Abstract
Purpose
Inflammatory bowel diseases are associated with an increase in the whole-body protein turnover, thus possibly requiring an additional supply of dietary proteins. Our aim was to evaluate whether increasing dietary protein content could alleviate protein metabolism alterations in the injured splanchnic and peripheral tissues during colitis and spontaneous mucosal healing.
Methods
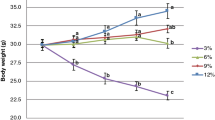
Mice with acute chemically induced colitis received either a normal protein (P14, 14% as energy), a moderately (P30, 30%) and a very high-protein (P53, 55%) diets. At different times after the challenge, protein synthesis rate was determined in tissues using a flooding dose of 13C valine.
Results
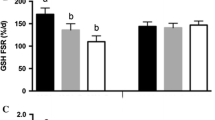
Colon, liver and spleen protein synthesis rates were significantly increased after colitis induction, while being decreased in the caecum, kidneys and muscle. Contrastingly to the two other diets, P30 diet consumption allowed faster recovery of the animals, and this coincided with a rapid resaturation of the initial protein synthesis in the colon. In the other tissues studied, the high-protein diets show different effects depending on the dietary protein content consumed and on the examined tissues, with a general trend of P53 in lowering anabolism rates.
Conclusion
This study highlights the severe impact of acute colonic inflammation on protein metabolism in different organs. In addition, dietary protein content modulated the recovery of the initial protein synthesis rate in the various tissues following colitis induction. P30 diet consumption notably showed a better ability to alleviate protein metabolism perturbations induced by colitis, that may explain its documented beneficial effect on colon mucosal healing.




Similar content being viewed by others
References
Hartman C, Eliakim R, Shamir R (2009) Nutritional status and nutritional therapy in inflammatory bowel diseases. World J Gastroenterol 15:2570–2578. https://doi.org/10.3748/wjg.15.2570
Scaldaferri F, Pizzoferrato M, Lopetuso LR et al (2017) Nutrition and IBD: malnutrition and/or sarcopenia? A practical guide. Gastroenterol Res Pract. https://doi.org/10.1155/2017/8646495
Yarur AJ, Quintero MA, Jain A et al (2017) Serum amyloid a as a surrogate marker for mucosal and histologic inflammation in patients with Crohn’s disease. Inflamm Bowel Dis 23:158–164. https://doi.org/10.1097/MIB.0000000000000991
El Yousfi M, Breuillé D, Papet I et al (2003) Increased tissue protein synthesis during spontaneous inflammatory bowel disease in HLA-B27 rats. Clin Sci (Lond) 105:437–446. https://doi.org/10.1042/CS20020313
Han PD, Burke A, Baldassano RN et al (1999) Nutrition and inflammatory bowel disease. Gastroenterol Clin North Am 28(423–43):ix. https://doi.org/10.1016/s0889-8553(05)70063-7
Vavricka SR, Schoepfer A, Scharl M et al (2015) Extraintestinal manifestations of inflammatory bowel disease. Inflamm Bowel Dis 21:1982–1992. https://doi.org/10.1097/MIB.0000000000000392
Corica D, Romano C (2015) Renal involvement in inflammatory bowel diseases. J Crohn’s Colitis 10:1–10. https://doi.org/10.1093/ecco-jcc/jjv138
Soeters PB, van de Poll MCG, van Gemert WG, Dejong CHC (1582S) Amino acid adequacy in pathophysiological states. J Nutr 134:1575S–1582S. https://doi.org/10.1093/jn/134.6.1575S
Mercier S, Breuillé D, Mosoni L et al (2002) Chronic inflammation alters protein metabolism in several organs of adult rats. J Nutr 132:1921–1928. https://doi.org/10.1093/jn/132.7.1921
Heys SD, Park KG, McNurlan MA et al (1992) Protein synthesis rates in colon and liver: stimulation by gastrointestinal pathologies. Gut 33:976–981. https://doi.org/10.1136/gut.33.7.976
Bischoff SC, Escher J, Hébuterne X et al (2020) ESPEN practical guideline: Clinical Nutrition in inflammatory bowel disease. Clin Nutr 39:632–653. https://doi.org/10.1016/j.clnu.2019.11.002
Lan A, Blachier F, Benamouzig R et al (2015) Mucosal healing in inflammatory bowel diseases: is there a place for nutritional supplementation? Inflamm Bowel Dis 21:198–207. https://doi.org/10.1097/MIB.0000000000000177
Lan A, Blais A, Coelho D et al (2016) Dual effects of a high-protein diet on DSS-treated mice during colitis resolution phase. Am J Physiol Gastrointest Liver Physiol 311:G624–G633. https://doi.org/10.1152/ajpgi.00433.2015
Vidal-Lletjós S, Andriamihaja M, Blais A et al (2019) Dietary protein intake level modulates mucosal healing and mucosa-adherent microbiota in mouse model of colitis. Nutrients 11:1–16. https://doi.org/10.3390/nu11030514
Chevalier L, Bos C, Gryson C et al (2009) High-protein diets differentially modulate protein content and protein synthesis in visceral and peripheral tissues in rats. Nutrition 25:932–939. https://doi.org/10.1016/j.nut.2009.01.013
Chevalier L, Bos C, Azzout-Marniche D et al (2010) Dietary protein regulates hepatic constitutive protein anabolism in rats in a dose-dependent manner and independently of energy nutrient composition. AJP Regul Integr Comp Physiol 299:R1720–R1730. https://doi.org/10.1152/ajpregu.00497.2010
Oberli M, Lan A, Khodorova N et al (2016) Compared with raw bovine meat, boiling but not grilling, barbecuing, or roasting decreases protein digestibility without any major consequences for intestinal mucosa in rats, although the daily ingestion of bovine meat induces histologic modifications in. J Nutr 146:1506–1513. https://doi.org/10.3945/jn.116.230839
Breuille C, Rose F, Arnal M et al (1994) Sepsis modifies the contribution of different organs to whole-body protein synthesis in rats. Clin Sci 86:663–669. https://doi.org/10.1042/cs0860663
Vidal-Lletjós S, Andriamihaja M, Blais A et al (2019) Mucosal healing progression after acute colitis in mice. World J Gastroenterol 25:3572–3589. https://doi.org/10.3748/wjg.v25.i27.3572
Essén P, McNurlan MA, Wernerman J et al (1992) Short-term starvation decreases skeletal muscle protein synthesis rate in man. Clin Physiol 12:287–299. https://doi.org/10.1111/j.1475-097X.1992.tb00834.x
Merritt EK, Cross JM, Bamman MM (2012) Inflammatory and protein metabolism signaling responses in human skeletal muscle after burn injury. J Burn Care Res 33:291–297. https://doi.org/10.1097/BCR.0b013e3182331e4b
Massafra V, Ijssennagger N, Plantinga M et al (2016) Splenic dendritic cell involvement in FXR-mediated amelioration of DSS colitis. Biochim Biophys Acta Mol Basis Dis 1862:166–173. https://doi.org/10.1016/j.bbadis.2015.11.001
Liu X, Beaumont M, Walker F et al (2013) Beneficial effects of an amino acid mixture on colonic mucosal healing in rats. Inflamm Bowel Dis 19:2895–2905. https://doi.org/10.1097/01.MIB.0000435849.17263.c5
Blachier F, Beaumont M, Andriamihaja M et al (2017) Changes in the luminal environment of the colonic epithelial cells and physiopathological consequences. Am J Pathol 187:476–486. https://doi.org/10.1016/j.ajpath.2016.11.015
Acknowledgements
The authors greatly acknowledge Nicolas Sorhaindo from the Centre de Recherche sur l’Inflammation UMR 1149, Morgane Dufay who helped to take care of the animals and Sarah Mekiri, Koralyn Octavien-Paul and Luc Gérard, for their involvement in the experiments. Also, they want to thank Armor Protéines (Saint-Brice-en-Cogles, France) for providing the dietary proteins used to prepare diets.
Funding
This work was supported by the INRAE, AgroParisTech, the Société Française de Nutrition and the Association François Aupetit.
Author information
Authors and Affiliations
Contributions
S.V.L., M.P. and A.L. performed the experiments. S.V.L., M.P., N.K. and A.L., analysed the data. A.L., C.G. and F.B. conceived and supervised the study. S.V.L. and A.L. drafted the manuscript. All the authors have read and approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflict of interest.
Ethics approval
This study was conducted in accordance to the European directive for the use and care of laboratory animals (2010/63/UE), and received the agreement of the local animal ethics committee and of the ministerial committee for animal experimentation (registration number: APAFIS#3987–2016012214388658).
Rights and permissions
About this article
Cite this article
Vidal-Lletjós, S., Khodorova, N.V., Piscuc, M. et al. Tissue-specific effect of colitis on protein synthesis in mice: impact of the dietary protein content. Eur J Nutr 60, 1669–1677 (2021). https://doi.org/10.1007/s00394-020-02365-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-020-02365-3