Abstract
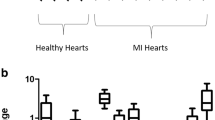
Enzymes involved in the metabolism nitric oxide (NO) and reactive oxygen species (ROS) may play a role for the decreased availability of NO in atherosclerosis. We, therefore, hypothesized that the pattern of gene expression of these enzymes is altered in atherosclerosis. Myocardial tissue from patients with coronary heart disease (CHD) or without CHD (control group) was investigated. The level of enzymes related to NO/ROS metabolism was determined both at mRNA level and protein level by rt-PCR, real-time PCR, and western blot. The expression of NOS1–3 (synthesis of NO), arginase1 (reduction of l-arginine), p22phox (active subunit of NADPH oxidase), GTPCH (rate limiting enzyme for tetrahydrobiopterin), SOD1–3 (scavengers of superoxide anions), PRTMT1–3, and DDAH2 (involved in the metabolism of ADMA) was determined. All enzymes were found to be expressed in human myocardium. NOS isoforms were decreased in CHD in protein level, but only the downregulation of NOS3 expression reached statistical significance. The expression of PRMT1 and PRMT3 was increased. In addition, the expression of DDAH2 was reduced, both theoretically leading to an increase of ADMA concentration. SOD3 was downregulated in tissue from patients with CHD. Taken together, in myocardial tissue from patients with atherosclerosis, the expression of genes increasing ADMA levels is enhanced in contrast to a reduced expression of genes promoting NO synthesis. These results may contribute to the explanation of increased oxidative stress in atherosclerosis on the level of gene expression.




Similar content being viewed by others
References
Asakura T, Karino T (1990) Flow patterns, spatial distribution of atherosclerotic lesions in human coronary arteries. Circ Res 66(4):1045–1066
Azumi H, Inoue N, Takeshita S, Rikitake Y, Kawashima S, Hayashi Y, Itoh H, Yokoyama M. (1999). Expression of NADH/NADPH oxidase p22phox in human coronary arteries. Circulation 100(14):1494–1498
Behr-Roussel D, Rupin A, Simonet S, Bonhomme E, Coumailleau S, Cordi A, Serkiz B, Fabiani JN, Verbeuren TJ (2000) Effect of chronic treatment with the inducible nitric oxide synthase inhibitor N-iminoethyl-l-lysine or with l-arginine on progression of coronary and aortic atherosclerosis in hypercholesterolemic rabbits. Circulation 102(9):1033–1038
Cai S, Alp NJ, McDonald D, Smith I, Kay J, Canevari L, Heales S, Channon KM. (2002) GTP cyclohydrolase I gene transfer augments intracellular tetrahydrobiopterin in human endothelial cells: effects on nitric oxide synthase activity, protein levels and dimerisation. Cardiovasc Res 55(4):838–849
Channon KM, Qian H, George SE (2000) Nitric oxide synthase in atherosclerosis and vascular injury: insights from experimental gene therapy. Arterioscler Thromb Vasc Biol 20(8):1873–1881
Chauhan A, More RS, Mullins PA, Taylor G, Petch C, Schofield PM (1996) Aging-associated endothelial dysfunction in humans is reversed by l-arginine. J Am Coll Cardiol 28(7):1796–1804
Cines DB, Pollak ES, Buck CA, Loscalzo J, Zimmerman GA, McEver RP, Pober JS, Wick TM, Konkle BA, Schwartz BS, Barnathan ES, McCrae KR, Hug BA, Schmidt AM, Stern DM (1998) Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood 91(10):3527–3561
Cominacini L, Rigoni A, Pasini AF, Garbin U, Davoli A, Campagnola M, Pastorino AM, Lo Cascio V, Sawamura T (2001) The binding of oxidized low density lipoprotein (ox-LDL) to ox-LDL receptor-1 reduces the intracellular concentration of nitric oxide in endothelial cells through an increased production of superoxide. J Biol Chem 276(17):13750–13755
Cromheeke KM, Kockx MM, De Meyer GR, Bosmans JM, Bult H, Beelaerts WJ, Vrints CJ, Herman AG (1999) Inducible nitric oxide synthase colocalizes with signs of lipid oxidation/peroxidation in human atherosclerotic plaques. Cardiovasc Res 43(3):744–754
Drexler H, Zeiher AM, Meinzer K, Just H (1991) Correction of endothelial dysfunction in coronary microcirculation of hypercholesterolaemic patients by l-arginine. Lancet 338(8782–8783):1546–1550
Gesierich A, Niroomand F, Tiefenbacher CP (2003) Role of human GTP cyclohydrolase I and its regulatory protein in tetrahydrobiopterin metabolism. Basic Res Cardiol 98(2):69–75
Goumas G, Tentolouris C, Tousoulis D, Stefanadis C, Toutouzas P (2001) Therapeutic modification of the l-arginine-eNOS pathway in cardiovascular diseases. Atherosclerosis 154(2):255–267
Gryglewski RJ, Palmer RM, Moncada S (1986) Superoxide anion is involved in the breakdown of endothelium-derived vascular relaxing factor. Nature 320(6061):454–456
Jenkinson CP, Grody WW, Cederbaum SD (1996) Comparative properties of arginases. Comp Biochem Physiol B Biochem Mol Biol 114(1):107–132
Kielstein JT, Bode-Boger SM, Frolich JC, Haller H, Boger RH (2001) Relationship of asymmetric dimethylarginine to dialysis treatment and atherosclerotic disease. Kidney Int Suppl 78:S9–13
Kimoto M, Whitley GS, Tsuji H, Ogawa T (1995) Detection of NG, NG-dimethylarginine dimethylaminohydrolase in human tissues using a monoclonal antibody. J Biochem 117(2):237–238
Landmesser U, Merten R, Spiekermann S, Buttner K, Drexler H, Hornig B (2000) Vascular extracellular superoxide dismutase activity in patients with coronary artery disease: relation to endothelium-dependent vasodilation. Circulation 101(19):2264–2270
Leiper JM, Santa Maria J, Chubb A, MacAllister RJ, Charles IG, Whitley GS, Vallance P (1999) Identification of two human dimethylarginine dimethylaminohydrolases with distinct tissue distributions and homology with microbial arginine deiminases. Biochem J 343( Pt 1):209–214
MacAllister RJ, Parry H, Kimoto M, Ogawa T, Russell RJ, Hodson H, Whitley GS, Vallance P (1996) Regulation of nitric oxide synthesis by dimethylarginine dimethylaminohydrolase. Br J Pharmacol 119(8):1533–1540
Marletta MA (1994) Nitric oxide synthase: aspects concerning structure and catalysis. Cell 78(6):927–930
Masuda H, Tsujii T, Okuno T, Kihara K, Goto M, Azuma H. (2001) Involvement of accumulated endogenous NOS inhibitors and decreased NOS activity in the impaired neurogenic relaxation of the rabbit proximal urethra with ischaemia. Br J Pharmacol 133(1):97–106
Price DT, Vita JA, Keaney JF, Jr. (2000) Redox control of vascular nitric oxide bioavailability. Antioxid Redox Signal 2(4):919–935
Ravalli S, Albala A, Ming M, Szabolcs M, Barbone A, Michler RE, Cannon PJ (1998) Inducible nitric oxide synthase expression in smooth muscle cells and macrophages of human transplant coronary artery disease. Circulation 97(23):2338–2345
Richartz BM, Werner GS, Ferrari M, Figulla HR (2001) Reversibility of coronary endothelial vasomotor dysfunction in idiopathic dilated cardiomyopathy: acute effects of vitamin C. Am J Cardiol 88(9):1001–1005
Schmidt HH, Nau H, Wittfoht W, Gerlach J, Prescher KE, Klein MM, Niroomand F, Bohme E. (1998) Arginine is a physiological precursor of endothelium-derived nitric oxide. Eur J Pharmacol 154(2):213–216
Silacci P, Formentin K, Bouzourene K, Daniel F, Brunner HR, Hayoz D (2000) Unidirectional and oscillatory shear stress differentially modulate NOS III gene expression. Nitric Oxide 4(1):47–56
Stuhlinger MC, Tsao PS, Her JH, Kimoto M, Balint RF, Cooke JP (2001) Homocysteine impairs the nitric oxide synthase pathway: role of asymmetric dimethylarginine. Circulation 104(21):2569–2575
Tang J, Gary JD, Clarke S, Herschman HR. (1998) PRMT 3, a type I protein arginine N-methyltransferase that differs from PRMT1 in its oligomerization, subcellular localization, substrate specificity, and regulation. J Biol Chem 273(27):16935–16945
Tiefenbacher CP, Bleeke T, Vahl C, Amann K, Vogt A, Kübler W (2000) Endothelial dysfunction of coronary resistance arteries is improved by tetrahydrobiopterin in atherosclerosis. Circulation 102: 2172–2179
Tran CT, Fox MF, Vallance P, Leiper JM (2000) Chromosomal localization, gene structure, and expression pattern of DDAH1: comparison with DDAH2 and implications for evolutionary origins. Genomics 68(1):101–105
Tzeng E, Kim YM, Pitt BR, Lizonova A, Kovesdi I, Billiar TR (1997) Adenoviral transfer of the inducible nitric oxide synthase gene blocks endothelial cell apoptosis. Surgery 122(2):255–263
Vallance P, Leone A, Calver A, Collier J, Moncada S (1992) Accumulation of an endogenous inhibitor of nitric oxide synthesis in chronic renal failure. Lancet 339(8793):572–575
Warnholtz A, Mollnau H, Oelze M, Wendt M, Munzel T (2001) Antioxidants and endothelial dysfunction in hyperlipidemia. Curr Hypertens Rep 3(1):53–60
Xu KY, Huso DL, Dawson TM, Bredt DS, Becker LC (1999) Nitric oxide synthase in cardiac sarcoplasmic reticulum. Proc Natl Acad Sci USA 96(2):657–62
Author information
Authors and Affiliations
Corresponding author
Additional information
Returned for 1st revision: 15 August 2005 Returned for 2nd revision: 29 November 2005 2nd revision received: 22 December 2005
Rights and permissions
About this article
Cite this article
Chen, X., Niroomand, F., Liu, Z. et al. Expression of nitric oxide related enzymes in coronary heart disease. Basic Res Cardiol 101, 346–353 (2006). https://doi.org/10.1007/s00395-006-0592-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-006-0592-5