Abstract.
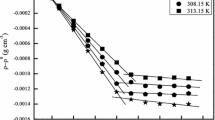
Mixed micelles formed by zwitterionic surfactant dimethyldodecylammniopropane sulfonate and short-chain phospholipid 1,2-diheptanoyl-sn-glycero-3-phosphocholine in different proportions in an aqueous medium have been studied physicochemically at an air/water interface and in the bulk by using interfacial tension and pyrene fluorescence intensity measurements, respectively. The critical micellar concentration and free energies of micellization and of interfacial adsorption have been determined. The interfacial study reveals that a mixed monolayer is formed at the air/water interface by the adsorption of surfactant and phospholipid monomers. This has been confirmed by evaluating the interfacial parameters; the maximum surface excess, the minimum area per molecule of a surface-active compound, and the Gibbs surface excess related to surface pressure. The nonideality of mixing, expressed in the terms of the regular solution interaction parameter, β, has negative values over the whole mole fraction range. The negative β values indicate the mutual synergism between the surfactant and phospholipid monomers. The equilibrium distribution of components between micelle and monomer phases was evaluated using a theoretical treatment based on excess thermodynamics quantities evaluated by Motomura's formulation.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Sehgal, P., Doe, H. & Bakshi, M. Interfacial and micellar properties of binary mixtures of surfactant and phospholipid in an aqueous medium. Colloid Polym Sci 281, 275–282 (2003). https://doi.org/10.1007/s00396-002-0791-z
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00396-002-0791-z