Abstract
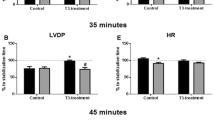
There is accumulating evidence that thyroid hormone metabolism is altered after myocardial infarction (AMI) but its physiological relevance remains largely unknown. The present study investigated the possible role of thyroid hormone signaling in the response of the post-infarcted heart to ischaemia-reperfusion. Wistar rats were subjected to left coronary artery ligation (AMI), or sham operation (SHAM). After 8 weeks, hearts from AMI and SHAM rats were perfused in Langendorff mode and subjected to 20 min of zero-flow global ischaemia (I) and 45 min of reperfusion (R); AMI(I/R), n = 7 and SHAM(I/R), n = 7. Basal left ventricular pressure (LVDP), +dp/dt, and –dp/dt were significantly reduced. Left ventricular weight of the viable myocardium was increased by 14% in the AMI as compared to SHAM hearts, P < 0.05. T3 and T4 plasma levels in nM were 1.83 (0.08) and 53.3 (2.9) for SHAM and 1.76 (0.06) and 59.4 (5.2) for AMI rats, respectively, P > 0.05. TRα1 and TRβ1 expression levels were 1.3- and 1.8-fold less in AMI than in SHAM hearts, P < 0.05. Furthermore, SERCA and NHE1 expression levels were 2.1- and 1.8-fold less in AMI than in SHAM, P < 0.05. PKCε was 1.35-fold more in AMI compared to SHAM, P < 0.05. Myocardial glycogen content (in µmol/g) was 7.8 (1.2) in AMI as compared to 4.4 (0.5) for SHAM hearts, P < 0.05. After I/R, left ventricular end-diastolic pressure at 45 min of R (LVEDP45 in mmHg) was 20.3 (3.2) for AMI(I/R) vs 50.6 (4.8) mmHg for SHAM(I/R), P < 0.05. LDH release per gram of tissue was 251 (103) for AMI(I/R) and 762 (74) for SHAM(I/R), P < 0.05. In conclusion, TRα1 and TRβ1 are downregulated after myocardial infarction and this was associated with altered expression of thyroid hormone responsive genes and increased tolerance of the post-infarcted heart to ischaemia-reperfusion injury.
Similar content being viewed by others
References
Abe M, Obata H, Tanaka H (1992) Functional and metabolic responses to ischaemia in the isolated perfused hypothyroid rat heart. Jpn Circ J 56 (7):671–680
Avkiran M (2001) Protection of the ischemic myocardium by Na+/H+ exchange inhibitors: potential mechanisms of action. Bas Res Cardiol 96:306–311
Bricknell OL, Daries PS, Opie LH (1981) A relationship between adenosine triphosphate, glycolysis and ischaemic contracture in the isolated rat heart. J Mol Cell Cardiol 13:941–945
Correia H, Rivero J, Herrera F (1997) Isolation of nuclear proteins from human brains. Biotechniques 22:848–850
Cross HR, Murphy E, Bolli R, Ping P, Steenbergen C (2002) Expression of activated PKC epsilon (PKCε) protects the ischaemic heart, without attenuating ischaemic H+ production. J Mol Cell Cardiol 34:361–367
Cross HR, Opie LH, Radda GK, Clarke K (1996) Is high glycogen content beneficial or detrimental to the ischaemic rat heart? A controversy resolved. Circ Res 78:482–491
Friberg L, Werner S, Eggertsen G, Ahnve S (2002) Rapid downregulation of thyroid hormones in acute myocardial infarction. Is it cardioprotective in patients with angina? Arch Intern Med 162:1388–1394
Franklyn JA, Gammage MD, Raymsden DB, Sheppard MC (1984) Thyroid status in patients after acute myocardial infarction. Clin Sci 67:585–590
Geenen DL, Malhotra A, Scheuer J (1989) Regional variation in rat cardiac myosin isoenzymes and ATPase activity after infarction. Am J Physiol 256:H745–H750
Harvey CB, Williams GR (2002) Mechanism of thyroid hormone action. Thyroid 12 (6):441–446
Hu K, Gaudron P, Ertl G (2004) Effects of high and low dose amiodarone on mortality, left ventricular remodeling, and hemodynamics in rats with experimental myocardial infarction. J Cardiovasc Pharmacol 44:627–630
Kingsley PB, Sako EY, Yang MQ, Zimmer SD, Ugurbil K, Foker JE, From AHL (1991) Ischemic contracture begins when anaerobic glycolysis stops: a 31P-NMR study of isolated rat hearts. Am J Physiol 261:H469–H478
Kinugawa K, Yonekura K, Ribeiro RC, Eto Y, Aoyagi T, Baxter JD, Camacho SA, Bristow MR, Long CS, Simpson PC (2001) Regulation of thyroid hormone receptor isoforms in physiological and pathological cardiac hypertrophy. Circ Res 89 (7):591–598
Klein I, Ojamma K (2001) Thyroid hormone targeting the heart. Endocrinology 142:11–12
Liu G, Cohen M, Mochly-Rosen D, Downey J (1999) Protein kinase C–ε is responsible for the protection of preconditioning in rabbit cardiomyocytes. J Mol Cell Cardiol 31:1937–1948
Morkin E, Ladenson P, Goldman S, Adamson C (2004) Thyroid hormone analogs for treatment of hypercholesterolemia and heart failure: past, present and future prospects. J Mol Cell Cardiol 37:1137–1146
Ojamaa K, Kenessey A, Shenoy R, Klein I (2000) Thyroid hormone metabolism and cardiac gene expression after acute myocardial infarction in the rat. Am J Physiol 279:E1319–E1324
Pantos C, Malliopoulou V, Mourouzis I, Karamanoli E, Tzeis SM, Carageorgiou H, Varonos D, Cokkinos DV (2001) Longterm thyroxine administration increases HSP70 mRNA expression and attenuates p38 MAP kinase activity in response to ischaemia. J Endocrinol 170:207–215
Pantos C, Malliopoulou V, Mourouzis I, Moraitis P, Tzeis S, Thempeyioti A, Paizis I, Cokkinos AD, Carageorgiou H, Varonos D, Cokkinos DV (2003) Involvement of p38 MAPK and JNK in the heat stress induced cardioprotection. Bas Res Cardiol 98:158–164
Pantos C, Malliopoulou V, Mourouzis I, Sfakianoudis K, Tzeis S, Doumba P, Xinaris C, Cokkinos AD, Carageorgiou H, Varonos D, Cokkinos DV (2003) Propylthiouracil induced hypothyroidism is associated with increased tolerance of the isolated rat heart to ischaemia-reperfusion. J Endocrinol 178 (3):427–435
Pantos C, Malliopoulou V, Mourouzis I, Karamanoli E, Moraitis P, Tzeis S, Paizis I, Carageorgiou H, Varonos D, Cokkinos DV (2003) Thyroxine pretreatment increases basal myocardial HSP27 expression and accelerates translocation and phosphorylation of this protein upon ischaemia. Eur J Pharmacol 478:53–60
Pantos C, Malliopoulou V, Varonos D, Cokkinos DV (2004) Thyroid hormone and phenotypes of cardioprotection. Basic Res Cardiol 99:101–120
Pantos C, Mourouzis I, Malliopoulou V, Paizis I, Tzeis S, Moraitis P, Sfakianoudis K, Varonos D, Cokkinos DV (2005) Dronedarone administration prevents body weight gain and increases tolerance of the heart to ischaemic stress: possible involvement of thyroid hormone receptor 1. Thyroid 15 (1):16–23
Pennock GD, Spooner PH, Summers CE, Litwin SE (2000) Prevention of abnormal sarcoplasmic reticulum calcium transport and protein expression in postinfarction heart failure using 3,5 diiodothyropropionic acid (DITPA). J Mol Cell Cardiol 32:1939–1953
Rybin V, Steinberg SF (1996) Thyroid hormone represses protein kinase C isoform expression and activity in rat cardiac myocytes. Circ Res 79:388–398
Slepkov E, Fliegel L (2004) Regulation of expression of the Na+/H+ exchanger by thyroid hormone. Vitam Horm 69:249–269
Sussman MA (2001) When the thyroid speaks the heart listens. Circ Res 89:557–559
Taegtmeyer H (2004) Glycogen in the heart-an expanded view. J Mol Cell Cardiol 37:43–50
Van Beeren HC, Bakker O, Wiersinga WM (1995) Desethylamiodarone is a competitive inhibitor of the binding of thyroid hormone to the thyroid hormone alpha 1-receptor protein. Mol Cell Endocrinol 112 (1):15–19
Van Beeren HC, Jong WM, Kaptein E, Visser TJ, Bakker O, Wiersinga WM (2003) Dronedarone acts as a selective inhibitor of 3,5,3’-triiodothyronine binding to thyroid hormone receptor α1: in vitro and in vivo evidence. Endocrinology 144 (2):552–558
Wang Y, Meyer JW, Ashraf M, Shull GE (2003) Mice with a null mutation in the NHE1 Na+-H+ exchanger are resistant to cardiac ischaemia-reperfusion injury. Circ Res 93:694–696
Zhang L, Parratt JR, Beastall GH, Pyne NJ, Furman BL (2002) Streptozotocin diabetes protects against arrhythmias in rat isolated hearts :role of hypothyroidism. Eur J Pharmacolol 435:269–276
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pantos, C., Mourouzis, I., Saranteas, T. et al. Thyroid hormone receptors α1 and β1 are downregulated in the post-infarcted rat heart: consequences on the response to ischaemia-reperfusion. Basic Res Cardiol 100, 422–432 (2005). https://doi.org/10.1007/s00395-005-0545-4
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00395-005-0545-4