Abstract
Purpose
The aim of this study was to evaluate the prevalence of thyroid dysfunction in a cohort of healthy individuals in Mexico City, as well as to investigate the potential associations of these results with their estimated iodine intake (EII) as reflected by their 24-hour urinary iodine excretion (24-h UIE).
Methods
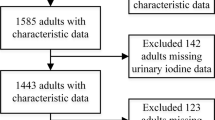
From the SALMEX cohort, 683 adults provided an appropriate 24-h urine sample. Thyroid function tests and thyroid antibody concentrations were determined in the participants’ sera. We analyzed discrepancies between the commonly used urinary parameters to determine the iodine intake status and the performance of thyroglobulin (Tg) as a biomarker of its status in the adult population.
Results
The prevalence of dysthyroidism was high, being similar to other studies. Subclinical hypothyroidism was detected in 5.0% of individuals, clinical hypothyroidism in 1.8% of individuals, and sub-clinical hyperthyroidism in 2.8% of individuals. The median EII was 285 μg/d (IQR 215.0–369.0); 94% of individuals had EII >150 µg/d recommended daily allowance (RDA) in adults. The urinary iodine concentration (UIC) and the UIE had relative biases in their averages of 34.4%. The Tg median was 7.21 ng/mL. The prevalence of increased Tg was 6.15%. There was no correlation between Tg and EII (r= 0.019, p= 0.606).
Conclusions
Thyroid dysfunction was highly prevalent in this population. Our cohort revealed a slight discrepancy between dysthyroidism manifestations and iodine intake markers; the latter represent a population with adequate iodine intake. Further studies are necessary to clearly define the prevalence of thyroid dysfunction as well as the iodine nutritional status in Mexico.



Similar content being viewed by others
References
Hoogendoorn EH, Hermus AR, de Vegt F, Ross HA, Verbeek AL, Kiemeney LA, Swinkels DW, Sweep FC, den Heijer M (2006) Thyroid function and prevalence of anti-thyroperoxidase antibodies in a population with borderline sufficient iodine intake: influences of age and sex. Clin Chem 52(1):104–11. https://doi.org/10.1373/clinchem.2005.055194
Vanderpump MP (2011) The epidemiology of thyroid disease. Br Med Bull 99:39–51. https://doi.org/10.1093/bmb/ldr030
Burek CL, Talor MV (2009) Environmental triggers of autoimmune thyroiditis. J Autoimmun 33(3–4):183–9. https://doi.org/10.1016/j.jaut.2009.09.001
Zimmermann MB, Andersson M (2012) Assessment of iodine nutrition in populations: past, present, and future. Nutr Rev 70(10):553–70. https://doi.org/10.1111/j.1753-4887.2012.00528.x
Teng W, Shan Z, Teng X, Guan H, Li Y, Teng D, Jin Y, Yu X, Fan C, Chong W, Yang F, Dai H, Yu Y, Li J, Chen Y, Zhao D, Shi X, Hu F, Mao J, Gu X, Yang R, Tong Y, Wang W, Gao T, Li C (2006) Effect of iodine intake on thyroid diseases in China. N Engl J Med 354(26):2783–93. https://doi.org/10.1056/NEJMoa054022
Teng X, Shan Z, Chen Y, Lai Y, Yu J, Shan L, Bai X, Li Y, Li N, Li Z, Wang S, Xing Q, Xue H, Zhu L, Hou X, Fan C, Teng W (2011) More than adequate iodine intake may increase subclinical hypothyroidism and autoimmune thyroiditis: a cross-sectional study based on two Chinese communities with different iodine intake levels. Eur J Endocrinol 164(6):943–50. https://doi.org/10.1530/EJE-10-1041
Camargo RY, Tomimori EK, Neves SC, G S Rubio I, Galrão AL, Knobel M, Medeiros-Neto G (2008) Thyroid and the environment: exposure to excessive nutritional iodine increases the prevalence of thyroid disorders in Sao Paulo. Brazil. Eur J Endocrinol 159(3):293–9. https://doi.org/10.1530/EJE-08-0192
Vejbjerg P, Knudsen N, Perrild H, Laurberg P, Andersen S, Rasmussen LB, Ovesen L, Jørgensen T (2009) Estimation of iodine intake from various urinary iodine measurements in population studies. Thyroid 19(11):1281–6. https://doi.org/10.1089/thy.2009.0094
The Iodine Global Network (2019) Global scorecard of iodine nutrition in 2019 in the general population based on school-age children (SAC) https://www.ign.org/cm_data/Global_Scorecard_MAP_2019_SAC.pdf. Iodine Global Network Webpage. Accessed 24 Oct 2019
Health Minister of Mexico (2003) Modificación a la Norma Oficial Mexicana NOM-040-SSA1-1993, Productos y servicios. Sal yodada y sal yodada fluorada. Especificaciones sanitarias. https://www.salud.gob.mx/unidades/cdi/nom/m040ssa193.html. Accesed 24 Oct 2019
Baloch Z, Carayon P, Conte-Devolx B, Demers LM, Feldt-Rasmussen U, Henry JF, LiVosli VA, Niccoli-Sire P, John R, Ruf J, Smyth PP, Spencer CA, Stockigt JR (2003) Laboratory medicine practice guidelines. Laboratory support for the diagnosis and monitoring of thyroid disease. Thyroid 13(1):3–126. https://doi.org/10.1089/105072503321086962
Colin-Ramirez E, Espinosa-Cuevas A, Miranda-Alatriste PV, Tovar-Villegas VI, Arcand J, Correa-Rotter R (2017) Food Sources of Sodium Intake in an Adult Mexican Population: A Sub-Analysis of the SALMEX Study. Nutrients. https://doi.org/10.3390/nu9080810
Vega-Vega O, Fonseca-Correa JI, Mendoza-De la Garza A, Rincón-Pedrero R, Espinosa-Cuevas A, Baeza-Arias Y, Dary O, Herrero-Bervera B, Nieves-Anaya I, Correa-Rotter R (2018) Contemporary Dietary Intake: Too Much Sodium, Not Enough Potassium, yet Sufficient Iodine: The SALMEX Cohort Results. Nutrients. https://doi.org/10.3390/nu10070816
Kesteloot H, Joossens JV (1996) On the determinants of the creatinine clearance: a population study. J Hum Hypertens 10(4):245–9
European Food Safety Autority (2006) Tolerable upper intake levels for vitamins and minerals. https://www.efsa.europa.eu/sites/default/files/efsa_rep/blobserver_assets/ndatolerableuil.pdf. Accesed 27 June 2019
Institute of Medicine (2001) Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. The National Academies Press, Washington https://doi.org/10.17226/10026
Canaris GJ, Manowitz NR, Mayor G, Ridgway EC (2000) The Colorado thyroid disease prevalence study. Arch Intern Med 160(4):526–34. https://doi.org/10.1001/archinte.160.4.526
O'Leary PC, Feddema PH, Michelangeli VP, Leedman PJ, Chew GT, Knuiman M, Kaye J, Walsh JP (2006) Investigations of thyroid hormones and antibodies based on a community health survey: the Busselton thyroid study. Clin Endocrinol (Oxf) 64(1):97–104. https://doi.org/10.1111/j.1365-2265.2005.02424.x
Hollowell JG, Staehling NW, Flanders WD, Hannon WH, Gunter EW, Spencer CA, Braverman LE (2002) Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab 87(2):489–99. https://doi.org/10.1210/jcem.87.2.8182
Bjoro T, Holmen J, Krüger O, Midthjell K, Hunstad K, Schreiner T, Sandnes L, Brochmann H (2000) Prevalence of thyroid disease, thyroid dysfunction and thyroid peroxidase antibodies in a large, unselected population. The Health Study of Nord-Trondelag (HUNT). Eur J Endocrinol 143(5):639–47. https://doi.org/10.1530/eje.0.1430639
Garduño-Garcia Jde J, Alvirde-Garcia U, López-Carrasco G, Padilla Mendoza ME, Mehta R, Arellano-Campos O, Choza R, Sauque L, Garay-Sevilla ME, Malacara JM, Gomez-Perez FJ, Aguilar-Salinas CA (2010) TSH and free thyroxine concentrations are associated with differing metabolic markers in euthyroid subjects. Eur J Endocrinol 163(2):273–8. https://doi.org/10.1530/EJE-10-0312
Guan H, Shan Z, Teng X, Li Y, Teng D, Jin Y, Yu X, Fan C, Chong W, Yang F, Dai H, Yu Y, Li J, Chen Y, Zhao D, Shi X, Hu F, Mao J, Gu X, Yang R, Chen W, Tong Y, Wang W, Gao T, Li C, Teng W (2008) Influence of iodine on the reference interval of TSH and the optimal interval of TSH: results of a follow-up study in areas with different iodine intakes. Clin Endocrinol (Oxf) 69(1):136–41. https://doi.org/10.1111/j.1365-2265.2007.03150.x
Caldwell KL, Jones R, Hollowell JG (2005) Urinary iodine concentration: United States National Health And Nutrition Examination Survey 2001–2002. Thyroid 15(7):692–9. https://doi.org/10.1089/thy.2005.15.692
Völzke H, Alte D, Kohlmann T, Lüdemann J, Nauck M, John U, Meng W (2005) Reference intervals of serum thyroid function tests in a previously iodine-deficient area. Thyroid 3:279–85. https://doi.org/10.1089/thy.2005.15.279
Institute of Medicine (2000) Dietary Reference Intakes: Applications in Dietary Assessment. Washington, DC: The National Academies Press. https://doi.org/10.17226/9956.
Johner SA, Shi L, Remer T (2010) Higher urine volume results in additional renal iodine loss. Thyroid 20(12):1391–7. https://doi.org/10.1089/thy.2010.0161
Johner SA, Thamm M, Schmitz R, Remer T (2016) Examination of iodine status in the German population: an example for methodological pitfalls of the current approach of iodine status assessment. Eur J Nutr 55(3):1275–82. https://doi.org/10.1007/s00394-015-0941-y
Andersen S, Waagepetersen R, Laurberg P (2015) Misclassification of iodine intake level from morning spot urine samples with high iodine excretion among Inuit and non-Inuit in Greenland. Br J Nutr 113(9):1433–40. https://doi.org/10.1017/S0007114515000653
Andersen S, Dehnfeld M, Laurberg P (2015) Ethnicity is import9ant for creatinine excretion among Inuit and Caucasians in Greenland. Scand J Clin Lab Invest 75(1):44–50. https://doi.org/10.3109/00365513.2014.965737
Zimmermann MB, Aeberli I, Andersson M, Assey V, Yorg JA, Jooste P, Jukić T, Kartono D, Kusić Z, Pretell E, San Luis TO, Jr Untoro J, Timmer A (2013) Thyroglobulin is a sensitive measure of both deficient and excess iodine intakes in children and indicates no adverse effects on thyroid function in the UIC range of 100–299 mug/L: a UNICEF/ICCIDD study group report. J Clin Endocrinol Metab 98(3):1271–80. https://doi.org/10.1210/jc.2012-3952
Pearce EN, Caldwell KL (2016) Urinary iodine, thyroid function, and thyroglobulin as biomarkers of iodine status. Am J Clin Nutr 104(Suppl 3):898S–901S. https://doi.org/10.3945/ajcn.115.110395
Vejbjerg P, Knudsen N, Perrild H, Laurberg P, Carlé A, Pedersen IB, Rasmussen LB, Ovesen L, Jørgensen T (2009) Thyroglobulin as a marker of iodine nutrition status in the general population. Eur J Endocrinol 161(3):475–81. https://doi.org/10.1530/EJE-09-0262
Spencer C, LoPresti J, Fatemi S (2014) How sensitive (second-generation) thyroglobulin measurement is changing paradigms for monitoring patients with differentiated thyroid cancer, in the absence or presence of thyroglobulin autoantibodies. Curr Opin Endocrinol Diabetes Obes 21(5):394–404. https://doi.org/10.1097/MED.0000000000000092
Ma ZF, Venn BJ, Manning PJ, Cameron CM, Skeaff SA (2018) The sensitivity and specificity of thyroglobulin concentration using repeated measures of urinary iodine excretion. Eur J Nutr 57(4):1313–20. https://doi.org/10.1007/s00394-017-1410-6
Li C, Peng S, Zhang X, Xie X, Wang D, Mao J, Teng X, Shan Z, Teng W (2016) The Urine Iodine to Creatinine as an Optimal Index of Iodine During Pregnancy in an Iodine Adequate Area in China. J Clin Endocrinol Metab 101(3):1290–8. https://doi.org/10.1210/jc.2015-3519
Konig F, Andersson M, Hotz K, Aeberli I, Zimmermann MB (2011) Ten repeat collections for urinary iodine from spot samples or 24-hour samples are needed to reliably estimate individual iodine status in women. J Nutr 141(11):2049–54. https://doi.org/10.3945/jn.111.144071
Acknowledgements
We gratefully acknowledge the support of P. Chem. Bertha Herrero Bervera from Department of Nephrology and Mineral Metabolism. Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán, Mexico City, Mexico.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical standards
his study was approved by the ethics committee and has been conducted in accordance with the ethical standards. All patients provided their informed consent prior to their inclusion in the study.
Rights and permissions
About this article
Cite this article
Flores-Rebollar, A., Pérez-Díaz, I., Vega-Vega, O. et al. Prevalence of thyroid dysfunction in healthy adults according to the estimated iodine intake in 24-hour urine samples: The SALMEX cohort. Eur J Nutr 60, 399–409 (2021). https://doi.org/10.1007/s00394-020-02254-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-020-02254-9