Abstract
Purpose
Increased levels of uric acid (UA), which is mainly excreted through the kidneys, are independently associated with higher mortality in end-stage renal disease (ESRD) patients. The uricolysis of gut microbiota plays an important role in extrarenal excretion of UA. This study aimed to examine the effect of inulin-type prebiotics (a type of fermentable dietary fiber) on intestinal microbiota modulation and serum UA levels in ESRD patients.
Methods
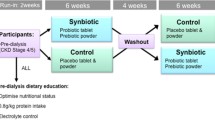
Continuous ambulatory peritoneal dialysis (CAPD) patients were recruited to a randomized, double-blind, placebo-controlled crossover trial of 12-week inulin-type prebiotics. Participants were visited before and after treatment with prebiotics or placebo. Serum UA levels, dietary purine intake, serum xanthine oxidase (XO) activity, daily “renal excretion” of UA, and fecal UA degradation capability were measured at each visit. Fecal metagenomic analysis was conducted to assess microbial composition and function.
Results
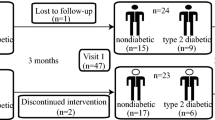
Sixteen participants (mean age = 37 y; 10 men and 6 women) completed the trial, and 64 specimens were analyzed. The average concentration of serum UA decreased by approximately 10% in the prebiotic intervention group in comparison to the placebo group (p = 0.047) without an increase in daily “renal excretion” of UA via urine and dialysate. There were no significant changes in purine intake or activity of XO. Notably, enhanced fecal UA degradation was observed after prebiotic intervention (p = 0.041), and the ratio of Firmicutes/Bacteroidetes, which was positively associated with fecal UA degradation, increased in the prebiotic period (p = 0.032). Furthermore, prebiotics enriched purine-degrading species in the gut microbiota, including unclassified_o_Clostridiales, Clostridium sp. CAG:7, Clostridium sp. FS41, Clostridium citroniae, Anaerostipes caccae, and Clostridium botulinum.
Conclusions
Inulin-type prebiotics is a promising therapeutic candidate to reduce serum UA levels in renal failure patients, and this urate-lowering effect could possibly be attributed to intestinal microbial degradation of UA.
Trial registry
This study was registered at the Chinese Clinical Trials Registry (http://www.chictr.org.cn/), registration ID: ChiCTR-INR-17013739, registration date: 6th Dec 2017.





Similar content being viewed by others
Data availability
Sequence data are available for download from the Sequence Read Archive with accession number PRJNA750234.
References
Ko J, Kang HJ, Kim DA, Kim MJ, Ryu ES, Lee S et al (2019) Uric acid induced the phenotype transition of vascular endothelial cells via induction of oxidative stress and glycocalyx shedding. Faseb j 33:13334–13345. https://doi.org/10.1096/fj.201901148R
Wu AH, Gladden JD, Ahmed M, Ahmed A, Filippatos G (2016) Relation of serum uric acid to cardiovascular disease. Int J Cardiol 213:4–7. https://doi.org/10.1016/j.ijcard.2015.08.110
Ichida K, Matsuo H, Takada T, Nakayama A, Murakami K, Shimizu T et al (2012) Decreased extra-renal urate excretion is a common cause of hyperuricemia. Nat Commun 3:764. https://doi.org/10.1038/ncomms1756
Saran R, Robinson B, Abbott KC, Agodoa LY, Albertus P, Ayanian J et al (2017) US renal data system 2016 annual data report: epidemiology of kidney disease in the United States. Am J Kidney Dis 69:A7-a8. https://doi.org/10.1053/j.ajkd.2016.12.004
Xia X, He F, Wu X, Peng F, Huang F, Yu X (2014) Relationship between serum uric acid and all-cause and cardiovascular mortality in patients treated with peritoneal dialysis. Am J Kidney Dis 64:257–264. https://doi.org/10.1053/j.ajkd.2013.08.027
Strilchuk L, Fogacci F, Cicero AF (2019) Safety and tolerability of available urate-lowering drugs: a critical review. Expert Opin Drug Saf 18:261–271. https://doi.org/10.1080/14740338.2019.1594771
Sato Y, Feig DI, Stack AG, Kang DH, Lanaspa MA, Ejaz AA et al (2019) The case for uric acid-lowering treatment in patients with hyperuricaemia and CKD. Nat Rev Nephrol 15:767–775. https://doi.org/10.1038/s41581-019-0174-z
White WB, Saag KG, Becker MA, Borer JS, Gorelick PB, Whelton A et al (2018) Cardiovascular safety of febuxostat or allopurinol in patients with gout. N Engl J Med 378:1200–1210. https://doi.org/10.1056/NEJMoa1710895
Yun Y, Yin H, Gao Z, Li Y, Gao T, Duan J et al (2017) Intestinal tract is an important organ for lowering serum uric acid in rats. PLoS One 12:e0190194. https://doi.org/10.1371/journal.pone.0190194
Shao T, Shao L, Li H, Xie Z, He Z, Wen C (2017) Combined signature of the fecal microbiome and metabolome in patients with gout. Front Microbiol 8:268. https://doi.org/10.3389/fmicb.2017.00268
Guo Z, Zhang J, Wang Z, Ang KY, Huang S, Hou Q et al (2016) Intestinal microbiota distinguish gout patients from healthy humans. Sci Rep 6:20602. https://doi.org/10.1038/srep20602
Guo Y, Yu Y, Li H, Ding X, Li X, Jing X et al (2021) Inulin supplementation ameliorates hyperuricemia and modulates gut microbiota in Uox-knockout mice. Eur J Nutr 60:2217–2230. https://doi.org/10.1007/s00394-020-02414-x
Wang J, Chen Y, Zhong H, Chen F, Regenstein J, Hu X et al (2021) The gut microbiota as a target to control hyperuricemia pathogenesis: Potential mechanisms and therapeutic strategies. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2021.1874287
Gibson GR, Hutkins R, Sanders ME, Prescott SL, Reimer RA, Salminen SJ et al (2017) Expert consensus document: the International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol 14:491–502. https://doi.org/10.1038/nrgastro.2017.75
Shoaib M, Shehzad A, Omar M, Rakha A, Raza H, Sharif HR et al (2016) Inulin: properties, health benefits and food applications. Carbohydr Polym 147:444–454. https://doi.org/10.1016/j.carbpol.2016.04.020
Roberfroid MB (2007) Inulin-type fructans: functional food ingredients. J Nutr 137:2493s-s2502. https://doi.org/10.1093/jn/137.11.2493S
Bonnema AL, Kolberg LW, Thomas W, Slavin JL (2010) Gastrointestinal tolerance of chicory inulin products. J Am Diet Assoc 110:865–868. https://doi.org/10.1016/j.jada.2010.03.025
Man S, Liu T, Yao Y, Lu Y, Ma L, Lu F (2021) Friend or foe? The roles of inulin-type fructans. Carbohydr Polym 252:117155. https://doi.org/10.1016/j.carbpol.2020.117155
Chung WS, Walker AW, Louis P, Parkhill J, Vermeiren J, Bosscher D et al (2016) Modulation of the human gut microbiota by dietary fibres occurs at the species level. BMC Biol 14:3. https://doi.org/10.1186/s12915-015-0224-3
Li L, Xiong Q, Zhao J, Lin X, He S, Wu N et al (2020) Inulin-type fructan intervention restricts the increase in gut microbiome-generated indole in patients with peritoneal dialysis: a randomized crossover study. Am J Clin Nutr 111:1087–1099. https://doi.org/10.1093/ajcn/nqz337
Ikizler TA, Burrowes JD, Byham-Gray LD, Campbell KL, Carrero JJ, Chan W et al (2020) KDOQI clinical practice guideline for nutrition in CKD: 2020 update. Am J Kidney Dis 76:S1-s107. https://doi.org/10.1053/j.ajkd.2020.05.006
Yang Y (2019) China food composition tables: standard edition, 6th edn. Peking University Medical Press, Beijing
Penne EL, van der Weerd NC, Grooteman MP, Mazairac AH, van den Dorpel MA, Nubé MJ et al (2011) Role of residual renal function in phosphate control and anemia management in chronic hemodialysis patients. Clin J Am Soc Nephrol 6:281–289. https://doi.org/10.2215/cjn.04480510
Kliger AS, Foley RN, Goldfarb DS, Goldstein SL, Johansen K, Singh A et al (2013) KDOQI US commentary on the 2012 KDIGO clinical practice guideline for anemia in CKD. Am J Kidney Dis 62:849–859. https://doi.org/10.1053/j.ajkd.2013.06.008
Li L, Liang W, Ye T, Chen Z, Zuo X, Du X et al (2016) The association between nutritional markers and biochemical parameters and residual renal function in peritoneal dialysis patients. PLoS One 11:e0156423. https://doi.org/10.1371/journal.pone.0156423
Sampson AL, Singer RF, Walters GD (2017) Uric acid lowering therapies for preventing or delaying the progression of chronic kidney disease. Cochrane Database Syst Rev 10:CD009460. https://doi.org/10.1002/14651858.CD009460.pub2
Maiuolo J, Oppedisano F, Gratteri S, Muscoli C, Mollace V (2016) Regulation of uric acid metabolism and excretion. Int J Cardiol 213:8–14. https://doi.org/10.1016/j.ijcard.2015.08.109
Gettman MT, Ogan K, Brinkley LJ, Adams-Huet B, Pak CY, Pearle MS (2005) Effect of cranberry juice consumption on urinary stone risk factors. J Urol 174:590–594. https://doi.org/10.1097/01.ju.0000165168.68054.f8
Guan H, Zheng Y, Zhou X, Xu Y, Fu C, Xiao J et al (2020) Efficacy of different urinary uric acid indicators in patients with chronic kidney disease. BMC Nephrol 21:290. https://doi.org/10.1186/s12882-020-01953-z
Hatch M, Vaziri ND (1994) Enhanced enteric excretion of urate in rats with chronic renal failure. Clin Sci (Lond) 86:511–516. https://doi.org/10.1042/cs0860511
Lun H, Yang W, Zhao S, Jiang M, Xu M, Liu F et al (2019) Altered gut microbiota and microbial biomarkers associated with chronic kidney disease. Microbiologyopen 8:e00678. https://doi.org/10.1002/mbo3.678
Ramezani A, Massy ZA, Meijers B, Evenepoel P, Vanholder R, Raj DS (2016) Role of the gut microbiome in uremia: a potential therapeutic target. Am J Kidney Dis 67:483–498. https://doi.org/10.1053/j.ajkd.2015.09.027
Sorensen LB (1965) Role of the intestinal tract in the elimination of uric acid. Arthritis Rheum 8:694–706. https://doi.org/10.1002/art.1780080429
Zhu L, Wu Q, Deng C, Zhang M, Zhang C, Chen H et al (2018) Adaptive evolution to a high purine and fat diet of carnivorans revealed by gut microbiomes and host genomes. Environ Microbiol 20:1711–1722. https://doi.org/10.1111/1462-2920.14096
Keenan RT (2020) The biology of urate. Semin Arthritis Rheum 50:S2-s10. https://doi.org/10.1016/j.semarthrit.2020.04.007
Hartwich K, Poehlein A, Daniel R (2012) The purine-utilizing bacterium clostridium acidurici 9a: a genome-guided metabolic reconsideration. PLoS One 7:e51662. https://doi.org/10.1371/journal.pone.0051662
Wu J, Wei Z, Cheng P, Qian C, Xu F, Yang Y et al (2020) Rhein modulates host purine metabolism in intestine through gut microbiota and ameliorates experimental colitis. Theranostics 10:10665–10679. https://doi.org/10.7150/thno.43528
Bian M, Wang J, Wang Y, Nie A, Zhu C, Sun Z et al (2020) Chicory ameliorates hyperuricemia via modulating gut microbiota and alleviating LPS/TLR4 axis in quail. Biomed Pharmacother 131:110719. https://doi.org/10.1016/j.biopha.2020.110719
Wang H, Mei L, Deng Y, Liu Y, Wei X, Liu M et al (2019) Lactobacillus brevis DM9218 ameliorates fructose-induced hyperuricemia through inosine degradation and manipulation of intestinal dysbiosis. Nutrition 62:63–73. https://doi.org/10.1016/j.nut.2018.11.018
Garcia-Arroyo FE, Gonzaga G, Munoz-Jimenez I, Blas-Marron MG, Silverio O, Tapia E et al (2018) Probiotic supplements prevented oxonic acid-induced hyperuricemia and renal damage. PLoS One 13:e0202901 https://doi.org/10.1371/journal.pone.0202901
Premachandra KH, Day RO, Roberts DM (2021) Managing hyperuricemia and gout in chronic kidney disease: a clinical conundrum. Curr Opin Nephrol Hypertens 30:245–251. https://doi.org/10.1097/mnh.0000000000000691
Xi Y, Yan J, Li M, Ying S, Shi Z (2019) Gut microbiota dysbiosis increases the risk of visceral gout in goslings through translocation of gut-derived lipopolysaccharide. Poult Sci 98:5361–5373. https://doi.org/10.3382/ps/pez357
Lee SC, Tang MS, Lim YA, Choy SH, Kurtz ZD, Cox LM et al (2014) Helminth colonization is associated with increased diversity of the gut microbiota. PLoS Negl Trop Dis 8:e2880. https://doi.org/10.1371/journal.pntd.0002880
Louis P, Hold GL, Flint HJ (2014) The gut microbiota, bacterial metabolites and colorectal cancer. Nat Rev Microbiol 12:661–672. https://doi.org/10.1038/nrmicro3344
Van Rymenant E, Abrankó L, Tumova S, Grootaert C, Van Camp J, Williamson G et al (2017) Chronic exposure to short-chain fatty acids modulates transport and metabolism of microbiome-derived phenolics in human intestinal cells. J Nutr Biochem 39:156–168. https://doi.org/10.1016/j.jnutbio.2016.09.009
Hamer HM, Jonkers DM, Bast A, Vanhoutvin SA, Fischer MA, Kodde A et al (2009) Butyrate modulates oxidative stress in the colonic mucosa of healthy humans. Clin Nutr 28:88–93. https://doi.org/10.1016/j.clnu.2008.11.002
Koh A, De Vadder F, Kovatcheva-Datchary P, Bäckhed F (2016) From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell 165:1332–1345. https://doi.org/10.1016/j.cell.2016.05.041
Acknowledgements
We thank all the participants in this clinical trial, and thank for the support from faculties and staffs in Department of Clinical Nutrition and Division of Nephrology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China.
Funding
This work was supported by National Natural Science Foundation of China [grant numbers: 81673161].
Author information
Authors and Affiliations
Contributions
All authors read the manuscript, interpreted the results, provided critical intellectual content, and approved the final manuscript. YC and ZX designed the project conception and development of the overall research plan. He S completed laboratory detection, analyzed the data and wrote the first draft of the manuscript. LW contributed to volunteer recruitment. XQ and TC revised the manuscript. LL contributed to statistics analysis. Other listed authors contributed to collecting samples and laboratory detection.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Ethical approval for this study was obtained from the Institutional Review Board of Tongji Hospital, Huazhong University of Science and Technology (TJ-IRB20171110), and registered at the Chinese Clinical Trials Registry (http://www.chictr.org.cn/. Registration ID: ChiCTR-INR-17013739). Informed consent was obtained before the study commenced in accordance with the Declaration of Helsinki.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
He, S., Xiong, Q., Tian, C. et al. Inulin-type prebiotics reduce serum uric acid levels via gut microbiota modulation: a randomized, controlled crossover trial in peritoneal dialysis patients. Eur J Nutr 61, 665–677 (2022). https://doi.org/10.1007/s00394-021-02669-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-021-02669-y