Abstract
Purpose
Insulin-like growth factor-I (IGF-I) is related to growth and its secretion is modified by protein intake in early infancy. We examined the relationship of dietary protein and circulating amino acids on plasma IGF-I levels and early growth.
Methods
Healthy formula-fed infants (n = 213) were randomly assigned to receive either a protein-reduced infant formula with alpha-lactalbumin-enriched whey and free tryptophan and phenylalanine (IF) or an isocaloric standard formula without free amino acids (CF) for the first 120 days of life. A group of breastfed (BF) infants was studied as a non-randomized reference cohort. Biochemical variables were measured shortly after birth (subpopulation) and at an age of 120 days. A path analysis was used to explore the relationship between IGF-I, insulin and amino acids. Results are derived from secondary analyses of a randomized controlled trial.
Results
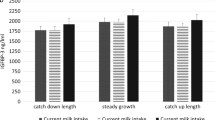
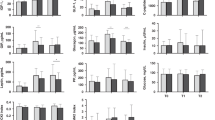
Plasma concentrations of IGF-I at 120 days were significantly higher in IF than in CF infants [58.5 (15.0) vs. 53.7 (9.95) ng/mL; p = 0.020]. BF infants showed lower IGF-I concentrations of 41.6 (10.7) ng/mL. All amino acids but Thr and Cit had a more marked effect on insulin than on IGF-I level. Considering weight, sex and feeding group, Trp explained an equal percentage of variance of IGF-I and insulin (total R 2 12.5 % of IGF-I and 12.3 % of insulin), while branched-chain AA explained an up to twofold higher variance of insulin than IGF-I. Compared to CF, IF explained 18.9 % of the IGF-I level (p = 0.03), while for insulin no direct effect was detectable.
Conclusion
Higher IGF-I concentrations and growth velocities in infants receiving protein-reduced IF indicate that the protein concentration of an infant formula alone does not control IGF-I levels and growth. Other components (e.g., selected amino acids) of infant formulae might control directly or indirectly via insulin influence IGF-I.


Similar content being viewed by others
Abbreviations
- AA:
-
Amino acid
- BF:
-
Breastfed
- CF:
-
Control formula
- FF:
-
Formula-fed
- IF:
-
Intervention formula
- IGF-I:
-
Insulin-like growth factor-I
- LC-PUFA:
-
Long-chain polyunsaturated fatty acids
- GIP:
-
Glucose-dependent insulinotropic peptide
References
Livingstone C (2013) Insulin-like growth factor-I (IGF-I) and clinical nutrition. Clin Sci 125(6):265–280
Thissen JP, Ketelslegers JM, Underwood LE (1994) Nutritional regulation of the insulin-like growth factors. Endocr Rev 15(1):80–101
Melnik BC, John SM, Schmitz G (2013) Milk is not just food but most likely a genetic transfection system activating mTORC1 signaling for postnatal growth. Nutr J 12(103):1475–2891
Rzehak P, Grote V, Lattka E, Weber M, Gruszfeld D, Socha P, Closa-Monasterolo R, Escribano J, Giovannini M, Verduci E, Goyens P, Martin F, Langhendries JP, Demmelmair H, Klopp N, Illig T, Koletzko B (2013) Associations of IGF-1 gene variants and milk protein intake with IGF-I concentrations in infants at age 6 months - results from a randomized clinical trial. Growth Horm IGF Res 23(5):149–158
Fliesen T, Maiter D, Gerard G, Underwood LE, Maes M, Ketelslegers JM (1989) Reduction of serum insulin-like growth factor-I by dietary protein restriction is age dependent. Pediatr Res 26(5):415–419
Koletzko B, von Kries R, Closa R, Escribano J, Scaglioni S, Giovannini M, Beyer J, Demmelmair H, Gruszfeld D, Dobrzanska A, Sengier A, Langhendries JP, Rolland Cachera MF, Grote V, European Childhood Obesity Trial Study G (2009) Lower protein in infant formula is associated with lower weight up to age 2 y: a randomized clinical trial. Am J Clin Nutr 89(6):1836–1845. doi:10.3945/ajcn.2008.27091
Smith PJ, Wise LS, Berkowitz R, Wan C, Rubin CS (1988) Insulin-like growth factor-I is an essential regulator of the differentiation of 3T3-L1 adipocytes. J Biol Chem 263(19):9402–9408
Nougues J, Reyne Y, Barenton B, Chery T, Garandel V, Soriano J (1993) Differentiation of adipocyte precursors in a serum-free medium is influenced by glucocorticoids and endogenously produced insulin-like growth factor-I. Int J Obes Relat Metab Disord 17(3):159–167
Weber M, Grote V, Closa-Monasterolo R, Escribano J, Langhendries JP, Dain E, Giovannini M, Verduci E, Gruszfeld D, Socha P, Koletzko B, European Childhood Obesity Trial Study G (2014) Lower protein content in infant formula reduces BMI and obesity risk at school age: follow-up of a randomized trial. Am J Clin Nutr 99(5):1041–1051. doi:10.3945/ajcn.113.064071
Owen CG, Martin RM, Whincup PH, Smith GD, Cook DG (2006) Does breastfeeding influence risk of type 2 diabetes in later life? A quantitative analysis of published evidence. Am J Clin Nutr 84(5):1043–1054
Newsholme P, Cruzat V, Arfuso F, Keane K (2014) Nutrient regulation of insulin secretion and action. J Endocrinol 221(3):13–0616
Fleddermann M, Demmelmair H, Grote V, Nikolic T, Trisic B, Koletzko B (2013) Infant formula composition affects energetic efficiency for growth: the BeMIM study, a randomized controlled trial. Clin Nutr 30(13):00330
Haschke F, van’t Hof MA (2000) Euro-growth references for length, weight, and body circumferences Euro-growth study group. J Pediatr Gastroenterol Nutr 31(1):S14–S38
Commission E (2006) Commission Directive 2006/141/EC of 22 December 2006 on infant formulae and follow-on formulae and amending Directive 1999/21/EC. Off J Eur Union, 401/401–401/433
Commission E (1999) Commission directive 1999/21/EC on infant formulae and follow-on formulae. Off J Eur Union
Harder U, Koletzko B, Peissner W (2011) Quantification of 22 plasma amino acids combining derivatization and ion-pair LC-MS/MS. J Chromatogr B Analyt Technol Biomed Life Sci 879(7–8):495–504
Bidlingmaier M, Friedrich N, Emeny RT, Spranger J, Wolthers OD, Roswall J, Korner A, Obermayer-Pietsch B, Hubener C, Dahlgren J, Frystyk J, Pfeiffer AF, Doering A, Bielohuby M, Wallaschofski H, Arafat AM (2014) Reference intervals for insulin-like growth factor-1 (igf-i) from birth to senescence: results from a multicenter study using a new automated chemiluminescence IGF-I immunoassay conforming to recent international recommendations. J Clin Endocrinol Metab 99(5):1712–1721
Frystyk J, Grofte T, Skjaerbaek C, Orskov H (1997) The effect of oral glucose on serum free insulin-like growth factor-I and -II in health adults. J Clin Endocrinol Metab 82(9):3124–3127
Sandstrom O, Lonnerdal B, Graverholt G, Hernell O (2008) Effects of alpha-lactalbumin-enriched formula containing different concentrations of glycomacropeptide on infant nutrition. Am J Clin Nutr 87(4):921–928
Runchey SS, Pollak MN, Valsta LM, Coronado GD, Schwarz Y, Breymeyer KL, Wang C, Wang CY, Lampe JW, Neuhouser ML (2012) Glycemic load effect on fasting and post-prandial serum glucose, insulin, IGF-1 and IGFBP-3 in a randomized, controlled feeding study. Eur J Clin Nutr 66(10):1146–1152
Leunissen RW, Kerkhof GF, Stijnen T, Hokken-Koelega A (2009) Timing and tempo of first-year rapid growth in relation to cardiovascular and metabolic risk profile in early adulthood. JAMA 301(21):2234–2242
Martin FP, Moco S, Montoliu I, Collino S, Da Silva L, Rezzi S, Prieto R, Kussmann M, Inostroza J, Steenhout P (2014) Impact of breast-feeding and high- and low-protein formula on the metabolism and growth of infants from overweight and obese mothers. Pediatr Res 75(4):535–543
Madsen AL, Schack-Nielsen L, Larnkjaer A, Molgaard C, Michaelsen KF (2010) Determinants of blood glucose and insulin in healthy 9-month-old term Danish infants; the SKOT cohort. Diabet Med 27(12):1350–1357
Yuksel B, Ozbek MN, Mungan NO, Darendeliler F, Budan B, Bideci A, Cetinkaya E, Berberoglu M, Evliyaoglu O, Yesilkaya E, Arslanoglu I, Darcan S, Bundak R, Ercan O (2011) Serum IGF-1 and IGFBP-3 levels in healthy children between 0 and 6 years of age. J Clin Res Pediatr Endocrinol 3(2):84–88
Larnkjaer A, Molgaard C, Michaelsen KF (2012) Early nutrition impact on the insulin-like growth factor axis and later health consequences. Curr Opin Clin Nutr Metab Care 15(3):285–292
Hawkes CP, Grimberg A (2013) Measuring growth hormone and insulin-like growth factor-I in infants: what is normal? Pediatr Endocrinol Rev 11(2):126–146
Ong KK, Langkamp M, Ranke MB, Whitehead K, Hughes IA, Acerini CL, Dunger DB (2009) Insulin-like growth factor I concentrations in infancy predict differential gains in body length and adiposity: the Cambridge baby growth study. Am J Clin Nutr 90(1):156–161
Socha P, Grote V, Gruszfeld D, Janas R, Demmelmair H, Closa-Monasterolo R, Subias JE, Scaglioni S, Verduci E, Dain E, Langhendries JP, Perrin E, Koletzko B (2011) Milk protein intake, the metabolic-endocrine response, and growth in infancy: data from a randomized clinical trial. Am J Clin Nutr 94(6 Suppl):17
Melnik BC (2012) Excessive Leucine-mTORC1-Signalling of cow milk-based infant formula: the missing link to understand early childhood obesity. J Obes 197653(10):19
Tikanoja T, Simell O (1983) Plasma amino acids after a feed of human milk or formula at three months of age. J Pediatr Gastroenterol Nutr 2(2):252–255
Dawson-Hughes B, Harris SS, Rasmussen HM, Dallal GE (2007) Comparative effects of oral aromatic and branched-chain amino acids on urine calcium excretion in humans. Osteoporos Int 18(7):955–961
Lonnerdal B (2014) Infant formula and infant nutrition: bioactive proteins of human milk and implications for composition of infant formulas. Am J Clin Nutr 99(3):22
Hsu JW, Ball RO, Pencharz PB (2007) Evidence that phenylalanine may not provide the full needs for aromatic amino acids in children. Pediatr Res 61(3):361–365
van Loon LJ, Saris WH, Verhagen H, Wagenmakers AJ (2000) Plasma insulin responses after ingestion of different amino acid or protein mixtures with carbohydrate. Am J Clin Nutr 72(1):96–105
Maggio M, De Vita F, Lauretani F, Butto V, Bondi G, Cattabiani C, Nouvenne A, Meschi T, Dall’Aglio E, Ceda GP (2013) IGF-1, the cross road of the nutritional, inflammatory and hormonal pathways to frailty. Nutrients 5(10):4184–4205
Lonnerdal B, Keen CL, Hurley LS (1985) Manganese binding proteins in human and cow’s milk. Am J Clin Nutr 41(3):550–559
Krachler M, Prohaska T, Koellensperger G, Rossipal E, Stingeder G (2000) Concentrations of selected trace elements in human milk and in infant formulas determined by magnetic sector field inductively coupled plasma-mass spectrometry. Biol Trace Elem Res 76(2):97–112
Hiney JK, Srivastava VK, Dees WL (2011) Manganese induces IGF-1 and cyclooxygenase-2 gene expressions in the basal hypothalamus during prepubertal female development. Toxicol Sci 121(2):389–396
Clegg MS, Donovan SM, Monaco MH, Baly DL, Ensunsa JL, Keen CL (1998) The influence of manganese deficiency on serum IGF-1 and IGF binding proteins in the male rat. Proc Soc Exp Biol Med 219(1):41–47
Karl JP, Alemany JA, Koenig C, Kraemer WJ, Frystyk J, Flyvbjerg A, Young AJ, Nindl BC (2009) Diet, body composition, and physical fitness influences on IGF-I bioactivity in women. Growth Horm IGF Res 19(6):491–496
Maggio M, Ceda GP, Lauretani F, Bandinelli S, Dall’Aglio E, Guralnik JM, Paolisso G, Semba RD, Nouvenne A, Borghi L, Ceresini G, Ablondi F, Benatti M, Ferrucci L (2010) Association of plasma selenium concentrations with total IGF-1 among older community-dwelling adults: the InCHIANTI study. Clin Nutr 29(5):674–677
Zhao JP, Levy E, Fraser WD, Julien P, Delvin E, Montoudis A, Spahis S, Garofalo C, Nuyt AM, Luo ZC (2014) Circulating docosahexaenoic acid levels are associated with fetal insulin sensitivity. PLoS One 9 (1)
Guelzim N, Huneau JF, Mathe V, Tesseraud S, Mourot J, Simon N, Hermier D (2014) N-3 fatty acids improve body composition and insulin sensitivity during energy restriction in the rat. Prostaglandins Leukot Essent Fatty Acids 91(5):203–211
Makrides M, Gibson RA, Udell T, Ried K (2005) Supplementation of infant formula with long-chain polyunsaturated fatty acids does not influence the growth of term infants. Am J Clin Nutr 81(5):1094–1101
Nommsen LA, Lovelady CA, Heinig MJ, Lonnerdal B, Dewey KG (1991) Determinants of energy, protein, lipid, and lactose concentrations in human milk during the first 12 mo of lactation: the darling study. Am J Clin Nutr 53(2):457–465
Zhang Z, Adelman AS, Rai D, Boettcher J, Lonnerdal B (2013) Amino acid profiles in term and preterm human milk through lactation: a systematic review. Nutrients 5(12):4800–4821
Food and Nutrition Board (2001) dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids. The National Academies Press, Washington
Jensen RG, Bitman J, Carlson SE, Couch SC, Hamosh M, Newburg DS (1995) Chapter 6—milk lipids: a. human milk lipids. In: Jensen RG (ed) Handbook of milk composition. Academic Press, San Diego, pp 495–542
Neville MC, Zhang P, Allen JC (1995) CHAPTER 7—minerals, ions, and trace elements in milk: a. Ionic Interactions in Milk. In: Jensen RG (ed) Handbook of Milk Composition. Academic Press, San Diego, pp 577–592
Acknowledgments
We are very grateful for the wonderful support of the study by Tatjana Nikolic and Dr. Branka Trisic during the preparation, recruitment and performance of the study in Belgrade, Serbia.
Source of funding
The clinical trial was financially supported by HiPP GmbH and Co Vertrieb KG (Pfaffenhofen, Germany). Work reported herein is carried out with partial financial support from the European Research Council Advanced Grant ERC-2012-AdG-No. 322605 META-GROWTH.
Author contribution
MB, PG and MB were responsible for IGF-I and insulin analysis; MF was responsible for amino acid analysis; VG and MF were responsible for statistical analyses; MF, HD, VG and BK undertook data interpretation; MF and HD were responsible for writing the manuscript. Finally, all authors critically revised the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have made substantial contributions to the conception and design of the study, or acquisition of data, or analysis and interpretation of data, drafting the article or revising it critically for important intellectual content. Each author has seen and approved the contents of the submitted manuscript. None of the authors had any personal or financial conflict of interest.
Ethical standards
Manuscripts submitted for publication include data of the BeMIM study Fleddermann et al. (2013). The study was conducted according to the guidelines laid down in the Declaration of Helsinki 1964 and all procedures involving human subjects were approved by the ethical committee of the Clinical Center Serbia Ethical Committee. Written informed consent was obtained from all parents prior to study start after the experimental protocol had been explained to them in detail. The ethical approval to do further analyses with anonymized data and blood samples was obtained by the Ethical Committee of the Medical Faculty of Ludwig Maximilians University of Munich, Germany.
Additional information
The study was registered at ClinicalTrials.gov (NCT01094080).
Rights and permissions
About this article
Cite this article
Fleddermann, M., Demmelmair, H., Grote, V. et al. Role of selected amino acids on plasma IGF-I concentration in infants. Eur J Nutr 56, 613–620 (2017). https://doi.org/10.1007/s00394-015-1105-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-015-1105-9