Abstract
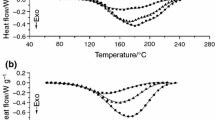
Enthalpy relaxation in a system containing the diglycidyl ether of bisphenol A (DGEBA) resin and a diamine, 1,3-bisaminomethylcyclohexane (1,3-BAC) as curing agent, has been investigated by differential scanning calorimetry (DSC). Samples fully cured were annealed at temperature Tg–15 °C for periods of time from 1 h to a maximum of 168 h. The enthalpy relaxation is analyzed by the peak shift method, in which the sample is heated at 10 °C/min following cooling at various rates through the glass transition region. The key parameters of structural relaxation determined were the non-linearity parameter x=0.47 ± 0.02, the apparent activation energy Δh*=1264 ± 48 kJ/mol or Δh*/R=152 ± 6 kK and the non-exponentiality parameter β ≈ 0.3. The results, obtained by the same method, were compared with those for other systems based on fully cured DGEBA. The correlations among these parameters with the peak shift model should be considered with caution. However, the results show that a correlation between crosslink lengths and the value of Δh* can be considered. The relaxation process for DGEBA/1,3-BAC proves to be highly cooperative.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 28 June 2000 Accepted: 6 September 2000
Rights and permissions
About this article
Cite this article
Ramírez, C., Abad, M., Cano, J. et al. Enthalpy relaxation in an epoxy-cycloaliphatic amine resin. Colloid Polym Sci 279, 184–189 (2001). https://doi.org/10.1007/s003960000419
Issue Date:
DOI: https://doi.org/10.1007/s003960000419