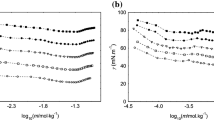
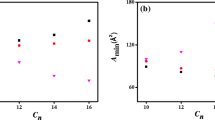
Abstract
Although ionic liquids have been studied as self-assembly media, the aggregation of fluorinated surfactants in ionic liquids has not been fully understood. In this work, the aggregation behavior of the fluorinated surfactant, N-ethylperfluorooctane sulfonamide (N-EtFOSA), in the aprotic ionic liquid, 1-ethyl-3-methylimidazolium ethylsulfate ([C2mim][EtSO4]), was studied using surface tension, 19F-NMR, dynamic light scattering (DLS), and cryo transmission electron microscopy (cryo-TEM). The surface properties and thermodynamic parameters were calculated based on the results of surface tension measurements. It was found that the fluorinated surfactant exhibits a considerable surface activity in aprotic ionic liquids, and its critical micelle concentration (cmc) increases with increasing temperature. In addition, the surface properties indicate that solvophobic interactions are the main driving force for micelle formation. The thermodynamic parameters for micelle formation derived from the temperature dependence of the cmc revealed that micelle formation is an enthalpy-driven process. DLS and cryo-TEM results indicated the presence of nanoscale micelles in the N-EtFOSA + [C2mim][EtSO4] system. The results of this work imply that this fluorinated surfactant exhibits a more pronounced surface activity than conventional hydrocarbon surfactants in [C2mim][EtSO4].

The self-assembly of fluorinated surfactants in aprotic ionic liquids






Similar content being viewed by others
References
Hao J, Zemb T (2007) Self-assembled structures and chemical reactions in room-temperature ionic liquids. Curr Opin Colloid Interface Sci 12:129–137
Greaves TL, Drummond CJ (2008) Ionic liquids as amphiphile self-assembly media. Chem Soc Rev 37:1709–1726
Tariq M, Freire MG, Saramago B, Coutinho JAP, Lopes JNC, Rebelo LPN (2012) Surface tension of ionic liquids and ionic liquid solutions. Chem Soc Rev 41:829–868
Greaves TL, Drummond CJ (2015) Protic ionic liquids: evolving structure-properties relationships and expanding applications. Chem Rev 115:11379–11448
Paul BK, Moulik SP (2015) Ionic liquid-based surfactant science: formulation, characterization, and applications. Wiley-VCH, Weinheim
Shi Y, Xiong D, Wang H, Zhao Y, Wang J (2016) Reversible switching of amphiphilic self-assemblies of ionic liquids between micelle and vesicle by CO2. Langmuir 32:6895–6901
Gu Y, Shi L, Cheng X, Lu F, Zheng L (2013) Aggregation behavior of 1-dodecyl-3-methylimidazolium bromide in aqueous solution: effect of ionic liquids with aromatic anions. Langmuir 29:6213–6220
More UU, Vaid ZS, Rajput SM, Malek NI, El Seoud OA (2017) Effects of 1-alkyl-3-methylimidazolium bromide ionic liquids on the micellar properties of [butanediyl-1,4-bis(dimethyldodecylammonium bromide)] gemini surfactant in aqueous solution. Colloid Polym Sci 295(12):2351–2361
Wang H, Zhang L, Wang J, Li Z, Zhang S (2013) The first evidence for unilamellar vesicle formation of ionic liquids in aqueous solutions. Chem Commun 49:5222–5224
Rao KS, Gehlot PS, Gupta H, Drechsler M, Kumar A (2015) Sodium bromide induced micelle to vesicle transitions of newly synthesized anionic surface active ionic liquids based on dodecylbenzenesulfonate. J Phys Chem B 119:4263–4274
Singh G, Kaur M, Drechsler M, Kang TS (2018) Unprecedented self-assembled architectures of surface-active ionic liquids in aqueous medium. Chem Commun 54:2432–2435
Greaves TL, Drummond CJ (2013) Solvent nanostructure, the solvophobic effect and amphiphile self-assembly in ionic liquids. Chem Soc Rev 42:1096–1120
Chen LG, Bermudez H (2012) Solubility and aggregation of charged surfactants in ionic liquids. Langmuir 28:1157–1162
López-Barrón CR, Li D, DeRita L, Basavaraj MG, Wagner NJW (2012) Spontaneous thermoreversible formation of cationic vesicles in a protic ionic liquid. J Am Chem Soc 134:20728–20732
Dolan A, Atkin R, Warr GG (2015) The origin of surfactant amphiphilicity and self-assembly in protic ionic liquids. Chem Sci 6:6189–6198
Thater JC, Stubenrauch C, Glatter O, Klemmer H, Sottmann T (2019) Microstructure of ionic liquid (EAN)-rich and oil-rich microemulsions studied by SANS. Phys Chem Chem Phys 21:160–170
Pei Y, Ru J, Yao K, Hao L, Li Z, Wang H, Zhu X, Wang J (2018) Nanoreactors stable up to 200°C: a class of high temperature microemulsions composed solely of ionic liquids. Chem Commun 54:6260–6263
Anderson JL, Pino V, Hagberg EC, Sheares VV, Armstrong DW (2003) Surfactant solvation effects and micelle formation in ionic liquids. Chem Commun 19:2444–2445
Fletcher KA, Pandey S (2004) Surfactant aggregation within room-temperature ionic liquid 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide. Langmuir 20:33–36
Patrascu C, Gauffre F, Nallet F, Bordes R, Oberdisse J, de Lauth-Viguerie N, Mingotaud C (2006) Micelles in ionic liquids: aggregation behavior of alkyl poly (ethyleneglycol)-ethers in 1-butyl-3-methyl-imidazolium type ionic liquids. ChemPhysChem 7:99–101
Inoue T, Maema K (2011) Self-aggregation of nonionic surfactants in imidazolium-based ionic liquids with trifluoromethanesulfonate anion. Colloid Polym Sci 289:1167–1175
Li N, Zhang SH, Zheng LQ, Dong B, Li XW, Yu L (2008) Aggregation behavior of long-chain ionic liquids in an ionic liquid. Phys Chem Chem Phys 10:4375–4377
Lindstrom AB, Strynar MJ, Libelo EL (2011) Polyfluorinated compounds: past, present, and future. Environ Sci Technol 45:7954–7961
Dichiarante V, Milani R, Metrangolo P (2018) Natural surfactants towards a more sustainable fluorine chemistry. Green Chem 20:13–27
Kissa E (2001) Fluorinated surfactants and repellents2nd edn. Marcel Dekker, Inc., New York
Li N, Zhang SH, Zheng LQ, Inoue T, Wu JP, Li XW, Yu L (2008) Aggregation behavior of a fluorinated surfactant in 1-butyl-3-methylimidazolium ionic liquids. J Phys Chem B 112:12453–12460
Li N, Zhang SH, Zheng LQ, Inoue T (2009) Aggregation behavior of a fluorinated surfactant in 1-butyl-3-methylimidazalium bis(trifluoromethylsulfonyl)imide ionic liquids. Langmuir 25:10473–10482
Long PF, Chen JF, Wang D, Hu ZQ, Gao XD, Li ZR, Hao JC (2012) Influence of counterions on micellization of tetramethylammonium perfluorononanoic carboxylate in 1-butyl-3-methylimidazolium ionic liquid. J Phys Chem B 116:7669–7675
Rojas O, Tiersch B, Rabe C, Stehle R, Hoell A, Arlt B, Koetz J (2013) Nonaqueous microemulsions based on N,N′-Alkylimidazolium alkylsulfate ionic liquids. Langmuir 29:6833–6839
Davis JH, Fox PA (2003) From curiosities to commodities: ionic liquids begin the transition. Chem Commun:1209–1212.
Li G, Gao Y, Li X, Liu J, Zheng L, Xing H, Xiao J (2010) Aggregation behavior of N-alkyl perfluorooctanesulfonamides in dimethyl sulfoxide solution. J Colloid Interface Sci 342:372–381
Wang X, Long P, Dong S, Hao J (2013) First fluorinated zwitterionic micelle with unusually slow exchange in an ionic liquid. Langmuir 29:14380–14385
Yue X, Chen X, Li Q (2012) Comparison of aggregation behaviors of a phytosterol ethoxylate surfactant in protic and aprotic ionic liquids. J Phys Chem 116:9439–9444
Holmberg K, Jonsson B, Kronberg B, Lindman B (2003) Surfactants and polymers in aqueous solution2nd edn. Wiley, Chichester
Rosen MJ, Kunjappu JT (2012) Surfactants and interfacial phenomena. Wiley-VCH, Weinheim
Shimizu S, Pires PAR, El Seoud OA (2004) Thermodynamics of micellization of benzyl(2-acylaminoethyl)dimethylammonium chloride surfactants in aqueous solutions: a conductivity and titration calorimetry study. Langmuir 20:9551–9559
Zhao Y, Gao S, Wang J, Tang J (2008) Aggregation of ionic liquids [Cnmim]Br (n ) 4, 6, 8, 10, 12) in D2O: a NMR study. J Phys Chem B 112:2031–2039
Funding
The authors are grateful to the financial support from the “Traditional Chinese Veterinary Medicine New Preparation Creation” Scientific and Technological Innovation Team Project of Henan University of Animal Husbandry and Economy (HUAHE2015009), the “Herbal Veterinary Medicines Pharmacognosy” Key Cultivate Discipline Team Project of Henan University of Animal Husbandry and Economy (41000003), and Key Scientific and Technological Project of Henan Provincial Science and Technology Department (162102310434).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 6818 kb).
Rights and permissions
About this article
Cite this article
Li, Y., Lian, R., Wang, X. et al. The aggregation behavior of fluorinated surfactant in an ionic liquid. Colloid Polym Sci 298, 1013–1021 (2020). https://doi.org/10.1007/s00396-020-04650-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-020-04650-3