Abstract
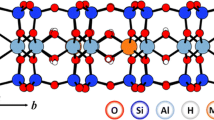
The adsorption of nitrobenzene, 2-chlorophenol, and 4-chlorophenol from water on organophilized derivatives of n-hexadecylpyridinium-montmorillonite (HDP-montmorillonite) was studied. Adsorption excess isotherms were obtained by the immersion method and were analyzed to determine the adsorption capacity of organic pollutants on the hydrophobized surface. The basal spacings of the clay minerals were determined by X-ray diffraction. The results of X-ray diffraction measurements are in good agreement with excess isotherms: whenever a region of the isotherm indicates an increase for the adsorption of organic component, an increase in basal spacing (interlamellar swelling) is also observed. By combining these two independent methods, composition and structure of the interlamellar space between the silicate layers could be accurately calculated. The free enthalpy of adsorption, the adsorption capacity, and the separation factor for adsorption are calculated by analyzing the adsorption isotherm on the basis of the Gibbs equation and Everett-Schay method. The results are utilizable for planning environmental procedures and systems (water clarification and soil remediation).
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 17 July 2000 Accepted: 5 October 2000
Rights and permissions
About this article
Cite this article
Farkas, A., Dékány, I. Interlamellar adsorption of organic pollutants in hydrophobic montmorillonite. Colloid Polym Sci 279, 459–467 (2001). https://doi.org/10.1007/s003960000442
Issue Date:
DOI: https://doi.org/10.1007/s003960000442