Abstract
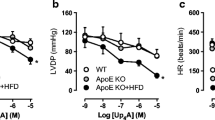
Accelerated development of coronary atherosclerosis is a defining characteristic of familial hypercholesterolemia (FH). However, the recent data highlight a significant cardiovascular risk prior to the development of critical coronary stenosis. We, therefore, examined the hypothesis that FH produces coronary microvascular dysfunction and impairs coronary vascular control at rest and during exercise in a swine model of FH. Coronary vascular responses to drug infusions and exercise were examined in chronically instrumented control and FH swine. FH swine exhibited ~tenfold elevation of plasma cholesterol and diffuse coronary atherosclerosis (20–60 % plaque burden). Similar to our recent findings in the systemic vasculature in FH swine, coronary smooth muscle nitric oxide sensitivity was increased in vivo and in vitro with maintained endothelium-dependent vasodilation in vivo in FH. At rest and during exercise, FH swine exhibited increased myocardial O2 extraction resulting in reduced coronary venous SO2 and PO2 versus control. During exercise in FH swine, the transmural distribution of coronary blood flow was unchanged; however, a shift toward anaerobic cardiac metabolism was revealed by increased coronary arteriovenous H+ concentration gradient. This shift was associated with a worsening of cardiac efficiency (relationship between cardiac work and O2 consumption) in FH during exercise owing, in part, to a generalized reduction in stroke volume which was associated with increased left atrial pressure in FH. Our data highlight a critical role for coronary microvascular dysfunction as a contributor to impaired myocardial O2 balance, cardiac ischemia, and impaired cardiac function prior to the development of critical coronary stenosis in FH.






Similar content being viewed by others
References
Ball RM, Bache RJ (1976) Distribution of myocardial blood flow in the exercising dog with restricted coronary artery inflow. Circ Res 38:60–66. doi:10.1161/01.res.38.2.60
Bender SB, de Beer VJ, Tharp DL, van Deel ED, Bowles DK, Duncker DJ, Laughlin MH, Merkus D (2014) Reduced contribution of endothelin to the regulation of systemic and pulmonary vascular tone in severe familial hypercholesterolaemia. J Physiol 592:1757–1769. doi:10.1113/jphysiol.2013.267351
Bender SB, Tune JD, Borbouse L, Long X, Sturek M, Laughlin MH (2009) Altered mechanism of adenosine-induced coronary arteriolar dilation in early-stage metabolic syndrome. Exp Biol Med (Maywood) 234:683–692. doi:10.3181/0812-RM-350
Bender SB, van Houwelingen MJ, Merkus D, Duncker DJ, Laughlin MH (2010) Quantitative analysis of exercise-induced enhancement of early and late systolic retrograde coronary blood flow. J Appl Physiol 108:507–514. doi:10.1152/japplphysiol.01096.2009
Berwick ZC, Dick GM, Moberly SP, Kohr MC, Sturek M, Tune JD (2012) Contribution of voltage-dependent K+ channels to metabolic control of coronary blood flow. J Mol Cell Cardiol 52:912–919. doi:10.1016/j.yjmcc.2011.07.004
Camici PG, d’Amati G, Rimoldi O (2015) Coronary microvascular dysfunction: mechanisms and functional assessment. Nat Rev Cardiol 12:48–62. doi:10.1038/nrcardio.2014.160
Checovich WJ, Fitch WL, Krauss RM, Smith MP, Rapacz J, Smith CL, Attie AD (1988) Defective catabolism and abnormal composition of low-density lipoproteins from mutant pigs with hypercholesterolemia. Biochemistry 27:1934–1941. doi:10.1021/bi00406a020
de Beer VJ, Merkus D, Bender SB, Tharp DL, Bowles DK, Duncker DJ, Laughlin MH (2013) Familial hypercholesterolemia impairs exercise-induced vasodilation due to reduced NO bioavailability. J Appl Physiol 115:1767–1776. doi:10.1152/japplphysiol.00619.2013
de Ferranti SD, Rodday AM, Mendelson MM, Wong JB, Leslie LK, Sheldrick RC (2016) Prevalence of familial hypercholesterolemia in the 1999–2012 United States National Health and Nutrition Examination Surveys (NHANES). Circulation 133:1067–1072. doi:10.1161/circulationaha.115.018791
Drexler H, Zeiher AM (1991) Endothelial function in human coronary arteries in vivo. Focus on hypercholesterolemia. Hypertension 18:II90–II99. doi:10.1161/01.HYP.18.4_Suppl.II90
Duncker DJ, Bache RJ (2008) Regulation of coronary blood flow during exercise. Physiol Rev 88:1009–1086. doi:10.1152/physrev.00045.2006
Duncker DJ, Beer VJ, Merkus D (2008) Alterations in vasomotor control of coronary resistance vessels in remodelled myocardium of swine with a recent myocardial infarction. Med Biol Eng Comput 46:485–497. doi:10.1007/s11517-008-0315-1
Duncker DJ, Koller A, Merkus D, Canty JM Jr (2015) Regulation of coronary blood flow in health and ischemic heart disease. Prog Cardiovasc Dis 57:409–422. doi:10.1016/j.pcad.2014.12.002
Duncker DJ, Stubenitsky R, Tonino PA, Verdouw PD (2000) Nitric oxide contributes to the regulation of vasomotor tone but does not modulate O2-consumption in exercising swine. Cardiovasc Res 47:738–748. doi:10.1016/S0008-6363(00)00143-7
Duncker DJ, Stubenitsky R, Verdouw PD (1998) Role of adenosine in the regulation of coronary blood flow in swine at rest and during treadmill exercise. Am J Physiol 275:H1663–H1672
Farhi ER, Canty JM, Klocke FJ (1989) Effects of graded reductions in coronary perfusion pressure on the diastolic pressure-segment length relation and the rate of isovolumic relaxation in the resting conscious dog. Circulation 80:1458–1468. doi:10.1161/01.cir.80.5.1458
Fleenor BS, Bowles DK (2009) Exercise training decreases the size and alters the composition of the neointima in a porcine model of percutaneous transluminal coronary angioplasty (PTCA). J Appl Physiol 107:937–945. doi:10.1152/japplphysiol.91444.2008
Fleenor BS, Bowles DK (2009) Negligible contribution of coronary adventitial fibroblasts to neointimal formation following balloon angioplasty in swine. Am J Physiol Heart Circ Physiol 296:H1532–H1539. doi:10.1152/ajpheart.00566.2008
Frisbee JC (2005) Reduced nitric oxide bioavailability contributes to skeletal muscle microvessel rarefaction in the metabolic syndrome. Am J Physiol Regul Integr Comp Physiol 289:R307–R316
Frisbee JC, Goodwill AG, Butcher JT, Olfert IM (2011) Divergence between arterial perfusion and fatigue resistance in skeletal muscle in the metabolic syndrome. Exp Physiol 96:369–383. doi:10.1113/expphysiol.2010.055418
Goldberg AC, Gidding SS (2016) Knowing the prevalence of familial hypercholesterolemia matters. Circulation 133:1054–1057. doi:10.1161/circulationaha.116.021673
Goodwill AG, Frisbee SJ, Stapleton PA, James ME, Frisbee JC (2009) Impact of chronic anticholesterol therapy on development of microvascular rarefaction in the metabolic syndrome. Microcirculation 16:667–684. doi:10.3109/10739680903133722
Guarini G, Kiyooka T, Ohanyan V, Pung YF, Marzilli M, Chen YR, Chen CL, Kang PT, Hardwick JP, Kolz CL, Yin L, Wilson GL, Shokolenko I, Dobson JG, Fenton R, Chilian WM (2016) Impaired coronary metabolic dilation in the metabolic syndrome is linked to mitochondrial dysfunction and mitochondrial DNA damage. Basic Res Cardiol 111:1–13. doi:10.1007/s00395-016-0547-4
Guth BD, Wisneski JA, Neese RA, White FC, Heusch G, Mazer CD, Gertz EW (1990) Myocardial lactate release during ischemia in swine. Relation to regional blood flow. Circulation 81:1948–1958. doi:10.1161/01.cir.81.6.1948
Heaps CL, Tharp DL, Bowles DK (2005) Hypercholesterolemia abolishes voltage-dependent K+ channel contribution to adenosine-mediated relaxation in porcine coronary arterioles. Am J Physiol Heart Circ Physiol 288:H568–H576. doi:10.1152/ajpheart.00157.2004
Henderson KK, Turk JR, Rush JW, Laughlin MH (2004) Endothelial function in coronary arterioles from pigs with early-stage coronary disease induced by high-fat, high-cholesterol diet: effect of exercise. J Appl Physiol 97:1159–1168. doi:10.1152/japplphysiol.00261.2004
Heusch G (2010) Adenosine and maximum coronary vasodilation in humans: myth and misconceptions in the assessment of coronary reserve. Basic Res Cardiol 105:1–5. doi:10.1007/s00395-009-0074-7
Kilroy JP, Klibanov AL, Wamhoff BR, Bowles DK, Hossack JA (2014) Localized in vivo model drug delivery with intravascular ultrasound and microbubbles. Ultrasound Med Biol 40:2458–2467. doi:10.1016/j.ultrasmedbio.2014.04.007
Kuo L, Davis MJ, Cannon MS, Chilian WM (1992) Pathophysiological consequences of atherosclerosis extend into the coronary microcirculation. Restoration of endothelium-dependent responses by l-arginine. Circ Res 70:465–476. doi:10.1161/01.RES.70.3.465
Lario FC, Miname MH, Tsutsui JM, Santos RD, Kowatsch I, Sbano JCN, Ramires JAF, Filho RK, Mathias W (2013) Atorvastatin treatment improves myocardial and peripheral blood flow in familial hypercholesterolemia subjects without evidence of coronary atherosclerosis. Echocardiography 30:64–71. doi:10.1111/j.1540-8175.2012.01810.x
Liyanage KE, Burnett JR, Hooper AJ, van Bockxmeer FM (2011) Familial hypercholesterolemia: epidemiology, neolithic origins and modern geographic distribution. Crit Rev Clin Lab Sci 48:1–18. doi:10.3109/10408363.2011.565585
Lowe SW, Checovich WJ, Rapacz J, Attie AD (1988) Defective receptor binding of low density lipoprotein from pigs possessing mutant apolipoprotein B alleles. J Biol Chem 263:15467–15473
McCommis KS, McGee AM, Laughlin MH, Bowles DK, Baines CP (2011) Hypercholesterolemia increases mitochondrial oxidative stress and enhances the MPT response in the porcine myocardium: beneficial effects of chronic exercise. Am J Physiol Regul Integr Comp Physiol 301:R1250–R1258. doi:10.1152/ajpregu.00841.2010
Merkus D, Sorop O, Houweling B, Boomsma F, van den Meiracker AH, Duncker DJ (2006) NO and prostanoids blunt endothelin-mediated coronary vasoconstrictor influence in exercising swine. Am J Physiol Heart Circ Physiol 291:H2075–H2081. doi:10.1152/ajpheart.01109.2005
Merkus D, Sorop O, Houweling B, Hoogteijling BA, Duncker DJ (2006) KCa channels contribute to exercise-induced coronary vasodilation in swine. Am J Physiol Heart Circ Physiol 291:H2090–H2097. doi:10.1152/ajpheart.00315.2006
Morrison ES, Scott RF, Lee WM, Frick J, Kroms M, Cheney CP (1977) Oxidative phosphorylation and aspects of calcium metabolism in myocardia of hypercholesterolaemic swine with moderate coronary atherosclerosis. Cardiovasc Res 11:547–553. doi:10.1093/cvr/11.6.547
Murthy VL, Naya M, Foster CR, Gaber M, Hainer J, Klein J, Dorbala S, Blankstein R, Di Carli MF (2012) Association between coronary vascular dysfunction and cardiac mortality in patients with and without diabetes mellitus. Circulation 126:1858–1868. doi:10.1161/CIRCULATIONAHA.112.120402
Naoumova RP, Kindler H, Leccisotti L, Mongillo M, Khan MT, Neuwirth C, Seed M, Holvoet P, Betteridge J, Camici PG (2007) Pioglitazone improves myocardial blood flow and glucose utilization in nondiabetic patients with combined hyperlipidemia—a randomized, double-blind, placebo-controlled study. J Am Coll Cardiol 50:2051–2058. doi:10.1016/j.jacc.2007.07.070
Nascimento AR, Machado M, de Jesus N, Gomes F, Lessa MA, Bonomo IT, Tibiriçá E (2013) Structural and functional microvascular alterations in a rat model of metabolic syndrome induced by a high-fat diet. Obesity 21:2046–2054. doi:10.1002/oby.20358
Nordestgaard BG, Chapman MJ, Humphries SE, Ginsberg HN, Masana L, Descamps OS, Wiklund O, Hegele RA, Raal FJ, Defesche JC, Wiegman A, Santos RD, Watts GF, Parhofer KG, Hovingh GK, Kovanen PT, Boileau C, Averna M, Borén J, Bruckert E, Catapano AL, Kuivenhoven JA, Pajukanta P, Ray K, Stalenhoef AFH, Stroes E, Taskinen M-R, Tybjærg-Hansen A (2013) Familial hypercholesterolaemia is underdiagnosed and undertreated in the general population: guidance for clinicians to prevent coronary heart disease. Consensus Statement of the European Atherosclerosis Society. Eur Heart J 34:3478–3490. doi:10.1093/eurheartj/eht273
Paulus WJ, Tschöpe C (2013) A novel paradigm for heart failure with preserved ejection fraction: comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J Am Coll Cardiol 62:263–271. doi:10.1016/j.jacc.2013.02.092
Prescott MF, McBride CH, Hasler-Rapacz J, Von Linden J, Rapacz J (1991) Development of complex atherosclerotic lesions in pigs with inherited hyper-LDL cholesterolemia bearing mutant alleles for apolipoprotein B. Am J Pathol 139:139–147
Pries AR, Badimon L, Bugiardini R, Camici PG, Dorobantu M, Duncker DJ, Escaned J, Koller A, Piek JJ, de Wit C (2015) Coronary vascular regulation, remodelling, and collateralization: mechanisms and clinical implications on behalf of the working group on coronary pathophysiology and microcirculation. Eur Heart J 36:3134–3146. doi:10.1093/eurheartj/ehv100
Rapacz J, Hasler-Rapacz J, Taylor KM, Checovich WJ, Attie AD (1986) Lipoprotein mutations in pigs are associated with elevated plasma cholesterol and atherosclerosis. Science 234:1573–1577. doi:10.1126/science.3787263
Reinhardt CP, Dalhberg S, Tries MA, Marcel R, Leppo JA (2001) Stable labeled microspheres to measure perfusion: validation of a neutron activation assay technique. Am J Physiol Heart Circ Physiol 280:H108–H116
Rodriguez-Porcel M, Lerman LO, Herrmann J, Sawamura T, Napoli C, Lerman A (2003) Hypercholesterolemia and hypertension have synergistic deleterious effects on coronary endothelial function. Arterioscler Thromb Vasc Biol 23:885–891. doi:10.1161/01.atv.0000069209.26507.bf
Schulz R, Oudiz RJ, Guth BD, Heusch G (1990) Minimal α1- and α2-adrenoceptor-mediated coronary vasoconstriction in the anaesthetized swine. Naunyn Schmiedebergs Arch Pharmacol 342:422–428. doi:10.1007/bf00169459
Shioiri H, Komaru T, Sato K, Takahashi K, Takeda S, Kanatsuka H, Watanabe J, Shirato K (2003) Impact of hypercholesterolemia on acidosis-induced coronary microvascular dilation. Basic Res Cardiol 98:76–83. doi:10.1007/s00395-003-0391-1
Stapleton PA, Goodwill AG, James ME, D’Audiffret AC, Frisbee JC (2010) Differential impact of familial hypercholesterolemia and combined hyperlipidemia on vascular wall and network remodeling in mice. Microcirculation 17:47–58. doi:10.1111/j.1549-8719.2009.00003.x
Tharp DL, Masseau I, Ivey J, Ganjam VK, Bowles DK (2009) Endogenous testosterone attenuates neointima formation after moderate coronary balloon injury in male swine. Cardiovasc Res 82:152–160. doi:10.1093/cvr/cvp038
Tharp DL, Wamhoff BR, Wulff H, Raman G, Cheong A, Bowles DK (2008) Local delivery of the KCa3.1 blocker, TRAM-34, prevents acute angioplasty-induced coronary smooth muscle phenotypic modulation and limits stenosis. Arterioscler Thromb Vasc Biol 28:1084–1089. doi:10.1161/atvbaha.107.155796
van de Hoef TP, van Lavieren MA, Damman P, Delewi R, Piek MA, Chamuleau SAJ, Voskuil M, Henriques JPS, Koch KT, de Winter RJ, Spaan JAE, Siebes M, Tijssen JGP, Meuwissen M, Piek JJ (2014) Physiological basis and long-term clinical outcome of discordance between fractional flow reserve and coronary flow velocity reserve in coronary stenoses of intermediate severity. Circ Cardiovasc Interv 7:301–311. doi:10.1161/circinterventions.113.001049
Winnik S, Gaul DS, Siciliani G, Lohmann C, Pasterk L, Calatayud N, Weber J, Eriksson U, Auwerx J, van Tits LJ, Lüscher TF, Matter CM (2016) Mild endothelial dysfunction in Sirt3 knockout mice fed a high-cholesterol diet: protective role of a novel C/EBP-β-dependent feedback regulation of SOD2. Basic Res Cardiol 111:1–15. doi:10.1007/s00395-016-0552-7
Yokoyama I, Murakami T, Ohtake T, Momomura S-i, Nishikawa J, Sasaki Y, Omata M (1996) Reduced coronary flow reserve in familial hypercholesterolemia. J Nucl Med 37:1937–1942
Zeiher AM, Drexler H, Wollschläger H, Just H (1991) Modulation of coronary vasomotor tone in humans. Progressive endothelial dysfunction with different early stages of coronary atherosclerosis. Circulation 83:391–401. doi:10.1161/01.cir.83.2.391
Acknowledgments
We gratefully acknowledge the assistance of Pam Thorne, Dave Harah, Jan Ivey, Sherrie Neff, Dr. Richard McAllister, Elza D. van Deel, and Rob van Bremen. This work was supported by the National Institutes of Health Grants HL-52490 (to MHL) and AR-048523 (to SBB), European Commission FP9-Health-2010 Grant MEDIA-261409 (to DJD and DM), the Netherlands Cardiovascular Research Initiative: an initiative of the Dutch Heart Foundation [CVON2012-08 PHAEDRA to DM; CVON2014-11 RECONNECT to DM and DJD], and the Department of Veterans Affairs Biomedical Laboratory Research and Development CDA-2 IK2 BX002030 (to SBB). This work was supported with resources and the use of facilities at the Harry S Truman Memorial Veterans Hospital in Columbia, MO.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
The manuscript does not contain clinical studies or patient data.
Rights and permissions
About this article
Cite this article
Bender, S.B., de Beer, V.J., Tharp, D.L. et al. Severe familial hypercholesterolemia impairs the regulation of coronary blood flow and oxygen supply during exercise. Basic Res Cardiol 111, 61 (2016). https://doi.org/10.1007/s00395-016-0579-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-016-0579-9