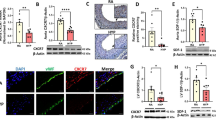
Abstract
Objectives
The response to hypoxia is primarily mediated by the transcription factor hypoxia-inducible factor-1 (HIF-1) which leads to the induction of a variety of adaptive gene products including vascular endothelial growth factor (VEGF) and endothelial nitric oxide synthase (eNOS). This study was designed to test the hypothesis that HIF-1 and its target genes would be upregulated in the ventricular myocardium of infants with cyanotic congenital cardiac defects.
Methods
14 infants with cyanotic (n = 7) or acyanotic cardiac defects (n = 7) were investigated. Samples from the right ventricular myocardium taken immediately after aortic clamping were studied for protein expression and DNA-binding activity.
Results
Protein levels of HIF-1α were significantly elevated in patients with cyanotic compared to acyanotic congenital heart disease and inversely correlated with the degree of hypoxemia. This response was accompanied by significantly enhanced HIF-1 DNA binding activity. Furthermore, protein levels of VEGF and eNOS were significantly higher in the myocardium of cyanotic than of acyanotic infants. To test the potential involvement of upstream regulatory pathways, activation of MAP kinases was determined. Intramyocardial levels of phosphorylated p38 MAP kinase, but not of ERK1/2 were significantly higher in infants with cyanotic compared to those with acyanotic congenital heart disease and inversely correlated to hypoxemia.
Conclusions
These findings show that chronic hypoxemia is associated with the induction and stabilization of the transcription factor HIF-1 as well as its target genes VEGF and eNOS in the myocardium of infants with cyanotic cardiac defects. Thus, stabilization of HIF-1 and induction of the adaptive hypoxia response could particularly participate in myocardial remodeling in children with congenital cardiac defects and chronic hypoxemia.
Similar content being viewed by others
References
Andrews NC, Faller DV (1991) A rapid micropreparation technique for extraction of DNA-binding proteins from limiting numbers of mammalian cells. Nucleic Acids Res 19:2499
BelAiba RS, Djordjevic T, Bonello S, Flugel D, Hess J, Kietzmann T, Gorlach A (2004) Redox-sensitive regulation of the HIF pathway under non-hypoxic conditions in pulmonary artery smooth muscle cells. Biol Chem 385:249–257
Bogoyevitch MA, Gillespie-Brown J, Ketterman AJ, Fuller SJ, Ben-Levy R, Ashworth A, Marshall CJ, Sugden PH (1996) Stimulation of the stress-activated mitogen-activated protein kinase subfamilies in perfused heart. p38/RK mitogen-activated protein kinases and c-Jun N-terminal kinases are activated by ischemia/reperfusion. Circ Res 79:162–173
Burton PB, Owen VJ, Hafizi S, Barton PJ, Carr-White G, Koh T, De Souza A, Yacoub MH, Pepper JR (2000) Vascular endothelial growth factor release following coronary artery bypass surgery: extracorporeal circulation versus ‘beating heart-surgery. Eur Heart J 21:1708–1713
Coulet F, Nadaud S, Agrapart M, Soubrier F (2003) Identification of hypoxia- response element in the human endothelial nitric-oxide synthase gene promoter. J Biol Chem 278:46230–46240
Couvelard A, O’Toole D, Leek R, Turley H, Sauvanet A, Degott C, Ruszniewski P, Belghiti J, Harris AL, Gatter K, Pezzella F (2005) Expression of hypoxia-inducible factors is correlated with the presence of a fibrotic focus and angiogenesis in pancreatic ductal adenocarcinomas. Histopathology 46:668–676
Felaco M, Grilli A, Gorbunov N, Di Napoli P, De Lutiis MA, Di Giulio C, Taccardi AA, Barsotti A, Barbacane RC, Reale M, Conti P (2000) Endothelial NOS expression and ischemia-reperfusion in isolated working rat heart from hypoxic and hyperoxic conditions. Biochim Biophys Acta 1524:203–211
Feng Q, Song W, Lu X, Hamilton JA, Lei M, Peng T, Yee SP (2002) Development of heart failure and congenital septal defects in mice lacking endothelial nitric oxide synthase. Circulation 106:873–879
Ferrara N (2004) Vascular endothelial growth factor: basic science and clinical progress. Endocr Rev 25:581–611
Ferreiro CR, Chagas AC, Carvalho MH, Dantas AP, Jatene MB, Bento De Souza LC, Lemos Da Luz P (2001) Influence of hypoxia on nitric oxide synthase activity and gene expression in children with congenital heart disease: a novel pathophysiological adaptive mechanism. Circulation 103:2272–2276
Forkel J, Chen X, Wandinger S, Keser F, Duschin A, Schwanke U, Frede S, Massoudy P, Schulz R, Jakob H, Heusch G (2004) Responses of chronically hypoxic rat hearts to ischemia: KATP channel blockade does not abolish increased RV tolerance to ischemia. Am J Physiol Heart Circ Physiol 286:545–551
Fukumura D, Gohongi T, Kadambi A, Izumi Y, Ang J, Yun CO, Buerk DG, Huang PL, Jain RK (2001) Predominant role of endothelial nitric oxide synthase in vascular endothelial growth factorinduced angiogenesis and vascular permeability. Proc Natl Acad Sci U S A 98:2604–2609
Giordano FJ, Gerber HP, Williams SP, VanBruggen N, Bunting S, Ruiz-Lozano P, Gu Y, Nath AK, Huang Y, Hickey R, Dalton N, Peterson KL, Ross J, Jr., Chien KR, Ferrara N (2001) A cardiac myocyte vascular endothelial growth factor paracrine pathway is required to maintain cardiac function. Proc Natl Acad Sci USA 98:5780–5785
Gorlach A, Diebold I, Schini-Kerth VB, Berchner-Pfannschmidt U, Roth U, Brandes RP, Kietzmann T, Busse R (2001) Thrombin activates the hypoxiainducible factor-1 signaling pathway in vascular smooth muscle cells: Role of the p22(phox)-containing NADPH oxidase. Circ Res 89:47–54
Hellwig-Burgel T, Stiehl DP,Wagner AE, Metzen E, Jelkmann W (2005) Review: hypoxia-inducible factor-1 (HIF-1): a novel transcription factor in immune reactions. J Interferon Cytokine Res 25:297–310
Himeno W, Akagi T, Furui J, Maeno Y, Ishii M, Kosai K, Murohara T, Kato H (2003) Increased angiogenic growth factor in cyanotic congenital heart disease. Pediatr Cardiol 24:127–132
Hood JD, Meininger CJ, Ziche M, Granger HJ (1998) VEGF upregulates ecNOS message, protein, and NO production in human endothelial cells. Am J Physiol 274:1054–1058
Hu J, Sun P, Ruan X, Chao A, Lin Y, Li XY (2005) Mechanism of myocardial microvessel formation in cyanotic congenital heart disease. Circ J 69:1089–1093
Jurgensen JS, Rosenberger C, Wiesener MS, Warnecke C, Horstrup JH, Grafe M, Philipp S, Griethe W, Maxwell PH, Frei U, Bachmann S, Willenbrock R, Eckardt KU (2004) Persistent induction of HIF- 1alpha and -2alpha in cardiomyocytes and stromal cells of ischemic myocardium. Faseb J 18:1415–1417
Kido M, Du L, Sullivan CC, Li X, Deutsch R, Jamieson SW, Thistlethwaite PA (2005) Hypoxia-inducible factor 1-alpha reduces infarction and attenuates progression of cardiac dysfunction after myocardial infarction in the mouse. J Am Coll Cardiol 46:2116–1124
Kietzmann T, Gorlach A (2005) Reactive oxygen species in the control of hypoxia- inducible factor-mediated gene expression. Semin Cell Dev Biol 16:474–486
Kietzmann T, Jungermann K, Gorlach A (2003) Regulation of the hypoxia-dependent plasminogen activator inhibitor 1 expression by MAP kinases. Thromb Haemost 89:666–673
Kim CH, Cho YS, Chun YS, Park JW, Kim MS (2002) Early expression of myocardial HIF-1alpha in response to mechanical stresses: regulation by stretch-activated channels and the phosphatidylinositol 3-kinase signaling pathway. Circ Res 90:25–33
Lee SH, Wolf PL, Escudero R, Deutsch R, Jamieson SW, Thistlethwaite PA (2000) Early expression of angiogenesis factors in acute myocardial ischemia and infarction. N Engl J Med 342:626–633
Mazure NM, Brahimi-Horn MC, Pouyssegur J (2003) Protein kinases and the hypoxia-inducible factor-1, two switches in angiogenesis. Curr Pharm Des 9:531–541
Namba T, Koike H, Murakami K, Aoki M, Makino H, Hashiya N, Ogihara T, Kaneda Y, Kohno M, Morishita R (2003) Angiogenesis induced by endothelial nitric oxide synthase gene through vascular endothelial growth factor expression in a rat hindlimb ischemia model. Circulation 108:2250–2257
Parisi Q, Biondi-Zoccai GG, Abbate A, Santini D, Vasaturo F, Scarpa S, Bussani R, Leone AM, Petrolini A, Silvestri F, Biasucci LM, Baldi A (2005) Hypoxia inducible factor-1 expression mediates myocardial response to ischemia late after acute myocardial infarction. Int J Cardiol 99:337–339
Philipp S, Cui L, Ludolph B, Kelm M, Schulz R, Cohen MV, Downey JM (2006) Desferoxamine and ethyl-3,4-dihydroxybenzoate protect myocardium by activating NOS and generating mitochondrial ROS. Am J Physiol Heart Circ Physiol 290:450–457
Qing M, Schumacher K, Heise R, Woltje M, Vazquez-Jimenez JF, Richter T, Arranda- Carrero M, Hess J, von Bernuth G, Seghaye MC (2003) Intramyocardial synthesis of pro- and anti-inflammatory cytokines in infants with congenital cardiac defects. J Am Coll Cardiol 41:2266–2274
Rapino C, Bianchi G, Di Giulio C, Centurione L, Cacchio M, Antonucci A, Cataldi A (2005) HIF-1alpha cytoplasmic accumulation is associated with cell death in old rat cerebral cortex exposed to intermittent hypoxia. Aging Cell 4:177–185
Semenza GL (2004) O2-regulated gene expression: transcriptional control of cardiorespiratory physiology by HIF-1. J Appl Physiol 96:1173–1177; discussion 117–1172
Shyu KG, Wang MT, Wang BW, Chang CC, Leu JG, Kuan P, Chang H (2002) Intramyocardial injection of naked DNA encoding HIF-1alpha/VP16 hybrid to enhance angiogenesis in an acute myocardial infarction model in the rat. Cardiovasc Res 54:576–583
Starnes SL, Duncan BW, Kneebone JM, Rosenthal GL, Jones TK, Grifka RG, Cecchin F, Owens DJ, Fearneyhough C, Lupinetti FM (2000) Vascular endothelial growth factor and basic fibroblast growth factor in children with cyanotic congenital heart disease. J Thorac Cardiovasc Surg 119:534–539
Tammela T, Enholm B, Alitalo K, Paavonen K (2005) The biology of vascular endothelial growth factors. Cardiovasc Res 65:550–563
Tomanek RJ, Zheng W, Yue X (2004) Growth factor activation in myocardial vascularization: therapeutic implications. Mol Cell Biochem 264:3–11
Zhao X, Lu X, Feng Q (2002) Deficiency in endothelial nitric oxide synthase impairs myocardial angiogenesis. Am J Physiol Heart Circ Physiol 283:2371–2378
Author information
Authors and Affiliations
Corresponding authors
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s00395-007-0654-3.
Rights and permissions
About this article
Cite this article
Quing, M., Görlach, A., Schumacher, K. et al. The hypoxia-inducible factor HIF-1 promotes intramyocardial expression of VEGF in infants with congenital cardiac defects. Basic Res Cardiol 102, 224–232 (2007). https://doi.org/10.1007/s00395-007-0639-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-007-0639-2