microgliosis in models of neurodegenerative diseases. Cell Mol Biol (Noisy-le-grand) 46:743–760
Sokolowski AL, Larsson BS, Lindquist NG, (1990) Distribution of 1-(3H)-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (3H-MPTP) in the frog: uptake in neuromelanin. Pharmacol Toxicol 66:252–258
Sulzer D, Bogulavsky J, Larsen KE, Behr G, Karatekin E, Kleinman MH, Turro N, Krantz D, Edwards RH, Greene LA, Zecca L (2000) Neuromelanin biosynthesis is driven by excess cytosolic catecholamines not accumulated by synaptic vesicles. Proc Natl Acad Sci U S A 97:11869–11874
Wilms H, Rosenstiel P, Sievers J, Deuschl G, Zecca L, Lucius R (2003) Activation of microglia by human neuromelanin is NF-kB dependent and involves p38 mitogen-activated protein kinase: implications for Parkinson's disease. FASEB J 17:500–502
Wu DC, Vila M, Tieu K, Teismann P, Vadseth C, Choi DK, Ischiropoulos H, Przedborski S (2002) Blockade of microglial activation is neuroprotective in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson disease. J Neurosci 22:1763–1771
Zecca L, Costi P, Mecacci C, Ito S, Terreni M, Sonnino S (2000) Interaction of human substantia nigra neuromelanin with lipids and peptides. J Neurochem 74:1758–1765
Zecca L, Fariello R, Riederer P, Sulzer D, Gatti A, Tampellini D (2002) The absolute concentration of nigral neuromelanin, assayed by a new sensitive method, increases throughout the life and is dramatically decreased in Parkinson's disease. FEBS Lett 510:216–220
Zecca L, Shima T, Stroppolo A, Goj C, Battiston GA, Gerbasi R, Sarna T, Swartz HM (1996) Interaction of neuromelanin and iron in substantia nigra and other areas of human brain. Neuroscience 73:407–415
Zecca L, Stroppolo A, Gatti A, Tampellini D, Toscani M, Gallorini M, Giaveri G, Arosio P, Santambrogio P, Fariello RG, Karatekin E, Kleinman MH, Turro N, Hornykiewicz O, Zucca FA (2004) The role of iron and copper molecules in the neuronal vulnerability of locus coeruleus and substantia nigra during aging. Proc Natl Acad Sci U S A 101:9843–9848
Zhang W, Shin EJ, Wang T, Lee PH, Pang H, Wie MB, Kim WK, Kim SJ, Huang WH, Wang Y, Zhang W, Hong JS, Kim HC (2006) 3-Hydroxymorphinan, a metabolite of dextromethorphan, protects nigrostriatal pathway against MPTP-elicited damage both in vivo and in vitro. FASEB J 20:2496–2511
Zucca FA, Giaveri G, Gallorini M, Albertini A, Toscani M, Pezzoli G, Lucius R, Wilms H, Sulzer D, Ito S, Waakamatsu K, Zecca L (2004) The neuromelanin of human substantia nigra: physiological and pathogenic aspects. Pigment Cell Res 17:610–617
Acknowledgments
The authors would like to thank M. Kölln and G. Jopp for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Luigi Zecca and Henrik Wilms contributed equally to this work.
This work was supported by grants from the Michael J. Fox Foundation (New York, NY, USA), the Hensel-Foundation, University of Kiel (to H.W. and R.L.) and a generous donation from the “Herbert und Inge Lampe Stiftung” (to H.W. and R.L.). L.Z., M.L.P. and F.A.Z thank for support the MIUR-FIRB project on Protein Folding and Aggregation: Metal and Biomolecules in Protein Conformational Diseases.
Rights and permissions
About this article
Cite this article
Zecca, L., Wilms, H., Geick, S. et al. Human neuromelanin induces neuroinflammation and neurodegeneration in the rat substantia nigra: implications for Parkinson's disease. Acta Neuropathol 116, 47–55 (2008). https://doi.org/10.1007/s00401-008-0361-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-008-0361-7