Abstract
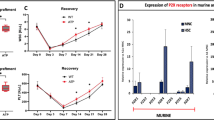
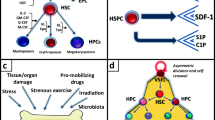
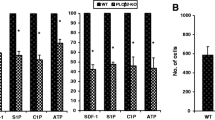
In addition to being a peptidase, the angiotensin-converting enzyme (ACE) can be phosphorylated and involved in signal transduction. We evaluated the role of ACE in granulocyte-colony-stimulating factor (G-CSF)-induced hematopoietic progenitor cell (HPC) mobilization and detected a significant increase in mice-lacking ACE. Transplantation experiments revealed that the loss of ACE in the HPC microenvironment rather than in the HPCs increased mobilization. Indeed, although ACE was expressed by a small population of bone-marrow cells, it was more strongly expressed by endosteal bone. Interestingly, there was a physical association of ACE with the G-CSF receptor (CD114), and G-CSF elicited ACE phosphorylation on Ser1270 in vivo and in vitro. A transgenic mouse expressing a non-phosphorylatable ACE (ACES/A) mutant demonstrated increased G-CSF-induced HPC mobilization and decreased G-CSF-induced phosphorylation of STAT3 and STAT5. These results indicate that ACE expression/phosphorylation in the bone-marrow niche interface negatively regulates G-CSF-induced signaling and HPC mobilization.






Similar content being viewed by others
References
Azizi M, Junot C, Ezan E, Menard J (2001) Angiotensin I-converting enzyme and metabolism of the haematological peptide N-acetyl-seryl-aspartyl-lysyl-proline. Clin Exp Pharmacol Physiol 28:1066–1069. https://doi.org/10.1046/j.1440-1681.2001.03560
Azizi M, Rousseau A, Ezan E, Guyene TT, Michelet S, Grognet JM, Lenfant M, Corvol P, Menard J (1996) Acute angiotensin-converting enzyme inhibition increases the plasma level of the natural stem cell regulator N-acetyl-seryl-aspartyl-lysyl-proline. J Clin Investig 97:839–844. https://doi.org/10.1172/JCI118484
Bernstein KE, Koronyo Y, Salumbides BC, Sheyn J, Pelissier L, Lopes DH, Shah KH, Bernstein EA, Fuchs DT, Yu JJ, Pham M, Black KL, Shen XZ, Fuchs S, Koronyo-Hamaoui M (2014) Angiotensin-converting enzyme overexpression in myelomonocytes prevents Alzheimer’s-like cognitive decline. J Clin Investig 124:1000–1012. https://doi.org/10.1172/JCI66541
Bocchietto E, Guglielmetti A, Silvagno F, Taraboletti G, Pescarmona GP, Mantovani A, Bussolino F (1993) Proliferative and migratory responses of murine microvascular endothelial cells to granulocyte-colony-stimulating factor. J Cell Physiol 155:89–95. https://doi.org/10.1002/jcp.1041550112
Bryant CD, Zhang NN, Sokoloff G, Fanselow MS, Ennes HS, Palmer AA, McRoberts JA (2008) Behavioral differences among C57BL/6 substrains: implications for transgenic and knockout studies. J Neurogenet 22:315–331
Campbell DJ, Alexiou T, Xiao HD, Fuchs S, McKinley MJ, Corvol P, Bernstein KE (2004) Effect of reduced angiotensin-converting enzyme gene expression and angiotensin-converting enzyme inhibition on angiotensin and bradykinin peptide levels in mice. Hypertension 43:854–859. https://doi.org/10.1161/01.HYP.0000119190.06968.f1
Cole J, Ertoy D, Lin H, Sutliff RL, Ezan E, Guyene TT, Capecchi M, Corvol P, Bernstein KE (2000) Lack of angiotensin II-facilitated erythropoiesis causes anemia in angiotensin-converting enzyme-deficient mice. J Clin Investig 106:1391–1398. https://doi.org/10.1172/JCI10557
Cole JM, Xiao H, Adams JW, Disher KM, Zhao H, Bernstein KE (2003) New approaches to genetic manipulation of mice: tissue-specific expression of ACE. Am J Physiol Ren Physiol 284:F599–F607. https://doi.org/10.1152/ajprenal.00308.2002
Donella-Deana A, Cesaro L, Sarno S, Ruzzene M, Brunati AM, Marin O, Vilk G, Doherty-Kirby A, Lajoie G, Litchfield DW, Pinna LA (2003) Tyrosine phosphorylation of protein kinase CK2 by Src-related tyrosine kinases correlates with increased catalytic activity. Biochem J 372:841–849. https://doi.org/10.1042/BJ20021905
Durik M, Seva PB, Roks AJ (2012) The renin–angiotensin system, bone marrow and progenitor cells. Clin Sci (Lond) 123:205–223. https://doi.org/10.1042/CS20110660
Fleming I, Kohlstedt K, Busse R (2005) New fACEs to the renin–angiotensin system. Physiology (Bethesda) 20:91–95. https://doi.org/10.1152/physiol.00003.2005
Garcia P, Schwenzer S, Slotta JE, Scheuer C, Tami AE, Holstein JH, Histing T, Burkhardt M, Pohlemann T, Menger MD (2010) Inhibition of angiotensin-converting enzyme stimulates fracture healing and periosteal callus formation—role of a local renin-angiotensin system. Br J Pharmacol 159:1672–1680. https://doi.org/10.1111/j.1476-5381.2010.00651.x
Gould AB, Goodman SA (1990) Effect of an angiotensin-converting enzyme inhibitor on blood pressure and erythropoiesis in rats. Eur J Pharmacol 181:225–234. https://doi.org/10.1016/0014-2999(90)90082-H
Griffing GT, Melby JC (1982) Enalapril (MK-421) and the white cell count and haematocrit. Lancet 1:1361. https://doi.org/10.1016/S0140-6736(82)92430-8
Hirakata H, Onoyama K, Iseki K, Kumagai H, Fujimi S, Omae T (1984) Worsening of anemia induced by long-term use of captopril in hemodialysis patients. Am J Nephrol 4:355–360. https://doi.org/10.1159/000166851
Hoggatt J, Pelus LM (2011) Mobilization of hematopoietic stem cells from the bone marrow niche to the blood compartment. Stem Cell Res Ther 2:13. https://doi.org/10.1186/scrt54
Hubert C, Savary K, Gasc JM, Corvol P (2006) The hematopoietic system: a new niche for the renin–angiotensin system. Nat Clin Pract Cardiovasc Med 3:80–85. https://doi.org/10.1038/ncpcardio0449
Izu Y, Mizoguchi F, Kawamata A, Hayata T, Nakamoto T, Nakashima K, Inagami T, Ezura Y, Noda M (2009) Angiotensin II type 2 receptor blockade increases bone mass. J Biol Chem 284:4857–4864. https://doi.org/10.1074/jbc.M807610200
Jokubaitis VJ, Sinka L, Driessen R, Whitty G, Haylock DN, Bertoncello I, Smith I, Peault B, Tavian M, Simmons PJ (2008) Angiotensin-converting enzyme (CD143) marks hematopoietic stem cells in human embryonic, fetal, and adult hematopoietic tissues. Blood 111:4055–4063. https://doi.org/10.1182/blood-2007-05-091710
Kamezaki K, Shimoda K, Numata A, Haro T, Kakumitsu H, Yoshie M, Yamamoto M, Takeda K, Matsuda T, Akira S, Ogawa K, Harada M (2005) Roles of Stat3 and ERK in G-CSF signaling. Stem Cells 23:252–263. https://doi.org/10.1634/stemcells.2004-0173a
Kendrick TS, Bogoyevitch MA (2007) Activation of mitogen-activated protein kinase pathways by the granulocyte colony-stimulating factor receptor: mechanisms and functional consequences. Front Biosci 12:591–607. https://doi.org/10.2741/2085
Kohlstedt K, Brandes RP, Muller-Esterl W, Busse R, Fleming I (2004) Angiotensin-converting enzyme is involved in outside-in signaling in endothelial cells. Circ Res 94:60–67. https://doi.org/10.1161/01.RES.0000107195.13573.E4
Kohlstedt K, Busse R, Fleming I (2005) Signaling via the angiotensin-converting enzyme enhances the expression of cyclooxygenase-2 in endothelial cells. Hypertension 45:126–132. https://doi.org/10.1161/01.HYP.0000150159.48992.11
Kohlstedt K, Gershome C, Friedrich M, Muller-Esterl W, Alhenc-Gelas F, Busse R, Fleming I (2006) Angiotensin-converting enzyme (ACE) dimerization is the initial step in the ACE inhibitor-induced ACE signaling cascade in endothelial cells. Mol Pharmacol 69:1725–1732. https://doi.org/10.1124/mol.105.020636
Kohlstedt K, Shoghi F, Muller-Esterl W, Busse R, Fleming I (2002) CK2 phosphorylates the angiotensin-converting enzyme and regulates its retention in the endothelial cell plasma membrane. Circ Res 91:749–756. https://doi.org/10.1161/01.RES.0000038114.17939.C8
Kohlstedt K, Trouvain C, Boettger T, Shi L, Fisslthaler B, Fleming I (2013) AMP-activated protein kinase regulates endothelial cell angiotensin-converting enzyme expression via p53 and the post-transcriptional regulation of microRNA-143/145. Circ Res 112:1150–1158. https://doi.org/10.1161/CIRCRESAHA.113.301282
Lee M, Aoki M, Kondo T, Kobayashi K, Okumura K, Komori K, Murohara T (2005) Therapeutic angiogenesis with intramuscular injection of low-dose recombinant granulocyte-colony stimulating factor. Arterioscler Thromb Vasc Biol 25:2535–2541. https://doi.org/10.1161/01.ATV.0000190609.28293.17
Li J (2013) JAK-STAT and bone metabolism. JAKSTAT 2(3):e23930. https://doi.org/10.4161/jkst.23930
Lin C, Datta V, Okwan-Duodu D, Chen X, Fuchs S, Alsabeh R, Billet S, Bernstein KE, Shen XZ (2010) Angiotensin-converting enzyme is required for normal myelopoiesis. FASEB J 25:1145–1155. https://doi.org/10.1096/fj.10-169433
Nicholson A, Reifsnyder PC, Malcolm RD, Lucas CA, MacGregor GR, Zhang W, Leiter EH (2010) Diet-induced obesity in two C57BL/6 substrains with intact or mutant nicotinamide nucleotide transhydrogenase (Nnt) gene. Obesity 18:1902–1905
Park TS, Zambidis ET (2009) A role for the renin–angiotensin system in hematopoiesis. Haematologica 94:745–747. https://doi.org/10.3324/haematol.2009.006965
Pratt MC, Lewis-Barned NJ, Walker RJ, Bailey RR, Shand BI, Livesey J (1992) Effect of angiotensin converting enzyme inhibitors on erythropoietin concentrations in healthy volunteers. Br J Clin Pharmacol 34:363–365. https://doi.org/10.1111/j.1365-2125.1992.tb05644.x
Richter R, Forssmann W, Henschler R (2017) Current developments in mobilization of hematopoietic stem and progenitor cells and their interaction with niches in bone marrow. Transfus Med Hemother 44:151–164. https://doi.org/10.1159/000477262
Rodgers KE, diZerega GS (2013) Contribution of the local RAS to hematopoietic function: a novel therapeutic target. Front Endocrinol (Lausanne) 4:157. https://doi.org/10.3389/fendo.2013.00157
Rodgers KE, Xiong S, diZerega GS (2002) Accelerated recovery from irradiation injury by angiotensin peptides. Cancer Chemother Pharmacol 49:403–411. https://doi.org/10.1007/s00280-002-0434-6
Rodgers KE, Xiong S, Steer R, diZerega GS (2000) Effect of angiotensin II on hematopoietic progenitor cell proliferation. Stem Cells 18:287–294. https://doi.org/10.1634/stemcells.18-4-287
Rousseau-Plasse A, Lenfant M, Potier P (1996) Catabolism of the hemoregulatory peptide N-acetyl-Ser-Asp-Lys-Pro: a new insight into the physiological role of the angiotensin-I-converting enzyme N-active site. Bioorg Med Chem 4:1113–1119. https://doi.org/10.1016/0968-0896(96)00104-6
Semerad CL, Christopher MJ, Liu F, Short B, Simmons PJ, Winkler I, Levesque JP, Chappel J, Ross FP, Link DC (2005) G-CSF potently inhibits osteoblast activity and CXCL12 mRNA expression in the bone marrow. Blood 106:3020–3027. https://doi.org/10.1182/blood-2004-01-0272
Shen XZ, Bernstein KE (2011) The peptide network regulated by angiotensin converting enzyme (ACE) in hematopoiesis. Cell Cycle 10:1363–1369. https://doi.org/10.4161/cc.10.9.15444
Simon MM, Greenaway S, White JK, Fuchs H, Gailus-Durner V, Wells S, Sorg T, Wong K, Bedu E, Cartwright EJ, Dacquin R, Djebali S, Estabel J, Graw J, Ingham NJ, Jackson IJ, Lengeling A, Mandillo S, Marvel J, Meziane H, Preitner F, Puk O, Roux M, Adams DJ, Atkins S, Ayadi A, Becker L, Blake A, Brooker D, Cater H, Champy MF, Combe R, Danecek P, di Fenza A, Gates H, Gerdin AK, Golini E, Hancock JM, Hans W, Hölter SM, Hough T, Jurdic P, Keane TM, Morgan H, Müller W, Neff F, Nicholson G, Pasche B, Roberson LA, Rozman J, Sanderson M, Santos L, Selloum M, Shannon C, Southwell A, Tocchini-Valentini GP, Vancollie VE, Westerberg H, Wurst W, Zi M, Yalcin B, Ramirez-Solis R, Steel KP, Mallon AM, de Angelis MH, Herault Y, Brown SD (2013) A comparative phenotypic and genomic analysis of C57BL/6J and C57BL/6N mouse strains. Genome Biol 14:R82. https://doi.org/10.1186/gb-2013-14-7-r82
Sinka L, Biasch K, Khazaal I, Peault B, Tavian M (2012) Angiotensin-converting enzyme (CD143) specifies emerging lympho-hematopoietic progenitors in the human embryo. Blood 119:3712–3723. https://doi.org/10.1182/blood-2010-11-314781
Skidgel RA, Engelbrecht S, Johnson AR, Erdos EG (1984) Hydrolysis of substance P and neurotensin by converting enzyme and neutral endopeptidase. Peptides 5:769–776. https://doi.org/10.1016/0196-9781(84)90020-2
Vlahakos DV, Canzanello VJ, Madaio MP, Madias NE (1991) Enalapril-associated anemia in renal transplant recipients treated for hypertension. Am J Kidney Dis 17:199–205. https://doi.org/10.1016/S0272-6386(12)81129-2
Zambidis ET, Park TS, Yu W, Tam A, Levine M, Yuan X, Pryzhkova M, Peault B (2008) Expression of angiotensin-converting enzyme (CD143) identifies and regulates primitive hemangioblasts derived from human pluripotent stem cells. Blood 112:3601–3614. https://doi.org/10.1182/blood-2008-03-144766
Acknowledgements
The authors are indebted to Isabel Winter, Marie von Reutern, Mechthild Piepenbrock, and Katharina Herbig for expert technical assistance. This work was supported by the Deutsche Forschungsgemeinschaft (FL 364/1-3 to I.F., KO 4263/2-1 to K.K., SFB 834/Z1 to R.H., and Exzellenzcluster 147 “Cardio-Pulmonary Systems”). The authors declare no competing financial interests.
Author information
Authors and Affiliations
Contributions
KK planned experiments, interpreted data, and drafted the manuscript. TF was responsible for designing the generation of the transgenic mouse. CT performed experiments and interpreted data. TM performed immunohistochemistry for ACE. RH provided valuable expertise and laboratory facilities for the study of progenitor cell populations. IF planned experiments, interpreted data, and took a lead role in writing the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kohlstedt, K., Trouvain, C., Frömel, T. et al. Role of the angiotensin-converting enzyme in the G-CSF-induced mobilization of progenitor cells. Basic Res Cardiol 113, 18 (2018). https://doi.org/10.1007/s00395-018-0677-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-018-0677-y