Abstract
Purpose
The Mediterranean diet rich in fruits, vegetables and olive oil has been related to a lower osteoporosis incidence and accordingly to a reduced fracture risk. These observations might be mediated by the active constituents of extra virgin olive oil, and especially polyphenols. In the context of exploring the features of olive oil active constituents on postmenopausal osteoporosis, an extra virgin olive oil total polyphenolic fraction (TPF) was isolated and its effect on the bone loss attenuation was investigated.
Methods
Female Lewis rats were ovariectomized and fed a diet enriched with a total phenolic extract of extra virgin olive oil in a concentration of 800 mg/kg diet.
Results
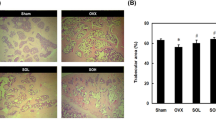
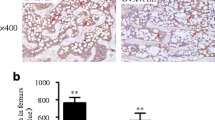
Oleocanthal, one compound of the polyphenolic fraction, showed a higher relative estrogen receptor binding affinity to the ERα compared to the ERβ. While the TPF only slightly induced the uterine wet weight (490.7 ± 53.7 vs. 432.7 ± 23, p = 0.058), TPF regulated estrogen response genes in the uterus (progesterone receptor, antigen identified by monoclonal antibody Ki67, complement C3). Comparing the quantified bone parameters, the oral TPF substitution did not attenuate the ovariectomy-induced bone loss.
Conclusions
The administration of extra virgin olive oil polyphenols regulated uterine estrogen response marker genes in an E2-agonistic manner. The bone loss induced by estrogen ablation was not mitigated by treatment with the polyphenolic extract.




Similar content being viewed by others
Abbreviations
- EVOO:
-
Extra virgin olive oil
- TPF:
-
Total polyphenolic fraction
References
Buckland G, Travier N, Cottet V et al (2012) Adherence to the Mediterranean diet and risk of breast cancer in the European prospective investigation into cancer and nutrition cohort study. Int J Cancer. doi:10.1002/ijc.27958
Yusof AS, Isa ZM, Shah SA (2012) Dietary patterns and risk of colorectal cancer: a systematic review of cohort studies (2000–2011). Asian Pac J Cancer Prev 13:4713–4717
Dilis V, Katsoulis M, Lagiou P et al (2012) Mediterranean diet and CHD: the Greek European prospective investigation into cancer and nutrition cohort. Br J Nutr 108:699–709. doi:10.1017/S0007114512001821
Buckland G, Travier N, Barricarte A et al (2012) Olive oil intake and CHD in the European prospective investigation into cancer and nutrition Spanish cohort. Br J Nutr 108:2075–2082. doi:10.1017/S000711451200298X
Ahluwalia N, Andreeva VA, Kesse-Guyot E, Hercberg S (2012) Dietary patterns, inflammation and the metabolic syndrome. Diabetes Metab. doi:10.1016/j.diabet.2012.08.007
Viscogliosi G, Cipriani E, Liguori ML et al (2013) Mediterranean dietary pattern adherence: associations with prediabetes, metabolic syndrome, and related micro inflammation. Metab Syndr Relat Disord. doi:10.1089/met.2012.0168
Johnell O, Gullberg B, Allander E, Kanis DJA (1992) The apparent incidence of hip fracture in Europe: a study of national register sources. Osteoporos Int 2:298–302. doi:10.1007/BF01623186
Kanis JA (1993) The incidence of hip fracture in Europe. Osteoporos Int 3:10–15. doi:10.1007/BF01621853
Benetou V, Orfanos P, Pettersson-Kymmer U et al (2012) Mediterranean diet and incidence of hip fractures in a European cohort. Osteoporos Int 24:1587–1598. doi:10.1007/s00198-012-2187-3
Perez-Martinez P, Garcia-Rios A, Delgado-Lista J et al (2011) Mediterranean diet rich in olive oil and obesity, metabolic syndrome and diabetes mellitus. Curr Pharm Des 17:769–777. doi:10.2174/138161211795428948
Lopez S, Bermudez B, Varela LM et al (2012) Olives and olive oil: diet and health impacts. CAB Rev. doi:10.1079/PAVSNNR20127034
Covas M-I (2007) Olive oil and the cardiovascular system. Pharmacol Res 55:175–186. doi:10.1016/j.phrs.2007.01.010
Cicerale S, Conlan XA, Sinclair AJ, Keast RSJ (2008) Chemistry and health of olive oil phenolics. Crit Rev Food Sci Nutr 49:218–236. doi:10.1080/10408390701856223
Puel C, Quintin A, Agalias A et al (2004) Olive oil and its main phenolic micronutrient (oleuropein) prevent inflammation-induced bone loss in the ovariectomised rat. Br J Nutr 92:119–127. doi:10.1079/BJN20041181
Puel C, Mathey J, Agalias A et al (2006) Dose–response study of effect of oleuropein, an olive oil polyphenol, in an ovariectomy/inflammation experimental model of bone loss in the rat. Clin Nutr 25:859–868. doi:10.1016/j.clnu.2006.03.009
Puel C, Mardon J, Kati-coulibaly S et al (2007) Black Lucques olives prevented bone loss caused by ovariectomy and talc granulomatosis in rats. Br J Nutr 97:1012–1020. doi:10.1017/S0007114507659030
Puel C, Mardon J, Agalias A et al (2008) Major phenolic compounds in olive oil modulate bone loss in an ovariectomy/inflammation experimental model. J Agric Food Chem 56:9417–9422. doi:10.1021/jf801794q
Saleh NK, Saleh HA (2011) Olive oil effectively mitigates ovariectomy-induced osteoporosis in rats. BMC Complem Altern Med 11:10. doi:10.1186/1472-6882-11-10
Hagiwara K, Goto T, Araki M et al (2011) Olive polyphenol hydroxytyrosol prevents bone loss. Eur J Pharmacol 662:78–84. doi:10.1016/j.ejphar.2011.04.023
Santiago-Mora R, Casado-Díaz A, Castro MDD, Quesada-Gómez JM (2011) Oleuropein enhances osteoblastogenesis and inhibits adipogenesis: the effect on differentiation in stem cells derived from bone marrow. Osteoporos Int 22:675–684. doi:10.1007/s00198-010-1270-x
Beauchamp GK, Keast RSJ, Morel D et al (2005) Phytochemistry: ibuprofen-like activity in extra-virgin olive oil. Nature 437:45–46. doi:10.1038/437045a
Smith AB, Han Q, Breslin PAS, Beauchamp GK (2005) Synthesis and assignment of absolute configuration of (−)-oleocanthal: a potent, naturally occurring non-steroidal anti-inflammatory and anti-oxidant agent derived from extra virgin olive oils. Org Lett 7:5075–5078. doi:10.1021/ol052106a
Iacono A, Gómez R, Sperry J et al (2010) Effect of oleocanthal and its derivatives on inflammatory response induced by lipopolysaccharide in a murine chondrocyte cell line. Arthritis Rheum 62:1675–1682. doi:10.1002/art.27437
Scotece M, Gómez R, Conde J et al (2012) Further evidence for the anti-inflammatory activity of oleocanthal: inhibition of MIP-1α and IL-6 in J774 macrophages and in ATDC5 chondrocytes. Life Sci 91:1229–1235. doi:10.1016/j.lfs.2012.09.012
Cicerale S, Lucas L, Keast R (2012) Antimicrobial, antioxidant and anti-inflammatory phenolic activities in extra virgin olive oil. Curr Opin Biotechnol 23:129–135. doi:10.1016/j.copbio.2011.09.006
Pitt J, Roth W, Lacor P et al (2009) Alzheimer’s-associated Aβ oligomers show altered structure, immunoreactivity and synaptotoxicity with low doses of oleocanthal. Toxicol Appl Pharmacol 240:189–197. doi:10.1016/j.taap.2009.07.018
Li W, Sperry JB, Crowe A et al (2009) Inhibition of tau fibrillization by oleocanthal via reaction with the amino groups of tau. J Neurochem 110:1339–1351. doi:10.1111/j.1471-4159.2009.06224.x
Monti MC, Margarucci L, Tosco A et al (2011) New insights on the interaction mechanism between tau protein and oleocanthal, an extra-virgin olive-oil bioactive component. Food Funct 2:423–428. doi:10.1039/C1FO10064E
Monti MC, Margarucci L, Riccio R, Casapullo A (2012) Modulation of Tau protein fibrillization by oleocanthal. J Nat Prod 75:1584–1588. doi:10.1021/np300384h
Abuznait AH, Qosa H, Busnena BA et al (2013) Olive-oil-derived oleocanthal enhances β-amyloid clearance as a potential neuroprotective mechanism against Alzheimer’s disease. In vitro and in vivo studies. ACS Chem Neurosci. doi:10.1021/cn400024q
Khanal P, Oh W-K, Yun HJ et al (2011) p-HPEA-EDA, a phenolic compound of virgin olive oil, activates AMP-activated protein kinase to inhibit carcinogenesis. Carcinogenesis 32:545–553. doi:10.1093/carcin/bgr001
Elnagar A, Sylvester P, El Sayed K (2011) (−)-Oleocanthal as a c-Met inhibitor for the control of metastatic breast and prostate cancers. Planta Med 77:1013–1019. doi:10.1055/s-0030-1270724
Parzonko A, Naruszewicz M (2012) Oleacein protects endothelial progenitor cells against angiotensin II—induced oxidative stress. Planta Med. doi:10.1055/s-0032-1320446
Kanakis P, Termentzi A, Michel T et al (2012) From drupes to olive oil: how do bioactives variate during a single production procedure? Planta Med. doi:10.1055/s-0032-1321201
Nelson HD Menopause. The Lancet 371:760–770. doi: 10.1016/S0140-6736(08)60346-3
(1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO study group. World Health Organ Tech Rep Ser 843:1–129
(2000) Osteoporosis prevention, diagnosis, and therapy. NIH Consens Statement 17:1–45
Kanis JA, McCloskey EV, Johansson H et al (2008) A reference standard for the description of osteoporosis. Bone 42:467–475. doi:10.1016/j.bone.2007.11.001
Kalu DN (1991) The ovariectomized rat model of postmenopausal bone loss. Bone Miner 15:175–191
Turner RT, Maran A, Lotinun S et al (2001) Animal models for osteoporosis. Rev Endocr Metab Disord 2:117–127
Jee WS, Yao W (2001) Overview: animal models of osteopenia and osteoporosis. J Musculoskelet Neuronal Interact 1:193–207
Lelovas PP, Xanthos TT, Thoma SE et al (2008) The laboratory rat as an animal model for osteoporosis research. Comp Med 58:424–430
Andreadou I, Iliodromitis EK, Mikros E et al (2006) The olive constituent oleuropein exhibits anti-ischemic, antioxidative, and hypolipidemic effects in anesthetized rabbits. J Nutr 136:2213–2219
Agalias A, Magiatis P, Skaltsounis A-L et al (2007) A new process for the management of olive oil mill waste water and recovery of natural antioxidants. J Agric Food Chem 55:2671–2676. doi:10.1021/jf063091d
Karkoula E, Skantzari A, Melliou E, Magiatis P (2012) Direct measurement of oleocanthal and oleacein levels in olive oil by quantitative 1H NMR. establishment of a new index for the characterization of extra virgin olive oils. J Agric Food Chem 60:11696–11703. doi:10.1021/jf3032765
Bouxsein ML, Boyd SK, Christiansen BA et al (2010) Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res 25:1468–1486. doi:10.1002/jbmr.141
Sirianni R, Chimento A, De Luca A et al (2010) Oleuropein and hydroxytyrosol inhibit MCF-7 breast cancer cell proliferation interfering with ERK1/2 activation. Mol Nutr Food Res 54:833–840. doi:10.1002/mnfr.200900111
Kuiper GGJM, Carlsson B, Grandien K et al (1997) Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors α and β. Endocrinology 138:863–870. doi:10.1210/en.138.3.863
Kuiper GGJM, Lemmen JG, Carlsson B et al (1998) Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor β. Endocrinology 139:4252–4263. doi:10.1210/en.139.10.4252
Trichopoulou A, Georgiou E, Bassiakos Y et al (1997) Energy intake and monounsaturated fat in relation to bone mineral density among women and men in Greece. Prev Med 26:395–400. doi:10.1006/pmed1997.0160
Wauquier F, Philippe C, Leotoing L et al (2013) The free fatty acid receptor G protein-coupled receptor 40 (GPR40) Protects from bone loss through inhibition of osteoclast differentiation. J Biol Chem 288:6542–6551. doi:10.1074/jbc.M112.429084
Acknowledgments
The present work was funded by SYNERGASIA 2009 PROGRAMME. This Programme is co-funded by the European Regional Development Fund and National Resources (Project code: OSTEOPRO-1076).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keiler, A.M., Zierau, O., Bernhardt, R. et al. Impact of a functionalized olive oil extract on the uterus and the bone in a model of postmenopausal osteoporosis. Eur J Nutr 53, 1073–1081 (2014). https://doi.org/10.1007/s00394-013-0609-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-013-0609-4