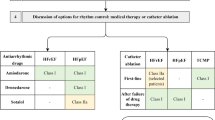
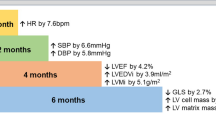
Summary
Sudden cardiac death accounts for 100,000 victims in Germany per year. Predominantely, patients with structural heart disease such as coronary artery disease or dilated cardiomyopathy are affected. However, approximately 5–10% of sudden deaths hit patients without structural disease of the heart. The proportion of young patients (< 40 years of age) in this group is even higher (10–20%). In younger patients significantly more diseases like hypertrophic cardiomyopathy, arrhythmogenic right ventricular dysplasia and primary electrical diseases of the heart could be observed such as long QT syndrome, short QT syndrome, Brugada syndrome and catecholaminergic polymorphic ventricular tachycardia. The primary electrical diseases are different concerning their electrocardiographical pattern, clinical triggers of arrhythmias, results of invasive diagnostics and therapy. Meanwhile, molecular genetic screening can reveal specific mutations of ion channels and can identify consecutive functional defects. The significance of programmed ventricular stimulation is at present unclear concerning risk stratification in patients with Brugada syndrome and short QT syndrome and of no significance in long QTsyndrome and catecholaminergic polymorphic ventricular tachycardias. The implantable cardioverter defibrillator is the therapy of choice in most symptomatic patients. With increasing knowledge as a result of sophisticated molecular genetic screening, identification of underlying ion channel defects and new details of the mechanisms of arrhythmogenesis, a potential genotype-guided therapy will gain more importance in the future.
Zusammenfassung
In Deutschland sterben pro Jahr mehr als 100 000 Menschen an einem plötzlichen Herztod. Bei einer Großzahl der verstorbenen Personen liegt ursächlich eine strukturelle Herzerkrankung wie beispielsweise eine koronare Herzerkrankung oder eine dilatative Kardiomyopathie vor. Etwa 5–10% der plötzlichen Herztodesfälle betreffen allerdings strukturell völlig herzgesunde Personen. Der Anteil jüngerer Menschen (< 40 Lebensjahre) beträgt in dieser Gruppe sogar 10–20%. Bei älteren Personen sind koronare Herzerkrankung und dilatative Kardiomyopathie für den überwiegenden Teil der plötzlichen Herztodesfälle verantwortlich. Bei jüngeren Patienten sind neben strukturellen Erkrankungen des Herzmuskels primär elektrische Erkrankungen des Herzens als wesentliche Differentialdiagnose in Betracht zu ziehen. Dazu gehören Long QT-Syndrom, Short QT-Syndrom, Brugada-Syndrom und die katecholaminerge polymorphe ventrikuläre Tachykardie. Diese Erkrankungen unterscheiden sich elektrokardiographisch, teilweise durch spezifische klinische Trigger der Arrhythmieentstehung, in der invasiven Diagnostik und im therapeutischen Vorgehen. Molekulargenetische Untersuchungen konnten bei einem Teil der jeweiligen Erkrankungen Mutationen spezifischer Ionenkanäle aufdecken. Der Stellenwert der programmierten Kammerstimulation in der Risikostratifizierung der Patienten wird derzeit beim Brugada-Syndrom diskutiert und könnte beim Short QT-Syndrom einen Beitrag leisten. Bei den anderen Erkrankungen ist sie obsolet. Der implantierbare Kardioverter- Defibrillator steht bei der Therapie der Erkrankungen in der Verhinderung des plötzlichen Herztodes an erster Stelle. Durch die zunehmenden Kenntnisse im Rahmen molekulargenetischer Untersuchungen mit Aufdeckung sowohl der zugrundeliegenden spezifischen Ionenkanaldefekte als auch der Mechanismen der Arrhythmogenese gewinnt eine medikamentöse Genotyp-spezifische Therapie an Bedeutung.
Similar content being viewed by others
References
Wever EF, Robles de Medina EO (2004) Sudden death in patients without structural heart disease. J Am Coll Cardiol 43:1137–1144
Moss AJ, Kass RS (2005) Long QT syndrome: from channels to cardiac arrhythmias. J Clin Invest 115:2018–2024
Gussak I, Brugada P, Brugada J, Wright RS, Kopecky SL, Chaitman BR, Bjerregaard P (2000) Idiopathic short QT interval: a new clinical syndrome? Cardiology 94:99–102
Gaita F, Giustetto C, Bianchi F, Wolpert C, Schimpf R, Riccardi R, Grossi S, Richiardi E, Borggrefe M (2003) Short QT syndrome: A familial cause of sudden death. Circulation 108:965–970
Wilde AA, Antzelevitch C, Borggrefe M, Brugada J, Brugada R, Brugada P, Corrado D, Hauer RN, Kass RS, Nademanee K, Priori SG, Towbin JA (2002) Proposed diagnostic criteria for the Brugada syndrome: consensus report. Circulation 106:2514–2519
Priori SG, Napolitano C, Memmi M, Colombi B, Drago F, Gasparini M, DeSimone L, Coltorti F, Bloise R, Keegan R, Cruz Filho FE, Vignati G, Benatar A, DeLogu A (2002) Clinical and molecular characterization of patients with catecholaminergic polymorphic ventricular tachycardia. Circulation 106:69–74
Witzenbichler B, Schulze-Bahr E, Haverkamp W, Breithardt G, Sticherling C, Behrens S, Schultheiss HP (2003) 18-year old patient with anti-epileptic therapy and sudden cardiac death. Z Kardiol 92:747–753
Tan HL, Hofman N, van Langen IM, van der Wal AC, Wilde AM (2005) Sudden unexplained death: Heritability and diagnostic yield of cardiological and genetic examination in surviving relatives. Circulation 112:207–213
Antzelevitch C (2005) Modulation of transmural repolarization. Ann NY Acad Sci 1047:314–323
Watanabe N, Kobayashi Y, Tanno K, Miyoshi F, Asano T, Kawamura M, Mikami Y, Adachi T, Ryu S, Miyata A, Katagiri T (2004) Transmural dispersion of repolarization and ventricular tachyarrhythmias. J Electrocardiol 37:191–200
Roden DM (2001) Pharmacogenetics and drug-induced arrhythmias. Cardiovasc Res 50:224–231
Schwartz PJ, Priori SG, Spazzolini C, Moss AJ, Vincent GM, Napolitano C, Denjoy I, Guicheney P, Breithardt G, Keating MT, Towbin JA, Beggs AH, Brink P, Wilde AA, Toivonen L, Zareba W, Robinson JL, Timothy KW, Corfield V, Wattanasirichaigoon D, Corbett C, Haverkamp W, Schulze-Bahr E, Lehmann MH, Schwartz K, Coumel P, Bloise R (2001) Genotypephenotype correlation in the long-QT syndrome: gene-specific triggers for life-threatening arrhythmias. Circulation 103:89–95
Zhang L, Timothy KW, Vincent MG, Lehmann MH, Fox J, Giuli LC, Shen J, Splawski I, Priori SG, Compton SJ, Yanowitz F, Benhorin J, Moss AJ, Schwartz PJ, Robinson JL, Wang Q, Zareba W, Keating MT, Towbin JA, Napolitano C, Medina A (2000) Spectrum of ST-T-wave patterns and repolarization parameters in congential long-QT syndrome: ECG findings identify genotypes. Circulation 102:2849–2855
Takenaka K, Ai T, Shimizu W, Kobori A, Ninomiya T, Otani H, Kubota T, Takaki H, Kamakura S, Horie M (2003) Exercise stress test amplifies genotype-phenotype correlation in the LQT1 and LQT2 forms of the long-QT syndrome. Circulation 107:838–844
Priori SG, Schwartz PJ, Napolitano C, Bloise R, Ronchetti E, Grillo M, Vicentini A, Spazzolini C, Nastoli J, Bottelli G, Folli R, Cappelletti D (2003) Risk stratification in the long QT syndrome. N Engl J Med 348:1866–1874
Shimizu W, Antzelevitch C (1998) Cellular basis for the ECG features of the LQT1 form of the long-QT syndrome: effects of beta-adrenergic agonists and antagonists and sodium channel blockers on transmural dispersion of repolarization and torsade de pointes. Circulation 98:2314–2322
Yamauchi M, Watanabe E, Yasui K, Takeuchi H, Terasawa T, Sawada K, Hishida H, Kodama I (2005) Prevention of ventricular extrasystole by mexiletine in patients with normal QT intervals is associated with a reduction of transmural dispersion of repolarization. Int J Cardiol 103:92–97
Priori SG, Napolitano C, Schwartz PJ, Grillo M, Bloise R, Ronchetti E, Moncalvo C, Tulipani C, Veia A, Bottelli G, Nastoli J (2004) Association of long QT syndrome loci and cardiac events among patients treated with beta-blockers. JAMA 292:1341–1344
Shimizu W, Antzelevitch C (1997) Sodium channel block with mexiletine is effective in reducing dispersion of repolarization and preventing torsade des pointes in LQT2 and LQT3 models of the long-QT syndrome. Circulation 96:2038–2047
Wehrens XH, Lehnart SE, Marks AR (2005) Ryanodine receptor-targeted anti-arrhythmic therapy. Ann NY Acad Sci 1047:366–375
Nam GB, Burashnikov A, Antzelevitch C (2005) Cellular mechanisms underlying the development of catecholaminergic ventricular tachycardia circulation. 111:2727–2733
Sumitomo N, Harada K, Nagashima M, Yasuda T, Nakamura Y, Aragaki Y, Saito A, Kurosaki K, Jouo K, Koujiro M, Konishi S, Matsuoka S, Oono T, Hayakawa S, Miura M, Ushinohama H, Shibata T, Niimura I (2003) Catecholaminergic polymorphic ventricular tachycardia: electrocardiographic characteristics and optimal therapeutic strategies to prevent sudden death. Heart 89:66–70
Swan H, Laitinen P, Kontula K, Toivonen L (2005) Calcium channel antagonism reduces exercise-induced ventricular arrhythmias in catecholaminergic polymorphic ventricular tachycardia patients with RyR2 mutations. J Cardiovasc Electrophysiol 16:162–166
Schimpf R, Wolpert C, Gaita F, Giustetto C, Borggrefe M (2005) Short QT Syndrome. Cardiovasc Res 67:357–366
Brugada R, Hong K, Dumaine R, Cordeiro J, Gaita F, Borggrefe M, Menendez TM, Brugada J, Pollevick GD, Wolpert C, Burashnikov E, Matsuo K, Wu YS, Guerchicoff A, Bianchi F, Giustetto C, Schimpf R, Brugada P, Antzelevitch C (2004) Sudden death associated with short QT syndrome linked to mutations in HERG. Circulation 109:30–35
Bellocq C, van Ginneken AC, Bezzina CR, Alders M, Escande D, Mannens MM, Baro I, Wilde AA (2004) Mutation in the KCNQ1 gene leading to the short QT-interval syndrome. Circulation 109:2394–2397
Extramiana F, Antzelevitch C (2004) Amplified transmural dispersion of repolarization as the basis for arrhythmogenesis in a canine ventricular-wedge model of short-QT syndrome. Circulation 110:3661–3666
Priori SG, Pandit SV, Rivolta I, Berenfeld O, Ronchetti E, Dhamoon A, Napolitano C, Anumonwo J, di Barletta MR, Gudapakkam S, Bosi G, Stramba-Badiale M, Jalife J (2005) A novel form of short QT syndrome (SQT3) is caused by a mutation in the KCNJ2 gene. Circ Res 96:800–807
Schimpf R, Bauersfeld U, Gaita F, Borggrefe M, Wolpert C (2005) Short QT syndrome: Successful prevention of sudden cardiac death in an adolescent by implantable cardioverter defibrillator treatment for primary prophylaxis. Heart Rhythm 2:416–417
Gaita F, Giustetto C, Bianchi F, Schimpf R, Haissaguerre M, Calo L, Brugada R, Antzelevitch C, Borggrefe M, Wolpert C (2004) Short QT syndrome: pharmacological treatment. J Am Coll Cardiol 43:1494–1499
Wolpert C, Schimpf R, Giustetto C, Antzelevitch C, Cordeiro J, Dumaine R, Brugada R, Hong K, Bauersfeld U, Gaita F, Borggrefe M (2005) Further insights into the effect of quinidine in short QT syndrome caused by a mutation in HERG. Journal Cardiovasc Electrophysiol 16:54–58
Antzelevitch C, Brugada P, Borggrefe M, Brugada J, Brugada R, Corrado D, Gussak I, LeMarec H, Nademanee K, Perez Riera AR, Shimizu W, Schulze-Bahr E, Tan H, Wilde A (2005) Brugada syndrome: report of the second consensus conference. Heart Rhythm 4:429–440
Schimpf R, Hsu LF, Veltmann C, Giustetto C, Gaita F, Haissaguerre M, Wolpert C (2005) Routine ECG testing and intravenous drug challenge in patients with syncope, family history of syncope and/or sudden cardiac death should always be performed with additional precordial chest leads to exclude a Brugada syndrome. Circulation 112:375 (abstract)
Brugada J, Brugada R, Brugada P (2003) Determinats of sudden cardiac death in individuals with electrocardiographic pattern of Brugada syndrome and no previous cardiac arrest. Circulation 108:3092–3096
Priori SG, Napolitano C (2005) Should patients with an asymptomatic Brugada electrocardiogram undergo pharmacological and electrophysiological testing? Circulation 112:279–292
Eckardt L, Probst V, Smits JP, Bahr ES, Wolpert C, Schimpf R, Wichter T, Boisseau P, Heinecke A, Breithardt G, Borggrefe M, LeMarec H, Bocker D, Wilde AA (2005) Long-term prognosis of individuals with right precordial ST-segment-elevation Brugada syndrome. Circulation 111:257–263
Antzelevitch C (2001) The Brugada Syndrome: Ionic basis and arrhythmia mechanisms. J Cardiovasc Electrophysiol 12:268–272
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schimpf, R., Kuschyk, J., Veltmann, C. et al. Primär elektrische Herzerkrankungen im Erwachsenenalter—. Herzschr. Elektrophys. 16, 250–259 (2005). https://doi.org/10.1007/s00399-005-0492-7
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00399-005-0492-7