Abstract
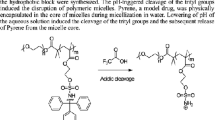
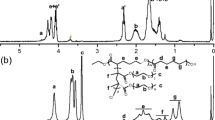
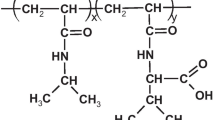
New pH-sensitive graft copolymers based on poly(2-hydroxyethyl aspartamide) (PHEA) were prepared by attaching various cationic monomers, such as 4-(aminomethyl)pyridine (PY), 1-(3-aminopropyl)imidazole (IM), and N-(3-aminopropyl)dibuthylamine (BU), as pH-sensitive units and octadecylamine (C18) as a hydrophobic segment on poly(succinimide). Phase transition of each copolymer solution occurred at a vicinity of the pK a value of the cationic groups, and their insoluble pH ranges were broadened as the feed amount of pH-sensitive moieties was increased. Depending on the cationic grafts having different pK a values, the pH ranges where the copolymer became insoluble could be tuned. Copolymers PHEA-g-C18-PY, PHEA-g-C18-IM, and PHEA-g-C18-BU exhibited phase separations in solutions at pH ranges of 4∼6, 6∼8, and 9∼12, respectively. These polymers have the unique feature of their pH sensitivity profiles being identified to three regimes. Under low pH conditions (below pK a ), the polymer solution is transparent. At medium pH (around pK a ), polymer precipitation occurred in solution. At pH > pK a , the polymer solution is gradually dissolved again.






Similar content being viewed by others
References
Qui Y, Park K (2001) Adv Drug Deliv Rev 53:321
Jeong B, Gutowska A (2002) Trends Biotechnol 20:305
Feng W, Patel SH, Young M-Y, Zunino JL, Xanthos M (2007) Adv Poly Technol 26:1
Zhang H, Ito Y (2002) Smart materials using signal-responsive polyelectrolytes. In: Tripathy SK, Kumar J, Nalwa HS (eds) Handbook of polyelectrolytes and their applications Volume 1: polyelectrolyte-based multilayers, self-assembly and nanostructures. American Scientific Publishers, California, p 183
Sethuraman VA, Lee MC, Bae YH (2008) Pharm Res 25:657
Wang C-H, Wang C-H, Hsiue G-H (2005) J Control Release 108:140
Klee SK, Lersch P (2006) SÖFW-J 132:2
Kang SI, Bae YH (2002) J Control Release 80:145
Jiang T-Y, Wang Z-Y, Tang L-X, Mo F-K, Chen C (2006) J Appl Polym Sci 99:2702
Castelli F, Messina C, Pignatello R, Puglisi G (2001) Drug Deliv 8:173
Bae SK, Kim J-D (2002) J Biomed Mater Res 64:282
Yang SR, Lee HJ, Kim J-D (2006) J Control Release 114:60
Seo K, Kim D (2006) Macromol Biosci 6:758
Kang HS, Yang SR, Kim J-D, Han S-H, Chang I-S (2001) Langmuir 17:7501
Kang HS, Kim J-D, Han S-H, Chang I-S (2002) J Control Release 81:135
Yang SR, Jeong JH, Park K, Kim J-D (2003) Colloid Polym Sci 281:852
Jeong JH, Kang HS, Yang SR, Park K, Kim J-D (2005) Colloid Surf A 264:187
Molyneux BP, Frank HP (1961) J Am Chem Soc 83:3169
Yang SY, Schultz G, Green MM, Morawetz H (1999) Macromolecules 32:2577
Wang Y, Morawetz H (1986) Macromolecules 19:1925
Jang K-S, Lee HJ, Yang H-M, An EJ, Kim T-H, Choi S-M, Kim J-D (2008) Soft Matter 4:349
An EJ (2007) MS thesis, Korea Advanced Institute of Science and Technology, Korea
Kang HS, Shin M-S, Kim J-D, Yang J-W (2000) Polym Bull 45:39
Bell PH, Robin OJ (1942) J Am Chem Soc 64:2905
Gohy J-F, Antoun S, Jérôme R (2001) Macromolecules 34:7435
Khutoryanskiy VV, Mun GA, Nurkeeva ZS, Dubolazov AV (2004) Polym Int 53:1382
Acknowledgements
This work was part of the project of the Human Resource Development for Industrial Demand, and Basic R&D Research Program, financially supported by the Ministry of Education, Science, and Technology (MEST) and a Korea Science and Engineering Foundation (KOSEF) grant (R11-2007-050-03003-0).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, H.W., Jin, HS., Yang, S.Y. et al. Tunable phase transition behaviors of pH-sensitive polyaspartamides having various cationic pendant groups. Colloid Polym Sci 287, 919–926 (2009). https://doi.org/10.1007/s00396-009-2046-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-009-2046-8