Abstract
Purpose
Dietary fiber (DF) has a good application prospect in effectively restoring the integrity of the intestinal mucosal barrier. Ginseng-DF has good physicochemical properties and physiological activity and shows positive effects in enhancing immunity. The aim of this study was to investigate the protective effect of Ginseng-DF on intestinal mucosal barrier injury induced by cyclophosphamide (CTX) in immunosuppressed mice and its possible mechanism.
Methods
The effects of Gginseng-DF on immune function in mice were studied by delayed-type hypersensitivy, lymphocyte proliferation assay and NK cytotoxicity assay, the T lymphocyte differentiation and intestinal barrier integrity were analyzed by flow cytometry and western blot.
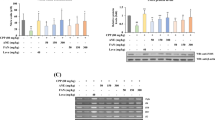
Results
Ginseng-DF (2.5% and 5%) could attenuate the inhibition of DTH response by CTX, promote the transformation and proliferation of lymphocytes, and stimulate NK effector cell activity. At the same time, Ginseng-DF could restore the proportion of CD4+/CD8+ T lymphocytes induced by CTX to different extents, improved spleen tissue damage, promoted the secretion of immunoglobulin IgG, and enhanced body immunity. More importantly, Ginseng-DF could up-regulate the contents of TNF-α, IFN-γ, IL-6 and IL-1β in serum and intestine of immunosuppressed mice to maintain the balance between Th1/Th2 cytokines, and improve the permeability of intestinal mucosal barrier. Meanwhile, Ginseng-DF could reduce intestinal epithelial cell apoptosis and improve intestinal adaptive immunity in CTX-induced immunosuppressed mice by regulating MAPK/NF-κB signaling pathway.
Conclusion
Ginseng-DF can be used as a safe dietary supplement to enhance body immunity and reduce intestinal mucosal injury caused by CTX.







Similar content being viewed by others
Data availability
The data that support the findings of this study are available upon request from the corresponding author.
Abbreviations
- DF:
-
Dietary fiber
- Th1/Th2:
-
T helper 1 and T helper 2
- IL-6:
-
Interleukin 6
- IFN-γ:
-
Interferon γ
- MAPK:
-
Mitogen-activated protein kinase
- Con-A:
-
Concanavalin A
- CytC:
-
Cytochrome C
- CTX:
-
Cyclophosphamide
- IgG:
-
Immunoglobulin G
- IL-1β:
-
Interleukin 1β
- TNF-α:
-
Tumor necrosis factor α
- NF-κB:
-
Nuclear factor-κB
- FBS:
-
Fetal bovine serum
- DTH:
-
Delayed-type hypersensitivity
References
Gray JI, Farber DL (2022) Tissue-resident immune cells in humans. Annu Rev Immunol 40:195–220. https://doi.org/10.1146/annurev-immunol-093019-112809
Emadi A, Jones RJ, Brodsky RA (2009) Cyclophosphamide and cancer: golden anniversary. Nat Rev Clin Oncol 6(11):638–647. https://doi.org/10.1038/nrclinonc.2009.146
Highley MS, Landuyt B, Prenen H, Harper PG, De Bruijn EA (2022) The nitrogen mustards. Pharmacol Rev 74(3):552–599. https://doi.org/10.1124/pharmrev.120.000121
Ponticelli C, Escoli R, Moroni G (2018) Does cyclophosphamide still play a role in glomerular diseases? Autoimmun Rev 17(10):1022–1027. https://doi.org/10.1016/j.autrev.2018.04.007
Suarez-Almazor ME, Belseck E, Shea B, Wells G, Tugwell P (2000) Cyclophosphamide for rheumatoid arthritis. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD001157
Fluckiger A, Daillere R, Sassi M, Sixt BS, Liu P, Loos F, Richard C, Rabu C, Alou MT, Goubet AG, Lemaitre F, Ferrere G, Derosa L, Duong CPM, Messaoudene M, Gagne A, Joubert P, De Sordi L, Debarbieux L, Simon S, Scarlata CM, Ayyoub M, Palermo B, Facciolo F, Boidot R, Wheeler R, Boneca IG, Sztupinszki Z, Papp K, Csabai I, Pasolli E, Segata N, Lopez-Otin C, Szallasi Z, Andre F, Iebba V, Quiniou V, Klatzmann D, Boukhalil J, Khelaifia S, Raoult D, Albiges L, Escudier B, Eggermont A, Mami-Chouaib F, Nistico P, Ghiringhelli F, Routy B, Labarriere N, Cattoir V, Kroemer G, Zitvogel L (2020) Cross-reactivity between tumor MHC class I-restricted antigens and an enterococcal bacteriophage. Science 369(6506):936–942. https://doi.org/10.1126/science.aax0701
Peterson LW, Artis D (2014) Intestinal epithelial cells: regulators of barrier function and immune homeostasis. Nat Rev Immunol 14(3):141–153. https://doi.org/10.1038/nri3608
Weitkamp JH, Koyama T, Rock MT, Correa H, Goettel JA, Matta P, Oswald-Richter K, Rosen MJ, Engelhardt BG, Moore DJ, Polk DB (2013) Necrotising enterocolitis is characterised by disrupted immune regulation and diminished mucosal regulatory (FOXP3)/effector (CD4, CD8) T cell ratios. Gut 62(1):73–82. https://doi.org/10.1136/gutjnl-2011-301551
Shakoor H, Feehan J, Apostolopoulos V, Platat C, Al Dhaheri AS, Ali HI, Ismail LC, Bosevski M, Stojanovska L (2021) Immunomodulatory effects of dietary polyphenols. Nutrients. https://doi.org/10.3390/nu13030728
Arthur JS, Ley SC (2013) Mitogen-activated protein kinases in innate immunity. Nat Rev Immunol 13(9):679–692. https://doi.org/10.1038/nri3495
Chen J, Chen X, Xuan Y, Shen H, Tang Y, Zhang T, Xu J (2023) Surface functionalization-dependent inflammatory potential of polystyrene nanoplastics through the activation of MAPK/NF-kappaB signaling pathways in macrophage Raw 264.7. Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2023.114520
Desai MS, Seekatz AM, Koropatkin NM, Kamada N, Hickey CA, Wolter M, Pudlo NA, Kitamoto S, Terrapon N, Muller A, Young VB, Henrissat B, Wilmes P, Stappenbeck TS, Nunez G, Martens EC (2016) A dietary fiber-deprived gut microbiota degrades the colonic mucus barrier and enhances pathogen susceptibility. Cell 167(5):1339-1353 e1321. https://doi.org/10.1016/j.cell.2016.10.043
Gao H, Song RJ, Jiang H, Zhang W, Han SF (2022) Oat fiber supplementation alleviates intestinal inflammation and ameliorates intestinal mucosal barrier via acting on gut microbiota-derived metabolites in LDLR(−/−) mice. Nutrition 95:111558. https://doi.org/10.1016/j.nut.2021.111558
Ejima R, Akiyama M, Sato H, Tomioka S, Yakabe K, Kimizuka T, Seki N, Fujimura Y, Hirayama A, Fukuda S, Hase K, Kim YG (2021) Seaweed dietary fiber sodium alginate suppresses the migration of colonic inflammatory monocytes and diet-induced metabolic syndrome via the gut microbiota. Nutrients. https://doi.org/10.3390/nu13082812
Liu J, Wang Z, Mai P, Hao Y, Wang Z, Wang J (2022) Quinoa bran soluble dietary fiber ameliorates dextran sodium sulfate induced ulcerative colitis in BALB/c mice by maintaining intestinal barrier function and modulating gut microbiota. Int J Biol Macromol 216:75–85. https://doi.org/10.1016/j.ijbiomac.2022.06.194
Huang J, Liu D, Wang Y, Liu L, Li J, Yuan J, Jiang Z, Jiang Z, Hsiao WW, Liu H, Khan I, Xie Y, Wu J, Xie Y, Zhang Y, Fu Y, Liao J, Wang W, Lai H, Shi A, Cai J, Luo L, Li R, Yao X, Fan X, Wu Q, Liu Z, Yan P, Lu J, Yang M, Wang L, Cao Y, Wei H, Leung EL (2022) Ginseng polysaccharides alter the gut microbiota and kynurenine/tryptophan ratio, potentiating the antitumour effect of antiprogrammed cell death 1/programmed cell death ligand 1 (anti-PD-1/PD-L1) immunotherapy. Gut 71(4):734–745. https://doi.org/10.1136/gutjnl-2020-321031
Chen LX, Qi YL, Qi Z, Gao K, Gong RZ, Shao ZJ, Liu SX, Li SS, Sun YS (2019) A comparative study on the effects of different parts of Panax ginseng on the immune activity of cyclophosphamide-induced immunosuppressed mice. Molecules. https://doi.org/10.3390/molecules24061096
He LX, Zhang ZF, Zhao J, Li L, Xu T, Bin S, Ren JW, Liu R, Chen QH, Wang JB, Salem MM, Pettinato G, Zhou JR, Li Y (2018) Ginseng oligopeptides protect against irradiation-induced immune dysfunction and intestinal injury. Sci Rep 8(1):13916. https://doi.org/10.1038/s41598-018-32188-6
Hua M, Sun Y, Shao Z, Lu J, Lu Y, Liu Z (2020) Functional soluble dietary fiber from ginseng residue: polysaccharide characterization, structure, antioxidant, and enzyme inhibitory activity. J Food Biochem 44(12):e13524. https://doi.org/10.1111/jfbc.13524
Hua M, Lu J, Qu D, Liu C, Zhang L, Li S, Chen J, Sun Y (2019) Structure, physicochemical properties and adsorption function of insoluble dietary fiber from ginseng residue: a potential functional ingredient. Food Chem 286:522–529. https://doi.org/10.1016/j.foodchem.2019.01.114
Hua M, Liu Z, Sha J, Li S, Dong L, Sun Y (2021) Effects of ginseng soluble dietary fiber on serum antioxidant status, immune factor levels and cecal health in healthy rats. Food Chem 365:130641. https://doi.org/10.1016/j.foodchem.2021.130641
Hua M, Fan ML, Li ZM, Sha JY, Li SS, Sun YS (2021) Ginseng soluble dietary fiber can regulate the intestinal flora structure, promote colon health, affect appetite and glucolipid metabolism in rats. J Funct Foods. https://doi.org/10.1016/j.jff.2021.104534
Patra K, Bose S, Sarkar S, Rakshit J, Jana S, Mukherjee A, Roy A, Mandal DP, Bhattacharjee S (2012) Amelioration of cyclophosphamide induced myelosuppression and oxidative stress by cinnamic acid. Chem Biol Interact 195(3):231–239. https://doi.org/10.1016/j.cbi.2012.01.001
Delmanto RD, de Lima PL, Sugui MM, da Eira AF, Salvadori DM, Speit G, Ribeiro LR (2001) Antimutagenic effect of Agaricus blazei Murrill mushroom on the genotoxicity induced by cyclophosphamide. Mutat Res 496(1–2):15–21. https://doi.org/10.1016/s1383-5718(01)00228-5
Jantan I, Haque MA, Ilangkovan M, Arshad L (2019) Zerumbone from Zingiber zerumbet inhibits innate and adaptive immune responses in Balb/C mice. Int Immunopharmacol 73:552–559. https://doi.org/10.1016/j.intimp.2019.05.035
Zhang J, Zhou HC, He SB, Zhang XF, Ling YH, Li XY, Zhang H, Hou DD (2021) The immunoenhancement effects of sea buckthorn pulp oil in cyclophosphamide-induced immunosuppressed mice. Food Funct 12(17):7954–7963. https://doi.org/10.1039/d1fo01257f
Han X, Bai B, Zhou Q, Niu J, Yuan J, Zhang H, Jia J, Zhao W, Chen H (2020) Dietary supplementation with polysaccharides from Ziziphus jujuba cv. Pozao intervenes in immune response via regulating peripheral immunity and intestinal barrier function in cyclophosphamide-induced mice. Food Funct 11(7):5992–6006. https://doi.org/10.1039/d0fo00008f
Hu JN, Yang JY, Jiang S, Zhang J, Liu Z, Hou JG, Gong XJ, Wang YP, Wang Z, Li W (2021) Panax quinquefolium saponins protect against cisplatin evoked intestinal injury via ROS-mediated multiple mechanisms. Phytomedicine 82:153446. https://doi.org/10.1016/j.phymed.2020.153446
Ren DD, Li SS, Lin HM, Xia YS, Li ZM, Bo PP, Mu R, Zhao LJ, Sun YS (2022) Panax quinquefolius polysaccharides ameliorate antibiotic-associated diarrhoea induced by lincomycin hydrochloride in rats via the MAPK signaling pathways. J Immunol Res 2022:4126273. https://doi.org/10.1155/2022/4126273
Chen DS, Mellman I (2017) Elements of cancer immunity and the cancer-immune set point. Nature 541(7637):321–330. https://doi.org/10.1038/nature21349
Xia L, Oyang L, Lin J, Tan S, Han Y, Wu N, Yi P, Tang L, Pan Q, Rao S, Liang J, Tang Y, Su M, Luo X, Yang Y, Shi Y, Wang H, Zhou Y, Liao Q (2021) The cancer metabolic reprogramming and immune response. Mol Cancer 20(1):28. https://doi.org/10.1186/s12943-021-01316-8
Madondo MT, Quinn M, Plebanski M (2016) Low dose cyclophosphamide: mechanisms of T cell modulation. Cancer Treat Rev 42:3–9. https://doi.org/10.1016/j.ctrv.2015.11.005
Sun Y, Liu Y, Ai C, Song S, Chen X (2019) Caulerpa lentillifera polysaccharides enhance the immunostimulatory activity in immunosuppressed mice in correlation with modulating gut microbiota. Food Funct 10(7):4315–4329. https://doi.org/10.1039/c9fo00713j
Camilleri M (2019) Leaky gut: mechanisms, measurement and clinical implications in humans. Gut 68(8):1516–1526. https://doi.org/10.1136/gutjnl-2019-318427
Dong J, Liang W, Wang T, Sui J, Wang J, Deng Z, Chen D (2019) Saponins regulate intestinal inflammation in colon cancer and IBD. Pharmacol Res 144:66–72. https://doi.org/10.1016/j.phrs.2019.04.010
Schulz O, Hammerschmidt SI, Moschovakis GL, Forster R (2016) Chemokines and chemokine receptors in lymphoid tissue dynamics. Annu Rev Immunol 34:203–242. https://doi.org/10.1146/annurev-immunol-041015-055649
Ilangkovan M, Jantan I, Bukhari SN (2016) Phyllanthin from Phyllanthus amarus inhibits cellular and humoral immune responses in Balb/C mice. Phytomedicine 23(12):1441–1450. https://doi.org/10.1016/j.phymed.2016.08.002
Liu YH, Qin HY, Zhong YY, Li S, Wang HJ, Wang H, Chen LL, Tang X, Li YL, Qian ZY, Li HY, Zhang L, Chen T (2021) Neutral polysaccharide from Panax notoginseng enhanced cyclophosphamide antitumor efficacy in hepatoma H22-bearing mice. BMC Cancer 21(1):37. https://doi.org/10.1186/s12885-020-07742-z
Villarreal DO, L’Huillier A, Armington S, Mottershead C, Filippova EV, Coder BD, Petit RG, Princiotta MF (2018) Targeting CCR8 induces protective antitumor immunity and enhances vaccine-induced responses in colon cancer. Cancer Res 78(18):5340–5348. https://doi.org/10.1158/0008-5472.CAN-18-1119
Chen X, Sun W, Xu B, Wu E, Cui Y, Hao K, Zhang G, Zhou C, Xu Y, Li J, Si H (2021) Polysaccharides from the roots of Millettia speciosa champ modulate gut health and ameliorate cyclophosphamide-induced intestinal injury and immunosuppression. Front Immunol 12:766296. https://doi.org/10.3389/fimmu.2021.766296
Linard C, Billiard F, Benderitter M (2012) Intestinal irradiation and fibrosis in a Th1-deficient environment. Int J Radiat Oncol Biol Phys 84(1):266–273. https://doi.org/10.1016/j.ijrobp.2011.11.027
Han Y, Zhang Y, Ouyang K, Chen L, Zhao M, Wang W (2022) Sulfated Cyclocarya paliurus polysaccharides improve immune function of immunosuppressed mice by modulating intestinal microbiota. Int J Biol Macromol 212:31–42. https://doi.org/10.1016/j.ijbiomac.2022.05.110
Zhuang Y, Wu H, Wang X, He J, He S, Yin Y (2019) Resveratrol attenuates oxidative stress-induced intestinal barrier injury through PI3K/Akt-mediated Nrf2 signaling pathway. Oxid Med Cell Longev 2019:7591840. https://doi.org/10.1155/2019/7591840
Gunther C, Buchen B, Neurath MF, Becker C (2014) Regulation and pathophysiological role of epithelial turnover in the gut. Semin Cell Dev Biol 35:40–50. https://doi.org/10.1016/j.semcdb.2014.06.004
Gunther C, Neumann H, Neurath MF, Becker C (2013) Apoptosis, necrosis and necroptosis: cell death regulation in the intestinal epithelium. Gut 62(7):1062–1071. https://doi.org/10.1136/gutjnl-2011-301364
Rincon M, Flavell RA, Davis RA (2000) The JNK and P38 MAP kinase signaling pathways in T cell-mediated immune responses. Free Radic Biol Med 28(9):1328–1337. https://doi.org/10.1016/s0891-5849(00)00219-7
Efferth T, Oesch F (2021) The immunosuppressive activity of artemisinin-type drugs towards inflammatory and autoimmune diseases. Med Res Rev 41(6):3023–3061. https://doi.org/10.1002/med.21842
Nenci A, Becker C, Wullaert A, Gareus R, van Loo G, Danese S, Huth M, Nikolaev A, Neufert C, Madison B, Gumucio D, Neurath MF, Pasparakis M (2007) Epithelial NEMO links innate immunity to chronic intestinal inflammation. Nature 446(7135):557–561. https://doi.org/10.1038/nature05698
Acknowledgements
This work was funded by National Key Research and Development Program (2023YFD1601600), Central Public-interest Scientific Institution Basal Research Fund (Grant No.: CAAS-ASTIP-2021-ISAPS), Jilin Province Science and Technology Development Program (202301ZYTS352), Jilin Provincial Department of Human Resources and Social Security (2023QN16).
Author information
Authors and Affiliations
Contributions
Ji-yue Sha: methodology, writing—original draft, project administration. Ke-Cheng Chen: conceptualization, writing—original draft. Zheng-bo Liu: investigation, data curation. Wei Li: conceptualization, supervision. Yu-shun Lu: data curation, methodology. Shuang Liu: software, validation, supervision. Jian-kai Ma: software, resources. Di Qu: software, resources. Yin-shi Sun: funding acquisition; writing review & editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
All experimental animal programs were performed in strict accordance with the Guide for the Care and Use of Laboratory Animals (2016) and approved by the Laboratory Animal Care and Ethics Committee of the Institute of Special Animals and Plants, Chinese Academy of Agricultural Sciences (NO. ISAPSAEC-2022-021).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sha, Jy., Chen, KC., Liu, Zb. et al. Ginseng-DF ameliorates intestinal mucosal barrier injury and enhances immunity in immunosuppressed mice by regulating MAPK/NF-κB signaling pathways. Eur J Nutr (2024). https://doi.org/10.1007/s00394-024-03378-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00394-024-03378-y