Abstract
Purpose
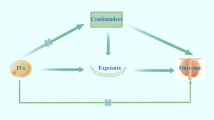
Previous observational studies showed that serum uric acid (SUA) was associated with prostate cancer, but the causal relationship is unclear. This study aimed to explore the potential causal association between SUA and prostate cancer risk using Mendelian randomization (MR) analyses in the East Asian populations.
Methods
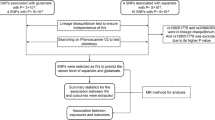
Publicly available summary-level genome-wide association studies (GWAS) data on SUA were obtained from a genome-wide meta-analysis of three Japanese cohorts (121,745 subjects). The GWAS data on prostate cancer were derived from Biobank Japan (109,347 subjects with 5,408 cases and 103,939 controls). A total of 34 SUA-related single-nucleotide polymorphisms (SNPs) (P value < 5 × 10–8) were identified as instrumental variables. The inverse variance weighted method was used as the primary method to compute the odds ratios (ORs) and 95% confidence intervals (95% CIs) for per standard deviation increase in SUA. MR Egger, weighted median, and weighted mode were also applied to test the robustness of the results.
Results
Genetically predicted SUA was positively associated with prostate cancer risk using inverse variance weighted (OR = 1.12; 95% CI 1.00–1.26; P = 0.043). The positive association was robust when MR Egger (OR = 1.16; 95% CI 1.01–1.34; P = 0.048), weighted median (OR = 1.18; 95% CI 1.03–1.36; P = 0.018), and weighted mode (OR = 1.14; 95% CI 1.01–1.29; P = 0.041) were used.
Conclusion
There were potential causal associations between higher genetically predicted SUA levels and increased prostate cancer risk. Further, MR studies with more valid SNPs and more cancer cases are needed. Validation of the findings is also recommended.
Similar content being viewed by others
Data availability
All data used in this study are publicly available without any access restrictions. One can also contact the corresponding author if they would like to assess them.
References:
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Jiang M, Ren L, Chen S, Li G (2021) Serum uric acid levels and risk of eight site-specific cancers: a Mendelian randomization study. Front Genet 12:608311. https://doi.org/10.3389/fgene.2021.608311
Li L, Zhang Y, Zeng C (2020) Update on the epidemiology, genetics, and therapeutic options of hyperuricemia. Am J Transl Res 12:3167–3181
Wang H, Zhang H, Sun L, Guo W (2018) Roles of hyperuricemia in metabolic syndrome and cardiac-kidney-vascular system diseases. Am J Transl Res 10:2749–2763
Battelli MG, Polito L, Bortolotti M, Bolognesi A (2016) Xanthine oxidoreductase in cancer: more than a differentiation marker. Cancer Med 5:546–557. https://doi.org/10.1002/cam4.601
Borghi C, Agabiti-Rosei E, Johnson RJ, Kielstein JT, Lurbe E, Mancia G et al (2020) Hyperuricaemia and gout in cardiovascular, metabolic and kidney disease. Eur J Intern Med 80:1–11. https://doi.org/10.1016/j.ejim.2020.07.006
Kim YR, Choi CK, Lee Y-H, Choi S-W, Kim H-Y, Shin M-H et al (2021) Association between albumin, total bilirubin, and uric acid serum levels and the risk of cancer: a prospective study in a Korean population. Yonsei Med J 62:792–798. https://doi.org/10.3349/ymj.2021.62.9.792
Wang A, Barber JR, Tin A, De Marzo AM, Kottgen A, Joshu CE et al (2019) Serum urate, genetic variation, and prostate cancer risk: atherosclerosis risk in communities (ARIC) study. Cancer Epidemiol Biomark Prev 28:1259–1261. https://doi.org/10.1158/1055-9965.Epi-19-0161
Davies NM, Holmes MV, Davey Smith G (2018) Reading Mendelian randomisation studies: a guide, glossary, and checklist for clinicians. BMJ 362:k601. https://doi.org/10.1136/bmj.k601
Skrivankova VW, Richmond RC, Woolf BAR et al (2021) Strengthening the reporting of observational studies in epidemiology using mendelian randomisation (STROBE-MR): explanation and elaboration. BMJ 375:n2233. https://doi.org/10.1136/bmj.n2233
Hemani G, Zheng J, Elsworth B et al (2018) The MR-base platform supports systematic causal inference across the human phenome. Elife 7:e34408. https://doi.org/10.7554/eLife.34408
Nakatochi M, Kanai M, Nakayama A, Hishida A, Kawamura Y, Ichihara S et al (2019) Genome-wide meta-analysis identifies multiple novel loci associated with serum uric acid levels in Japanese individuals. Commun Biol 2:115. https://doi.org/10.1038/s42003-019-0339-0
Ishigaki K, Akiyama M, Kanai M, Takahashi A, Kawakami E, Sugishita H et al (2020) Large-scale genome-wide association study in a Japanese population identifies novel susceptibility loci across different diseases. Nat Genet 52:669–679. https://doi.org/10.1038/s41588-020-0640-3
Xu L, Hao YT (2017) Effect of handgrip on coronary artery disease and myocardial infarction: a Mendelian randomization study. Sci Rep 7:954. https://doi.org/10.1038/s41598-017-01073-z
Mozaffarian D, Kabagambe EK, Johnson CO, Lemaitre RN, Manichaikul A, Sun Q et al (2015) Genetic loci associated with circulating phospholipid trans fatty acids: a meta-analysis of genome-wide association studies from the CHARGE consortium. Am J Clin Nutr 101:398–406. https://doi.org/10.3945/ajcn.114.094557
Burgess S, Davies NM, Thompson SG (2016) Bias due to participant overlap in two-sample Mendelian randomization. Genet Epidemiol 40:597–608. https://doi.org/10.1002/gepi.21998
Burgess S (2014) Sample size and power calculations in Mendelian randomization with a single instrumental variable and a binary outcome. Int J Epidemiol 43:922–929. https://doi.org/10.1093/ije/dyu005
Brion MJ, Shakhbazov K, Visscher PM (2013) Calculating statistical power in Mendelian randomization studies. Int J Epidemiol 42:1497–1501. https://doi.org/10.1093/ije/dyt179
Kolonel LN, Yoshizawa C, Nomura AM, Stemmermann GN (1994) Relationship of serum uric acid to cancer occurrence in a prospective male cohort. Cancer Epidemiol Biomark Prev 3:225–228
Taghizadeh N, Vonk JM, Boezen HM (2014) Serum uric acid levels and cancer mortality risk among males in a large general population-based cohort study. Cancer Causes Control 25:1075–1080. https://doi.org/10.1007/s10552-014-0408-0
Fini MA, Elias A, Johnson RJ, Wright RM (2012) Contribution of uric acid to cancer risk, recurrence, and mortality. Clin Transl Med 1:16. https://doi.org/10.1186/2001-1326-1-16
Ames BN, Cathcart R, Schwiers E, Hochstein P (1981) Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: a hypothesis. Proc Natl Acad Sci USA 78:6858–6862. https://doi.org/10.1073/pnas.78.11.6858
Tong L, Chuang CC, Wu S, Zuo L (2015) Reactive oxygen species in redox cancer therapy. Cancer Lett 367:18–25. https://doi.org/10.1016/j.canlet.2015.07.008
Seifried HE, Anderson DE, Fisher EI, Milner JA (2007) A review of the interaction among dietary antioxidants and reactive oxygen species. J Nutr Biochem 18:567–579. https://doi.org/10.1016/j.jnutbio.2006.10.007
Conti DV, Darst BF, Moss LC, Saunders EJ, Sheng X, Chou A et al (2021) Trans-ancestry genome-wide association meta-analysis of prostate cancer identifies new susceptibility loci and informs genetic risk prediction. Nat Genet 53:65–75. https://doi.org/10.1038/s41588-020-00748-0
Acknowledgements
We would like to express our gratitude to the Japan Multi-institutional Collaborative Cohort (J-MICC), the Kita-Nagoya Genomic Epidemiology (KING), and the BioBank Japan (BBJ) for providing publicly available summary-level genome-wide association studies (GWAS) data on serum uric acid. We also want to thank the BBJ for the publicly available summary-level GWAS data on prostate cancer, body mass index, smoking, total cholesterol, low-density-lipoprotein cholesterol, high-density-lipoprotein cholesterol, triglyceride, diastolic blood pressure, systolic blood pressure, mean arterial pressure, congestive heart failure, and type 2 diabetes.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
YYD conceived and designed this study. YYD conducted the data acquisition, analyzed the data, and drafted the manuscript. JJH proofread the data and manuscript. MCSW critically revised the manuscript. All authors approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
All related studies used in this study were approved by appropriate ethics review boards. Written informed consents were obtained from all participants. This study did not exceed the scope of the original ethics committee approval.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deng, Y., Huang, J. & Wong, M.C.S. Association between serum uric acid and prostate cancer risk in East Asian populations: a Mendelian randomization study. Eur J Nutr 62, 1323–1329 (2023). https://doi.org/10.1007/s00394-022-03076-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-022-03076-7