Abstract
Purpose
Atherosclerosis and its related clinical complications are the leading cause of death. MicroRNA (miR)-92a in the inflammatory endothelial dysfunction leads to atherosclerosis. Krüppel-like factor 2 (KLF2) is required for vascular integrity and endothelial function maintenance. Flavonoids possess many biological properties. This study investigated the vascular protective effects of chrysin in balloon-injured carotid arteries.
Materials and methods
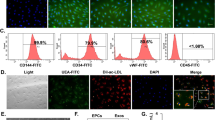
Exosomes were extracted from human coronary artery endothelial cell (HCAEC) culture media. Herb flavonoids and chrysin were the treatments in these atheroprotective models. Western blotting and real-time PCRs were performed. In situ hybridization, immunohistochemistry, and immunofluorescence analyses were employed.
Results
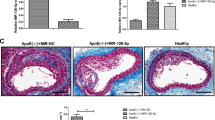
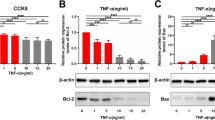
MiR-92a increased after balloon injury and was present in HCAEC culture media. Chrysin was treated, and significantly attenuated the miR-92a levels after balloon injury, and similar results were obtained in HCAEC cultures in vitro. Balloon injury-induced miR-92a expression, and attenuated KLF2 expression. Chrysin increased the KLF2 but reduced exosomal miR-92a secretion. The addition of chrysin and antagomir-92a, neointimal formation was reduced by 44.8 and 49.0% compared with balloon injury after 14 days, respectively.
Conclusion
Chrysin upregulated KLF2 expression in atheroprotection and attenuated endothelial cell-derived miR-92a-containing exosomes. The suppressive effect of miR-92a suggests that chrysin plays an atheroprotective role.
Graphic abstract
Proposed pathway for human coronary artery endothelial cell (HCAEC)-derived exosomes induced by chrysin to suppress microRNA (miR)-92a expression and counteract the inhibitory effect of miR-92a on KLF2 expression in HCAECs. This provides an outline of the critical role of the herbal flavonoid chrysin, which may serve as a valuable therapeutic supplement for atheroprotection.







Similar content being viewed by others
Abbreviations
- MiR-92a:
-
MicroRNA-92a
- KLF2:
-
Krüppel-like factor 2
- PCR:
-
Polymerase chain reaction
- HCAEC:
-
Human coronary artery endothelial cell
- EGCG:
-
Epigallocatechin gallate
- FA:
-
Ferulic acid
- DMSO:
-
Dimethyl sulfoxide
- SD:
-
Standard deviation
- ANOVA:
-
Analysis of variance
References
Gimbrone MA Jr, García-Cardeña G (2016) Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ Res 18(4):620–636
Jain MK, Sangwung P, Hamik A (2014) Regulation of an inflammatory disease: Krüppel-like factors and atherosclerosis. Arterioscler Thromb Vasc Biol 34(3):499–508
Navickas R, Gal D, Laucevičius A, Taparauskaitė A, Zdanytė M, Holvoet P (2016) Identifying circulating microRNAs as biomarkers of cardiovascular disease: a systematic review. Cardiovasc Res 111(4):322–337
Loyer X, Potteaux S, Vion AC et al (2014) Inhibition of microRNA-92a prevents endothelial dysfunction and atherosclerosis in mice. Circ Res 114(3):434–443
Feinberg MW, Moore KJ (2016) MicroRNA regulation of atherosclerosis. Circ Res 118(4):703–720
Fang Y, Davies PF (2012) Site-specific microRNA-92a regulation of Kruppel-like Factors 4 and 2 in atherosusceptible endothelium. Arterioscler Thromb Vasc Biol 32(4):979–987
Jansen F, Li Q, Pfeifer A, Werner N (2017) Endothelial- and immune cell-derived extracellular vesicles in the regulation of cardiovascular health and disease. JACC Basic Transl Sci 2(6):790–807
Lu X (2017) The role of exosomes and exosome-derived microRNAs in atherosclerosis. Curr Pharm Des 23(40):6182–6193
Chang YJ, Li YS, Wu CC et al (2019) Extracellular microRNA-92a mediates endothelial cell-macrophage communication. Arterioscler Thromb Vasc Biol 39(12):2492–2504
Shyu KG, Wang BW, Pan CM, Fang WJ, Lin CM (2019) Hyperbaric oxygen boosts long noncoding RNA MALAT1 exosome secretion to suppress microRNA-92a expression in therapeutic angiogenesis. Int J Cardiol 274:271–278
Liu Y, Li Q, Hosen MR et al (2019) Atherosclerotic conditions promote the packaging of functional microRNA-92a-3p into endothelial microvesicles. Circ Res 124(4):575–587
Kumar S, Pandey AK (2013) Chemistry and biological activities of flavonoids: an overview. Sci World J. https://doi.org/10.1155/2013/162750
Grassi D, Desideri G, Ferri C (2010) Flavonoids: antioxidants against atherosclerosis. Nutrients 2(8):889–902
Enaa F, Badole S, Menaa B, Menaa A, Bodhankar S (2012) Polyphenols, promising therapeutics for inflammatory diseases? In: Watson R, Preedy V (eds) Bioactive food as dietary interventions for arthritis and related inflammatory diseases, 1st edn. Academic Press, New York, pp 421–430
Farkhondeh T, Samarghandian S, Bafandeh F (2019) The cardiovascular protective effects of chrysin: a narrative review on experimental researches. Cardiovasc Hematol Agents Med Chem 17(1):17–27
Lin CM, Shyu KG, Wang BW, Chang H, Chen YH, Chiu JH (2010) Chrysin suppresses IL-6-induced angiogenesis via down-regulation of JAK1/STAT3 and VEGF: an in vitro and in ovo approach. J Agric Food Chem 58(11):7082–7087
Zhao S, Liang M, Wang Y et al (2019) Chrysin suppresses vascular endothelial inflammation via inhibiting the NF-κB signaling pathway. J Cardiovasc Pharmacol Ther 24(3):278–287
Lin CM, Wang BW, Pan CM et al (2018) Effects of flavonoids on microRNA 145 regulation through Klf4 and myocardin in neointimal formation in vitro and in vivo. J Nutr Biochem 52:27–35
Wang W, Li Z, Zheng Y, Yan M, Cui Y, Jiang J (2019) Circulating microRNA-92a level predicts acute coronary syndrome in diabetic patients with coronary heart disease. Lipids Health Dis 18(1):22. https://doi.org/10.1186/s12944-019-0964-0
Wang Z, Zhang J, Zhang S et al (2019) MiR-30e and miR-92a Are related to atherosclerosis by targeting ABCA1. Mol Med Rep 19(4):3298–3304
Vazhappilly CG, Ansari SA, Al-Jaleeli R et al (2019) Role of flavonoids in thrombotic, cardiovascular, and inflammatory diseases. Inflammopharmacology 27(5):863–869
Farkhondeh T, Samarghandian S, Bafandeh F (2019) Impact of chrysin on the molecular mechanisms underlying diabetic complications. J Cell Physiol 234(10):17144–17158
Andrade N, Andrade S, Silva C et al (2020) Chronic consumption of the dietary polyphenol chrysin attenuates metabolic disease in fructose-fed rats. Eur J Nutr 59(1):151–165
Zhong X, Zhang L, Li Y, Li P, Li J, Cheng G (2018) Kaempferol alleviates ox-LDL-induced apoptosis by up-regulation of miR-26a-5p via inhibiting TLR4/NF-κB pathway in human endothelial cells. Biomed Pharmacother 108:1783–1789
Zhang H, Zhao Z, Pang X et al (2017) MiR-34a/sirtuin-1/foxo3a is involved in genistein protecting against ox-LDL-induced oxidative damage in HUVECs. Toxicol Lett 277:115–122
Xu X, Tian L, Zhang Z (2019) Triptolide inhibits angiogenesis in microvascular endothelial cells through regulation of miR-92a. J Physiol Biochem 75(4):573–583
Chamorro-Jorganes A, Lee MY, Araldi E et al (2016) VEGF-induced expression of miR-17–92 cluster in endothelial cells is mediated by ERK/ELK1 activation and regulates angiogenesis. Circ Res 118(1):38–47
Sweets DR, Fan L, Hsieh PN, Jain MK (2018) Krüppel-like factors in vascular inflammation: mechanistic insights and therapeutic potential. Front Cardiovasc Med 5:6. https://doi.org/10.3389/fcvm.2018.00006.5,1
Martínez-Fernández L, Pons Z, Margalef M, Arola-Arnal A, Muguerza B (2015) Regulation of vascular endothelial genes by dietary flavonoids: structure-expression relationship studies and the role of the transcription factor KLF-2. J Nutr Biochem 26(3):277–284
Caton PW, Pothecary MR, Lees DM et al (2010) Regulation of vascular endothelial function by procyanidin-rich foods and beverages. J Agric Food Chem 58(7):4008–4013
Han JM, Li H, Cho MH et al (2017) Soy-leaf extract exerts atheroprotective effects via modulation of Krüppel-like factor 2 and adhesion molecules. Int J Mol Sci 18(2):373. https://doi.org/10.3390/ijms18020373
AnandhiR TPA, Geraldine P (2014) Evaluation of the anti-atherogenic potential of chrysin in Wistar rats. Mol Cell Biochem 385(1–2):103–113
Boulanger CM, Loyer X, Rautou PE, Amabile N (2017) Extracellular vesicles in coronary artery disease. Nat Rev Cardiol 14(5):259–272
Wang Y, Xie Y, Zhang A, Wang M, Fang Z, Zhang J (2019) Exosomes: an emerging factor in atherosclerosis. Biomed Pharmacother 115:108951. https://doi.org/10.1016/j.biopha.2019.108951
Yin M, Loyer X, Boulanger CM (2015) Extracellular vesicles as new pharmacological targets to treat atherosclerosis. Eur J Pharmacol 763(Pt A):90–103
Soleti R, Andriantsitohaina R, Martinez MC (2018) Impact of polyphenols on extracellular vesicle levels and effects and their properties as tools for drug delivery for nutrition and health. Arch Biochem Biophys 644:57–63
Camacho P, Fan HM, Liu ZM, He JQ (2016) Small mammalian animal models of heart disease. Am J Cardiovasc Dis 6(3):70–80
Acknowledgements
This work was partially supported, in part, by Shin Kong Wu Ho-Su Memorial Hospital (Taipei, Taiwan; SKH-8302-105-DR-23). CML, BWW, CMP, WJF, and KGS conceived and designed the experiments. CML, SKC, and KGS analyzed the data. CML wrote the paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Lin, CM., Wang, BW., Pan, CM. et al. Chrysin boosts KLF2 expression through suppression of endothelial cell-derived exosomal microRNA-92a in the model of atheroprotection . Eur J Nutr 60, 4345–4355 (2021). https://doi.org/10.1007/s00394-021-02593-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-021-02593-1