Abstract
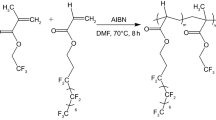
Amphiphilic fluorocopolymer P (PEGMA)-co-P (DFHM) is prepared by hydrophobic dodecafluorohrptyl methacrylate (DFHM) and hydrophilic poly (ethylene glycol) methyl ether methacrylate (PEGMA) monomers via atom transfer radical polymerization (ATRP). The self-assembled colloids of P (PEGMA)-co-P (DFHM) in ethanol, dimethylformamide (DMF), tetrahydrofuran (THF), and chloroform (CHCl3) solutions are discussed according to solvent dielectric constants (ε = 38.3–4.8). The hydrophilic/hydrophobic property, protein resistance, and adhesive strength of films formed by these colloids are evaluated based on the surface morphologies and chemical compositions. P (PEGMA)-co-P (DFHM) can self-assembly into core-shell micelles composed of P (PEGMA) shell and P (DFHM) core (~130 nm) as 500–600 nm in DMF, 300–400 nm in ethanol, and 200–250 nm in THF and CHCl3 solutions. These colloids provide the film surfaces with high fluorine content (39.40–41.70 wt%) to indicate the migration of fluorine-containing groups onto the film surface during the film formation and therefore the obvious hydrophilic/hydrophobic properties (94.3°–102.4° water contact angles and 58.5°–60.2° cetane contact angles). Comparatively, DMF-casted film gains the highest surface roughness (Ra = 0.98 nm), the lowest fluorine content (39.40 wt%), and the highest surface free energy (20.41 mN m−1) due to its large size of colloids, but CHCl3-casted film gives the lowest surface free energy (17.82 mN m−1). The adhesive strength for DMF- and ethanol-casted films (210 and 168 N) proves higher than THF- and CHCl3-casted films (105 and 63 N), while DMF-casted film shows much better protein resistance (Δf = −15.0 Hz) than other three films (Δf = −17–20 Hz). It is believed that the obtained P (P EGMA)-co-P(DFHM) could have a promising application as solvent-dependent and protein-resistance coatings.









Similar content being viewed by others
References
Ji XF, Dong SY, Wei PF, Xia DY, Huang FH (2013) A novel diblock copolymer with a supramolecular polymer block and a traditional polymer block: preparation, controllable self-assembly in water, and application in controlled release. Adv Mater 25:5725–5729
Tokarczyk KK, Junginger M, Belegrinou S, Taubert A (2010) Amphiphilic polymers at interfaces. Adv Polym Sci 242:151–201
He WN, Zhou B, Xu JT, Du BY, Fan ZQ (2012) Two growth modes of semicrystalline cylindrical poly (ε-caprolactone)-b-poly (ethylene oxide) micelles. Macromolecules 45:9768–9778
Garofalo C, Capuano G, Sottile R, Tallerico R, Adami R, Reverchon E, Carbone E, Izzo L, Pappalardo D (2014) Different insight into amphiphilic pEG-PLA copolymers: influence of macromolecular architecture on the micelle formation and cellular uptake. Biomacromolecules 15(1):403–415
Ray JG, Naik SS, Hoff EA, Johnson AJ, Ly JT, Easterling CP, Patton DL, Savin DA (2012) Stimuli-responsive peptide-based ABA-triblock copolymers: unique morphology transitions with Ph. Macromol Rapid Commun 33:819–826
Zhang J, Chen XF, Wei HB, Wan XH (2013a) Tunable assembly of amphiphilic rod–coil block copolymers in solution. Chem Soc Rev 42:9127–9154
Vera F, Torrent MM, Avci C, Arbiol J, Esquena J, Rovira C, Veciana J (2013) Robust molecular micro-capsules for encapsulating and releasing hydrophilic contents. Chem Commun 49:7827–7829
Zhang L, Feng Y, Tian H, Zhao M, Khan M, Guo J (2013b) Amphiphilic depsipeptide-based block copolymers as nanocarriers for controlled release of ibuprofen with soxorubicin. J Polym Sci Part A 51:3213–3226
Bensaid F, Thillaye BO, Amgoune A, Pradel C, Harivardhan RL, Didier E, Sable S, Louit G, Bazile D, Bourissou D (2013) Y-shaped mPEG-PLA cabazitaxel conjugates: well-controlled synthesis by organocatalytic approach and self-assembly into interface drug-loaded core−corona nanoparticles. Biomacromolecules 14:1189–1198
Feng C, Lu G, Li Y, Huang X (2013) Self-assembly of amphiphilic homopolymers bearing ferrocene and carboxyl functionalities: effect of polymer concentration, β-cyclodextrin and length of alkyl linker. Langmuir 29:10922–10931
Huang W, Wang Y, Zhang S, Huang L, Hua D, Zhu X (2013) A facile approach for controlled modification of chitosan under γ-ray irradiation for drug delivery. Macromolecules 46:814–818
Hatton FL, Chambon P, McDonald TO, Owen A, Rannard SP (2014) Hyperbranched polydendrons: a new controlled macromolecular architecture with self-assembly in water and organic solvents. Chem Sci 5:1844–8153
Ren CH, Liu XW, Jiang X, Sun G, Huang XY (2015) Polyisobutylene-b-poly (NN-diethylacrylamide) well-defined amphiphilic diblock copolymer: synthesis and thermo-responsive phase behavior. Polym Chem 53:1143–1150
Nakabayashi K, Oya H, Mori H (2012) Cross-linked core−shell nanoparticles based on amphiphilic block copolymers by RAFT polymerization and palladium-catalyzed suzuki coupling reaction. Macromolecules 45:3197–3240
Cao CW, Yang K, Wu F, Wei XQ, Lu LC, Cai YL (2010) Thermally induced swellability and acid-liable dynamic properties of microgels of copolymers based on PEGMA and aldehyde-functionalized monomer. Macromolecules 43:9511–9521
Huang C, Neoh KG, Kang ET (2012) Combined ATRP and ‘click’ chemistry for designing stable tumor-targeting superparamagnetic iron oxide nanoparticles. Langmuir 28:563–571
Guo WH, Zhu J, Cheng ZP, Zhang ZB, Zhu XL (2011) Anticoagulant surface of 316 L stainless steel modified by surface-initiated atom transfer radical polymerization. Appl Mater Interfaces 3:1675–1680
Liu JL, He WW, Zhang LF, Zhang ZB, Zhu J, Yuan L (2011) Bifunctional nanoparticles with fluorescence and magnetism via surface-initiated AGET ATRP mediated by an iron catalyst. Langmuir 27:12684–12692
Terashima T, Sugita T, Fukae K, Sawamoto M (2014) Synthesis and single-chain folding of amphiphilic random copolymers in water. Macromolecules 47:589–600
Lutz JF (2008) Polymerization of oligo (ethylene glycol) (meth) acrylates: toward new generations of smart biocompatible materials. J Polym Sci Part A 46:3459–3470
Chen X, Zhang G, Zhang Q, Zhan X, Chen F (2015) Preparation and performance of amphiphilic polyurethane copolymers with capsaicin-mimic and PEG moieties for protein resistance and antibacteria. Ind Eng Chem Fundam 54:3813–3820
Chiag YC, Chang Y, Chen WY, Ruaan RC (2012) Biofouling resistance of ultrafiltration membranes controlled by surface self-assembled coating with PEGylated copolymer. Langmuir 28:1399–1407
Zhan XL, Zhang GF, Zhang QH, Chen FQ (2014) Preparation, surface wetting properties, and protein adsorption resistance of well-defined amphiphilic fluorinated diblock copolymers. Appl Polym Sci 10:41167–41178
Martinelli E, Guazzelli E, Bartoli C, Gazzarri M, Chiellini F, Galli G, Callow ME, Callow JA, Finlay JA, Hill S (2015) Amphiphilic pentablock copolymers and their blends with PDMS for antibiofouling coatings. Polym Chem 53:1213–1225
Prakash R, Kaler KVIS, Papageorgiou DP, Papathanasiou AG (2012) Performance of multilayered fluoropolymer surface coating for DEP surface microfluidic devices microfluid. Nano 13:309–318
Wei ZJ, Liu WL, Xiao CL, Tian D, Fan ZP, Sun XL, Wang XQ (2010) Based on atom transfer radical polymerization method preparation of fluoropolymer superhydrophobic films. Thin Solid Films 518:6972–6976
Sun Y, Liu WQ, Ma ZQ (2013) Synthesis of epoxy-terminated fluoropolymer via ATRP and the properties of epoxy thermosets modified with it. Polym Bull 70:1531–1542
Zhang Y, Qi YH (2015) Synthesis of fluorinated acrylate polymer and preparation, and properties of antifouling coating. J Coat Technol Res 12(1):215–223
Sun Y, Liu WQ (2012) Preparation and properties of an organic–inorganic hybrid materials based on fluorinated block copolymer. J Mater Sci 47:1803–1810
Shim MH, Kim J, Park CH (2014) The effects of surface energy and roughness on the hydrophobicity of woven fabrics. Textile Research J 84(12):1268–1278
Li L, Xiong SD, Wang YX, Song GW, Wu SL, Chu PK, Xu ZS (2011) Interaction between fluorinated amphiphilic copolymer P (HFMA)-g-P (SPEG) and BSA. J Dispers Sci Technol 32:1185–1190
Zhao ZL, Ni HG, Han ZY, Jiang TF, Xu YJ, Lu XL, Ye P (2013) Effect of surface compositional heterogeneities and microphase segregation of fluorinated amphiphilic copolymers on antifouling performance. ACS Appl Mater Interfaces 5:7808–7818
Tan BH, Hussain H, Chaw KC, Dickinson GH, Gudipati CS, Birch WR, Teo SLM, He C, Liu Y, Davis TP (2010) Barnacle repellent nanostructured surfaces formed by the self-assembly of amphiphilic block copolymers. Polym Chem 1:276–279
Fu GD, Phua SJ, Kang ET, Neoh KG (2005) Tadpole-shaped amphiphilic block-graft copolymers prepared via consecutive atom transfer radical polymerizations. Macromolecules 38:2612–2619
Kawata T, Hashidzume A, Sato T (2007) Micellar structure of amphiphilic statistical copolymers bearing dodecyl hydrophobes in aqueous media. Macromolecules 40:1174–1180
Liu XY, Wang YH, Yi CL, Feng Y, Jiang JQ, Cui ZG, Chen MQ (2009) Self-assembly and emulsification behavior of photo-sensitive P (St/CS-alt-MA) copolymer. Acta Chim Sin 67(5):447–452
Dong X, He L, Wang N, Liang JY, Niu MJ, Zhao X (2012) Diblock fluoroacrylate copolymers from two initiators: synthesis, self-assembly and surface properties. J Mater Chem 22:23078–23090
Huang HP, Qu J, He L (2016) Amphiphilic silica/fluoropolymer nanoparticles: synthesis, tem-responsive and surface properties as protein-resistance coatings. J Polym Sci, Part A: Polym Chem 54:381–393
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was funded by the National Natural Science Foundation of China (NSFC Grants 51403171, 5157314, 551373133) and the National Basic Research Program of China (973 Program, No.2012CB720904). The authors also wish to express their gratitude for the MOE Key Laboratory for Non-equilibrium Condensed Matter and Quantum Engineering of Xi’an Jiaotong University.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Guo, H., He, L. Self-assembled colloid and solvent-responsive property of amphiphilic fluoropolymer for protein-resistance coatings. Colloid Polym Sci 295, 827–836 (2017). https://doi.org/10.1007/s00396-017-4065-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-017-4065-1