Abstract
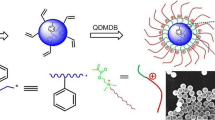
A two-stage synthesis of surface-functionalized monodisperse polystyrene (PS) microspheres in ethanol/water medium by introducing methacryloyloxyethyl dodecyl dimethyl ammonium bromide (QDMDB) to dispersion polymerization of styrene is reported. Polyvinylpyrrolidone (PVP) and fatty alcohol polyoxyethylene ether (AEO-9) are used as dispersion stabilizers. The novel microspheres are characterized by FT-IR, SEM, GPC, TGA, water contact angle (WCA), etc. The dispersion polymerization goes through successfully with the solid content up to 58.75%, in which the water content in the medium is 20%. The water content in the medium has a certain effect on the particle size and molecular weight of microspheres. Under the experimental conditions, the average diameters of the obtained microspheres are approximately 550–1200 nm with the number average molecular weight of up to 5.8 × 105. The introduction of QDMDB improves the wettability and thermal stability of the poly(St-co-QDMDB) microspheres. A mechanism of dispersion copolymerization based on XPS and 1H NMR is proposed.

ᅟ












Similar content being viewed by others
References
Zhang F, Cao L, Yang W (2010) Preparation of monodisperse and anion-charged polystyrene microspheres stabilized with polymerizable sodium styrene sulfonate by dispersion polymerization. Macromol Chem Phys 211:744–751
Lok KP, Ober CK (1985) Particle size control in dispersion polymerization of polystyrene. Can J Chem 63:209–216
Cho YS, Shin CH, Han S (2016) Dispersion polymerization of polystyrene particles using alcohol as reaction medium. Nanoscale Res Lett 11:46
Sponchioni M, Palmiero UC, Moscatelli D (2017) HPMA-PEG surfmers and their use in stabilizing fully biodegradable polymer nanoparticles. Macromol Chem Phys 218:1700380
Schurig V, Grosenick H (1994) Preparative enantiomer separation of enflurane and isoflurane by inclusion gas chromatography. J Chromatogr A 666:617–625
Davis SS, Illum L (1988) Polymeric microspheres as drug carriers. Biomaterials 9:111–115
Jia H, Zhu G, Wang P (2003) Catalytic behaviors of enzymes attached to nanoparticles: the effect of particle mobility. Biotechnol Bioeng 84:406–414
Lin J, Chen XY, Chen CY, Hu JT, Zhou CL, Cai XF, Wang W, Zheng C, Zhang PP, Cheng J, Guo ZH, Liu H (2018) Durably antibacterial and bacterially antiadhesive cotton fabrics coated by cationic fluorinated polymers. ACS Appl Mater Interfaces 10:6124–6136
Callow JA, Callow ME (2011) Trends in the development of environmentally friendly fouling-resistant marine coatings. Nat Commun 2:244
Fu Y, Jiang J, Zhang Q, Zhan X, Chen F (2017) Robust liquid-repellent coatings based on polymer nanoparticles with excellent self-cleaning and antibacterial performances. J Mater Chem A 5:275–284
Zhou H, Li F, Weir MD, Xu HHK (2013) Dental plaque microcosm response to bonding agents containing quaternary ammonium methacrylates with different chain lengths and charge densities. J Dent 41:1122–1131
Yang X, Liu L, Yang W (2012) Direct preparation of monodisperse core-shell microspheres with surface antibacterial property by using bicationic viologen surfmer. polymer 53:2190–2196
Liang K, Liu Q, Peng M (2015) Negatively charged microspheres from stabilizer-free dispersion copolymerization of styrene and hydroxyethyl acrylate. J Polym Mater 32:211–224
Zhang F, Bai Y, Ma Y, Yang W (2009) Preparing of monodisperse and cation-charged polystyrene particles stabilized with polymerizable quarternary ammonium by dispersion polymerization in a methanol-water medium. J Colloid Interface Sci 334:13–21
Tang Y, Xu J, Liu W, Xu L, Li H (2016) Preparation of monodispersed core-shell microspheres with surface antibacterial property employing N-(4-vinylbenzyl)-N,N-diethylamine hydrochloride as surfmer. Int J Polym Mater Polym Biomater 65:143–150
Albernaz VL, Bach M, Weber A, Southan A, Tovar GEM (2018) Active ester containing surfmer for one-stage polymer nanoparticle surface functionalization in mini-emulsion polymerization. Polymer 10:408
He J, Söderling E, Österblad M, Vallittu PK, Lassila LV (2011) Synthesis of methacrylate monomers with antibacterial effects against S. mutans. Molecules 16:9755–9763
Antonucci JM, Zeiger DN, Tang K, Lin-Gibson S, Fowler BO, Lin NJ (2012) Synthesis and characterization of dimethacrylates containing quaternary ammonium functionalities for dental applications. Dent Mater 28:219–228
Vidal ML, Rego GF, Viana GM, Cabral LM, Souza JPB, Silikas N, Schneider LF, Cavalcante LM (2018) Physical and chemical properties of model composites containing quaternary ammonium methacrylates. Dent Mater 34:143–151
Paine AJ, Luymes W, McNulty J (1990) Dispersion polymerization of styrene in polar solvents. 6. Influence of reaction parameters on particle size and molecular weight in poly(iV-vinylpyrrolidone)-stabilized reactions. Macromolecules 23:3104–3109
Sáenz JM, Saenz JMA (1998) Kinetics of the dispersion copolymerization of styrene and butyl acrylate. Macromolecules 31:5215–5222
Vengatesan S, Santhi S, Jeevanantham S, Sozhan G (2015) Quaternized poly (styrene-co-vinylbenzyl chloride) anion exchange membranes for alkaline water electrolysers. J Power Sources 284:361–368
Ma Y, Dai J, Wu L, Fang G, Guo Z (2017) Enhanced anti-ultraviolet, anti-fouling and anti-bacterial polyelectrolyte membrane of polystyrene grafted with trimethyl quaternary ammonium salt modified lignin. Polymer 114:113–121
Acknowledgements
We would like to thank the Analytical & Testing Center of Sichuan University for the SEM measurements. Thanks to Yongshuai Xie for testing FT-IR spectra.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lü, S., Jiang, W. & Li, J. Preparation of surface-modified monodisperse polystyrene microspheres with cationic functional comonomer. Colloid Polym Sci 297, 165–175 (2019). https://doi.org/10.1007/s00396-018-4452-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-018-4452-2