Abstract
Purpose
To understand the effects of consuming high-fat and low-fat dairy products on postprandial cardiometabolic risk factors and intestinal immune function, we used an established low birthweight (LBW) swine model of diet-induced insulin resistance.
Methods
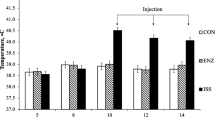
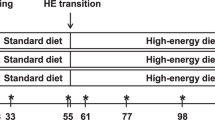
LBW piglets were randomized to consume one of the 3 experimental high fat diets and were fed for a total of 7 weeks: (1) Control high fat (LBW–CHF), (2) CHF diet supplemented with 3 servings of high-fat dairy (LBW–HFDairy) and (3) CHF diet supplemented with 3 servings of low-fat dairy (LBW–LFDairy). As comparison groups, normal birthweight (NBW) piglets were fed a CHF (NBW–CHF) or standard pig grower diet (NBW–Chow). At 11 weeks of age, all piglets underwent an established modified oral glucose and fat tolerance test. At 12 weeks of age, piglets were euthanized and ex vivo cytokine production by cells isolated from mesenteric lymph node (MLN) stimulated with mitogens was assessed.
Results
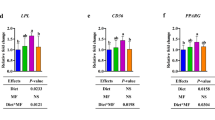
Dairy consumption did not modulate postprandial plasma lipid, inflammatory markers and glucose concentrations. A lower production of IL-2 and TNF-α after pokeweed mitogen (PWM) stimulation was observed in LBW–CHF vs NBW–Chow (P < 0.05), suggesting impaired MLN T cell function. While feeding high-fat dairy had minimal effects, feeding low-fat dairy significantly improved the production of IL-2 and TNF-α after PWM stimulation (P < 0.05).
Conclusions
Irrespective of fat content, dairy had a neutral effect on postprandial cardiometabolic risk factors. Low-fat dairy products improved intestinal T cell function to a greater extent than high-fat dairy in this swine model of obesity and insulin resistance.




Similar content being viewed by others
Change history
11 October 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00394-023-03263-0
Abbreviations
- APC:
-
Antigen presenting cell
- AUC:
-
Area under curve
- CHF:
-
Control high-fat diet
- CI:
-
Confidence interval
- CV:
-
Coefficient of variation
- CVD:
-
Cardiovascular disease
- ELISA:
-
Enzyme-linked immunosorbent assay
- FSC:
-
Forward scatter
- GALT:
-
Gut-associated lymphoid tissue
- HFDairy:
-
High-fat dairy diet
- IEC:
-
Intestinal epithelial cells
- IFN-γ:
-
Interferon-gamma
- IL:
-
Interleukin
- LBW:
-
Low birthweight
- LDL-C:
-
Low-density lipoprotein cholesterol
- LFDairy:
-
Low-fat dairy diet
- LPS:
-
Lipopolysaccharide
- MFI:
-
Median florescence intensity
- MFGM:
-
Milk fat globular membrane
- MHC:
-
Major histocompatibility complex
- MLN:
-
Mesenteric lymph node
- MOGTT:
-
Modified oral glucose and fat tolerance test
- NBW:
-
Normal birthweight
- PBMC:
-
Peripheral blood mononuclear cell
- PBS:
-
Phosphate-buffered saline
- PC:
-
Phosphatidylcholine
- PMA-I:
-
Phorbol 12-myristate 13-acetate plus ionomycin
- PP:
-
Peyer's patches
- PWM:
-
Pokeweed mitogen
- SEM:
-
Standard error of the mean
- SM:
-
Sphingomyelin
- S–MCFA:
-
Short–medium chain fatty acid
- SRTC:
-
Swine research technology centre
- SSC:
-
Side scatter
- TC:
-
Total cholesterol
- TG:
-
Triglycerides
- Th:
-
T helper cell
- TLR-4:
-
Toll-like receptor 4
- TNF-α:
-
Tumor necrosis factor-alpha
- Treg:
-
T regulatory cell
References
Calder PC (2013) Feeding the immune system. Proc Nutr Soc 72:299–309. https://doi.org/10.1017/S0029665113001286
Soderholm AT, Pedicord VA (2019) Intestinal epithelial cells: at the interface of the microbiota and mucosal immunity. Immunology 158:267–280. https://doi.org/10.1111/imm.13117
Winer DA, Luck H, Tsai S, Winer S (2016) The intestinal immune system in obesity and insulin resistance. Cell Metab 23:413–426. https://doi.org/10.1016/j.cmet.2016.01.003
Luck H, Tsai S, Chung J, Clemente-Casares X, Ghazarian M, Revelo XS et al (2015) Regulation of obesity-related insulin resistance with gut anti-inflammatory agents. Cell Metab 21:527–542. https://doi.org/10.1016/j.cmet.2015.03.001
Ruth MR, Proctor SD, Field CJ (2009) Effects of feeding fish oil on mesenteric lymph node cytokine responses in obese leptin receptor-deficient JCR:LA-cp rats. Int J Obes 33:96–103. https://doi.org/10.1038/ijo.2008.227
Blewett HJ, Gerdung CA, Ruth MR, Proctor SD, Field CJ (2009) Vaccenic acid favourably alters immune function in obese JCR:LA-cp rats. Br J Nutr 102:526–536. https://doi.org/10.1017/S0007114509231722
Bozzetto L, Della-Pepa G, Vetrani C, Rivellese AA (2020) Dietary impact on postprandial lipemia. Front Endocrinol 11:337. https://doi.org/10.3389/fendo.2020.00337
Mangat R, Warnakula S, Borthwick F, Hassanali Z, Uwiera RRE, Russell JC et al (2012) Arterial retention of remnant lipoproteins ex vivo is increased in insulin resistance because of increased arterial biglycan and production of cholesterol-rich atherogenic particles that can be improved by ezetimibe in the JCR:LA-cp rat. J Am Heart Assoc 1:e003434. https://doi.org/10.1161/JAHA.112.003434
van Oostrom AJHHM, Sijmonsma TP, Rabelink TJ, van Asbeck BS, Castro Cabezas M (2003) Postprandial leukocyte increase in healthy subjects. Metabolism 52:199–202. https://doi.org/10.1053/meta.2003.50037
Alipour A, van Oostrom AJHHM, Izraeljan A, Verseyden C, Collins JM, Frayn KN et al (2008) Leukocyte activation by triglyceride-rich lipoproteins. Arterioscler Thromb Vasc Biol 28:792–797. https://doi.org/10.1161/ATVBAHA.107.159749
Kris-Etherton PM, Fleming JA (2015) Emerging nutrition science on fatty acids and cardiovascular disease: nutritionists’ perspectives. Adv Nutr 6:326S-S337. https://doi.org/10.3945/an.114.006981
Colonic T, Homeostasis C, Smith PM, Howitt MR, Panikov N, Michaud M et al (2013) The microbial metabolites, short-chain fatty acids, regulate colonic treg cell homeostasis. Science 341:569–573. https://doi.org/10.1126/science.1241165
Peng L, Li ZR, Green RS, Holzman IR, Lin J (2009) Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers. J Nutr 139:1619–1625. https://doi.org/10.3945/jn.109.104638
Norris GH, Jiang C, Ryan J, Porter CM, Blesso CN (2016) Milk sphingomyelin improves lipid metabolism and alters gut microbiota in high fat diet-fed mice. J Nutr Biochem 30:93–101. https://doi.org/10.1016/j.jnutbio.2015.12.003
Li Y, Wu J, Niu Y, Chen H, Tang Q, Zhong Y et al (2019) Milk fat globule membrane inhibits NLRP3 inflammasome activation and enhances intestinal barrier function in a rat model of short bowel. J Parenter Enter Nutr 43:677–685. https://doi.org/10.1002/jpen.1435
Barrera JA, Patel D, Field C, Pouliot Y, Goruk S, Jacobs R, et al (2021) A Diet High in Lipid Soluble Forms of Choline Modulates Gut-Associated Immune Function in Sprague-Dawley Dams. Current Developments in Nutrition 5 (Supplement_2)
Imamura F, Fretts A, Marklund M, Ardisson Korat AV, Yang WS, Lankinen M et al (2018) Fatty acid biomarkers of dairy fat consumption and incidence of type 2 diabetes: a pooled analysis of prospective cohort studies. PLoS Med 15:e1002670. https://doi.org/10.1371/journal.pmed.1002670
She Y, Mangat R, Tsai S, Proctor SD, Richard C (2022) The interplay of obesity, dyslipidemia and immune dysfunction: a brief overview on pathophysiology, animal models, and nutritional modulation. Front Nutr 9:840209. https://doi.org/10.3389/fnut.2022.840209
Fontaine MA, Diane A, Singh VP, Mangat R, Krysa JA, Nelson R et al (2019) Low birth weight causes insulin resistance and aberrant intestinal lipid metabolism independent of microbiota abundance in Landrace-Large White pigs. FASEB J 33:9250–9262. https://doi.org/10.1096/fj.201801302RR
She Y, Wang K, Makarowski A, Mangat R, Tsai S, Willing BP et al (2022) Effect of high-fat and low-fat dairy products on cardiometabolic risk factors and immune function in a low birthweight swine model of diet-induced insulin resistance. Front Nutr 9:923120. https://doi.org/10.3389/fnut.2022.923120
Singh VP, Fontaine MA, Mangat R, Fouhse JM, Diane A, Willing BP et al (2021) High vaccenic acid content in beef fat attenuates high fat and high carbohydrate western diet induced changes in lipid metabolism and gut microbiota in pigs. Microorganisms 9:2517. https://doi.org/10.3390/microorganisms9122517
Richard C, Lewis ED, Zhao YY, Asomaning J, Jacobs RL, Field CJ et al (2016) Measurement of the total choline content in 48 commercial dairy products or dairy alternatives. J Food Compos Anal 45:1–8. https://doi.org/10.1016/j.jfca.2015.09.009
Azarcoya-Barrera J, Field CJ, Goruk S, Makarowski A, Curtis JM, Pouliot Y et al (2021) Buttermilk: an important source of lipid soluble forms of choline that influences the immune system development in Sprague-Dawley rat offspring. Eur J Nutr 60:2807–2818. https://doi.org/10.1007/s00394-020-02462-3
Azarcoya-Barrera J, Goruk S, Lewis ED, Pouliot Y, Curtis JM, Steele R et al (2020) Feeding buttermilk-derived choline forms during gestation and lactation modulates ex vivo t-cell response in rat dams. J Nutr 150:1958–1965. https://doi.org/10.1093/jn/nxaa089
le Huërou-Luron I, Bouzerzour K, Ferret-Bernard S, Ménard O, le Normand L, Perrier C et al (2018) A mixture of milk and vegetable lipids in infant formula changes gut digestion, mucosal immunity and microbiota composition in neonatal piglets. Eur J Nutr 57:463–476. https://doi.org/10.1007/s00394-016-1329-3
Ghezzal S, Postal BG, Quevrain E, Brot L, Seksik P, Leturque A et al (2020) Palmitic acid damages gut epithelium integrity and initiates inflammatory cytokine production. Biochim Biophys Acta Mol Cell Biol Lipids 1865:158530. https://doi.org/10.1016/j.bbalip.2019.158530
Schmidt KA, Cromer G, Burhans MS, Kuzma JN, Hagman DK, Fernando I et al (2021) The impact of diets rich in low-fat or full-fat dairy on glucose tolerance and its determinants: a randomized controlled trial. Am J Clin Nutr 113:534–547. https://doi.org/10.1093/ajcn/nqaa301
O’Connor S, Julien P, Weisnagel SJ, Gagnon C, Rudkowska I (2019) Impact of a high intake of dairy product on insulin sensitivity in hyperinsulinemic adults: a crossover randomized controlled trial. Curr Dev Nutr 3:083. https://doi.org/10.1093/cdn/nzz083
Engel S, Tholstrup T, Bruun JM, Astrup A, Richelsen B, Raben A (2018) Effect of high milk and sugar-sweetened and non-caloric soft drink intake on insulin sensitivity after 6 months in overweight and obese adults: a randomized controlled trial. Eur J Clin Nutr 72:358–366. https://doi.org/10.1038/s41430-017-0006-9
Thorning TK, Raziani F, Bendsen NT, Astrup A, Tholstrup T, Raben A (2015) Diets with high-fat cheese, high-fat meat, or carbohydrate on cardiovascular risk markers in overweight postmenopausal women: a randomized crossover trial. Am J Clin Nutr 102:573–581. https://doi.org/10.3945/ajcn.115.109116
Bohl M, Bjørnshave A, Larsen MK, Gregersen S, Hermansen K (2017) The effects of proteins and medium-chain fatty acids from milk on body composition, insulin sensitivity and blood pressure in abdominally obese adults. Eur J Clin Nutr 71:76–82. https://doi.org/10.1038/ejcn.2016.207
Clemente G, Mancini M, Nazzaro F, Lasorella G, Rivieccio A, Palumbo AM et al (2003) Effects of different dairy products on postprandial lipemia. Nutr Metab Cardiovasc Dis 13:377–383. https://doi.org/10.1016/s0939-4753(03)80007-8
Bohl M, Bjørnshave A, Rasmussen K, v., Schioldan AG, Amer B, Larsen MK, et al (2015) Dairy proteins, dairy lipids, and postprandial lipemia in persons with abdominal obesity (DairyHealth): a 12-wk, randomized, parallel-controlled, double-blinded, diet intervention study. Am J Clin Nutr 101:870–878. https://doi.org/10.3945/ajcn.114.097923
Wang K, She Y, Mangat R, Makarowski A, Roy B, Bruce H, et al (2021) Exploring Increased Intestinal Lipid Absorption and Identifying Strategies to Improve Pork Quality in Low-Birth-Weight Swine. Curr Dev Nutr 5 (Supplement_2)
Schmid A, Petry N, Walther B, Bütikofer U, Luginbüh W, Gille D et al (2015) Inflammatory and metabolic responses to high-fat meals with and without dairy products in men. Br J Nutr 113:1853–1861. https://doi.org/10.1017/S0007114515000677
Erridge C, Attina T, Spickett CM, Webb DJ (2007) A high-fat meal induces low-grade endotoxemia: evidence of a novel mechanism of postprandial inflammation. Am J Clin Nutr 86:1286–1292. https://doi.org/10.1093/ajcn/86.5.1286
Herieka M, Erridge C (2014) High-fat meal induced postprandial inflammation. Mol Nutr Food Res 58:136–146. https://doi.org/10.1002/mnfr.201300104
van Oostrom AJHHM, Sijmonsma TP, Verseyden C, Jansen EHJM, de Koning EJP, Rabelink TJ et al (2003) Postprandial recruitment of neutrophils may contribute to endothelial dysfunction. J Lipid Res 44:576–583. https://doi.org/10.1194/jlr.M200419-JLR200
Fritsche KL (2015) The science of fatty acids and inflammation. Adv Nutr 6:293S-301S. https://doi.org/10.3945/an.114.006940
Mkumbuzi L, Mfengu MMO, Engwa GA, Sewani-Rusike CR (2020) Insulin resistance is associated with gut permeability without the direct influence of obesity in young adults. Diabetes Metab Syndr Obes 13:2997–3008. https://doi.org/10.2147/DMSO.S256864
de Loera-Rodriguez CO, Delgado-Rizo V, Alvarado-Navarro A, Agraz-Cibrian JM, Segura-Ortega JE, Fafutis-Morris M (2014) Over-expression of TLR4-CD14, pro-inflammatory cytokines, metabolic markers and NEFAs in obese non-diabetic Mexicans. J Inflamm 11:39. https://doi.org/10.1186/s12950-014-0039-y
Akashi S, Shimazu R, Ogata H, Nagai Y, Takeda K, Kimoto M et al (2000) Cutting edge: cell surface expression and lipopolysaccharide signaling via the toll-like receptor 4-MD-2 complex on mouse peritoneal macrophages. J Immunol 64:3471–3475. https://doi.org/10.4049/jimmunol.164.7.3471
Vergadi E, Vaporidi K, Tsatsanis C (2018) Regulation of endotoxin tolerance and compensatory anti-inflammatory response syndrome by non-coding RNAs. Front Immunol 9:2705. https://doi.org/10.3389/fimmu.2018.02705
Lamas O, Martinez JA, Marti A (2002) T-helper lymphopenia and decreased mitogenic response in cafeteria diet-induced obese rats. Nutr Res 22:497–506
Richard C, Wadowski M, Goruk S, Cameron L, Sharma AM, Field CJ (2017) Individuals with obesity and type 2 diabetes have additional immune dysfunction compared with obese individuals who are metabolically healthy. BMJ Open Diabetes Res Care 5:e000379. https://doi.org/10.1136/bmjdrc-2016-000379
Ross SH, Cantrell DA (2018) Signaling and Function of Interleukin-2 in T Lymphocytes. Annu Rev Immunol 36:411–433. https://doi.org/10.1146/annurev-immunol-042617-053352
Zelová H, Hošek J (2013) TNF-α signalling and inflammation: Interactions between old acquaintances. Inflamm Res 62:641–651. https://doi.org/10.1007/s00011-013-0633-0
Tucureanu MM, Rebleanu D, Constantinescu CA, Deleanu M, Voicu G, Butoi E et al (2018) Lipopolysaccharide-induced inflammation in monocytes/macrophages is blocked by liposomal delivery of Gi-protein inhibitor. Int J Nanomed 13:63–76. https://doi.org/10.2147/IJN.S150918
Tibaes JRB, Silva MIB, Cervantes PB, Wollin B, Makarowski A, Vine D et al (2021) Expression of CD25 in individuals with obesity with or without insulin resistance after following a north american diet. Curr Dev Nutr 5(2):1122
Acknowledgements
This study was supported by grants from Dairy Farmers of Canada (RES0042193) and the Natural Sciences and Engineering Research Council of Canada Discovery Grants to both CR and SDP. YS is a recipient of a Ph.D. scholarship from China Scholarship Council (201807980001).
Funding
Dairy Farmers of Canada (Grant No. RES0042193), Natural Sciences and Engineering Research Council of Canada (Grant No. RES0038933).
Author information
Authors and Affiliations
Contributions
CR and SDP designed and obtained funding for this study. ST and BPW provided expertise on immunology and animal model. YS, AM, RM and KW conducted research and analyzed data. YS performed the statistical analysis and wrote the manuscript under the supervision of CR and SDP. CR has primary responsibility for final content. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
She, Y., Wang, K., Makarowski, A. et al. Low-fat dairy consumption improves intestinal immune function more than high-fat dairy in a diet-induced swine model of insulin resistance. Eur J Nutr 62, 699–711 (2023). https://doi.org/10.1007/s00394-022-03013-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-022-03013-8