Abstract
Purpose
Six-transmembrane epithelial antigen of prostate (STEAP)-4 and neutrophil gelatinase-associated lipocalin (NGAL) are novel adipokines related to iron homeostasis with potential roles in insulin resistance and inflammation. The aim of the present work was to evaluate the effect of obesity and iron status on gene expression levels of STEAP-4 and NGAL in visceral adipose tissue (VAT) and its implication in inflammation.
Methods
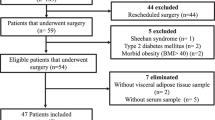
VAT biopsies obtained from 53 subjects were used in the study. Real-time PCR and Western-blot were performed to quantify the levels of STEAP4 and NGAL in VAT as well as the association with other genes implicated in inflammatory pathways. Circulating ferritin and free iron concentrations were also determined.
Results
Obese patients exhibited significantly increased STEAP4 and NGAL mRNA expression levels (P < 0.001) compared to lean subjects. Protein expression levels of NGAL (P < 0.05) and STEAP4 were also higher in the visceral fat depot of obese patients, although protein levels of STEAP4 did not reach statistical significance. A negative correlation (P < 0.05) between free iron concentrations and gene expression levels of both STEAP4 and NGAL was found, while circulating ferritin concentrations were positively correlated (P < 0.05) with NGAL mRNA after body fat (BF) adjustment. Furthermore, a significant positive association between STEAP4 and NGAL gene expression levels with inflammatory markers was also detected (P < 0.01).
Conclusion
These findings represent the first observation that STEAP4 and NGAL mRNA and protein levels in human VAT are related to iron status. Moreover, STEAP4 and NGAL are associated with pro-inflammatory markers suggesting their potential involvement in the low-grade chronic inflammation accompanying obesity.


Similar content being viewed by others
References
Frühbeck G, Gómez-Ambrosi J (2003) Control of body weight: a physiologic and transgenic perspective. Diabetologia 46:143–172
Haslam DW, James WP (2005) Obesity. Lancet 366:1197–1209
Fernández-Real JM, López-Bermejo A, Ricart W (2002) Cross-talk between iron metabolism and diabetes. Diabetes 51:2348–2354
Piperno A, Trombini P, Gelosa M, Mauri V, Pecci V, Vergani A, Salvioni A, Mariani R, Mancia G (2002) Increased serum ferritin is common in men with essential hypertension. J Hypertens 20:1513–1518
Menke A, Muntner P, Fernández-Real JM, Guallar E (2012) The association of biomarkers of iron status with mortality in US adults. Nutr Metab Cardiovasc Dis 22:734–740
Ford ES, Cogswell ME (1999) Diabetes and serum ferritin concentration among U.S. adults. Diabetes Care 22:1978–1983
Ferrannini E (2000) Insulin resistance, iron, and the liver. Lancet 355:2181–2182
Guillen C, McInnes IB, Kruger H, Brock JH (1998) Iron, lactoferrin and iron regulatory protein activity in the synovium; relative importance of iron loading and the inflammatory response. Ann Rheum Dis 57:309–314
Jehn M, Clark JM, Guallar E (2004) Serum ferritin and risk of the metabolic syndrome in U.S. adults. Diabetes Care 27:2422–2428
Freixenet N, Remacha A, Berlanga E, Caixas A, Gimenez-Palop O, Blanco-Vaca F, Bach V, Baiget M, Sanchez Y, Felez J, Gonzalez-Clemente JM (2009) Serum soluble transferrin receptor concentrations are increased in central obesity. Results from a screening programme for hereditary hemochromatosis in men with hyperferritinemia. Clin Chim Acta 400:111–116
Finch C (1994) Regulators of iron balance in humans. Blood 84:1697–1702
Anderson GJ, Darshan D, Wilkins SJ, Frazer DM (2007) Regulation of systemic iron homeostasis: how the body responds to changes in iron demand. Biometals 20:665–674
Swaminathan S, Fonseca VA, Alam MG, Shah SV (2007) The role of iron in diabetes and its complications. Diabetes Care 30:1926–1933
Gkouvatsos K, Papanikolaou G, Pantopoulos K (2012) Regulation of iron transport and the role of transferrin. Biochim Biophys Acta 1820:188–202
Bao G, Clifton M, Hoette TM, Mori K, Deng SX, Qiu A, Viltard M, Williams D, Paragas N, Leete T, Kulkarni R, Li X, Lee B, Kalandadze A, Ratner AJ, Pizarro JC, Schmidt-Ott KM, Landry DW, Raymond KN, Strong RK, Barasch J (2010) Iron traffics in circulation bound to a siderocalin (Ngal)-catechol complex. Nat Chem Biol 6:602–609
Wang Y, Lam KS, Kraegen EW, Sweeney G, Zhang J, Tso AW, Chow WS, Wat NM, Xu JY, Hoo RL, Xu A (2007) Lipocalin-2 is an inflammatory marker closely associated with obesity, insulin resistance, and hyperglycemia in humans. Clin Chem 53:34–41
Wellen KE, Fucho R, Gregor MF, Furuhashi M, Morgan C, Lindstad T, Vaillancourt E, Gorgun CZ, Saatcioglu F, Hotamisligil GS (2007) Coordinated regulation of nutrient and inflammatory responses by STAMP2 is essential for metabolic homeostasis. Cell 129:537–548
Flo TH, Smith KD, Sato S, Rodriguez DJ, Holmes MA, Strong RK, Akira S, Aderem A (2004) Lipocalin 2 mediates an innate immune response to bacterial infection by sequestrating iron. Nature 432:917–921
Torti SV, Torti FM (2011) Ironing out cancer. Cancer Res 71:1511–1514
Catalán V, Gómez-Ambrosi J, Rodríguez A, Ramírez B, Silva C, Rotellar F, Gil MJ, Cienfuegos JA, Salvador J, Frühbeck G (2009) Increased adipose tissue expression of lipocalin-2 in obesity is related to inflammation and matrix metalloproteinase-2 and metalloproteinase-9 activities in humans. J Mol Med 87:803–813
Esteve E, Ricart W, Fernández-Real JM (2009) Adipocytokines and insulin resistance: the possible role of lipocalin-2, retinol binding protein-4, and adiponectin. Diabetes Care 32(Suppl 2):S362–367
Frühbeck G, Gómez-Ambrosi J (2001) Rationale for the existence of additional adipostatic hormones. FASEB J 15:1996–2006
Moldes M, Lasnier F, Gauthereau X, Klein C, Pairault J, Feve B, Chambaut-Guerin AM (2001) Tumor necrosis factor-α-induced adipose-related protein (TIARP), a cell-surface protein that is highly induced by tumor necrosis factor-α and adipose conversion. J Biol Chem 276:33938–33946
Ohgami RS, Campagna DR, McDonald A, Fleming MD (2006) The Steap proteins are metalloreductases. Blood 108:1388–1394
Arner P, Stenson BM, Dungner E, Naslund E, Hoffstedt J, Ryden M, Dahlman I (2008) Expression of six transmembrane protein of prostate 2 in human adipose tissue associates with adiposity and insulin resistance. J Clin Endocrinol Metab 93:2249–2254
De Domenico I, McVey Ward D, Kaplan J (2008) Regulation of iron acquisition and storage: consequences for iron-linked disorders. Nat Rev Mol Cell Biol 9:72–81
Hotamisligil GS, Shargill NS, Spiegelman BM (1993) Adipose expression of tumor necrosis factor-α: direct role in obesity-linked insulin resistance. Science 259:87–91
Catalán V, Gómez-Ambrosi J, Rodríguez A, Salvador J, Frühbeck G (2009) Adipokines in the treatment of diabetes mellitus and obesity. Expert Opin Pharmacother 10:239–254
Catalán V, Gómez-Ambrosi J, Rodríguez A, Frühbeck G (2012) Role of extracellular matrix remodelling in adipose tissue pathophysiology. Relevance in the development of obesity. Histol Histopathol 27:1515–1528
Gómez-Ambrosi J, Catalán V, Ramírez B, Rodríguez A, Colina I, Silva C, Rotellar F, Mugueta C, Gil MJ, Cienfuegos JA, Salvador J, Frühbeck G (2007) Plasma osteopontin levels and expression in adipose tissue are increased in obesity. J Clin Endocrinol Metab 92:3719–3727
Gómez-Ambrosi J, Silva C, Galofré JC, Escalada J, Santos S, Millán D, Vila N, Ibañez P, Gil MJ, Valentí V, Rotellar F, Ramírez B, Salvador J, Frühbeck G (2012) Body mass index classification misses subjects with increased cardiometabolic risk factors related to elevated adiposity. Int J Obes 36:286–294
Ginde SR, Geliebter A, Rubiano F, Silva AM, Wang J, Heshka S, Heymsfield SB (2005) Air displacement plethysmography: validation in overweight and obese subjects. Obes Res 13:1232–1237
Gómez-Ambrosi J, Frühbeck G, Martínez JA (1999) Leptin, but not a β3-adrenergic agonist, upregulates muscle uncoupling protein-3 messenger RNA expression: short-term thermogenic interactions. Cell Mol Life Sci 55:992–997
Gómez-Ambrosi J, Salvador J, Rotellar F, Silva C, Catalán V, Rodríguez A, Gil MJ, Frühbeck G (2006) Increased serum amyloid A concentrations in morbid obesity decrease after gastric bypass. Obes Surg 16:262–269
Catalán V, Gómez-Ambrosi J, Ramírez B, Rotellar F, Pastor C, Silva C, Rodríguez A, Gil MJ, Cienfuegos JA, Frühbeck G (2007) Proinflammatory cytokines in obesity: impact of type 2 diabetes mellitus and gastric bypass. Obes Surg 17:1464–1474
Muruzábal FJ, Frühbeck G, Gómez-Ambrosi J, Archanco M, Burrell MA (2002) Immunocytochemical detection of leptin in non-mammalian vertebrate stomach. Gen Comp Endocrinol 128:149–152
Catalán V, Gómez-Ambrosi J, Rotellar F, Silva C, Rodríguez A, Salvador J, Gil MJ, Cienfuegos JA, Frühbeck G (2007) Validation of endogenous control genes in human adipose tissue: relevance to obesity and obesity-associated type 2 diabetes mellitus. Horm Metab Res 39:495–500
Gygi SP, Rochon Y, Franza BR, Aebersold R (1999) Correlation between protein and mRNA abundance in yeast. Mol Cell Biol 19:1720–1730
Guo Y, Xiao P, Lei S, Deng F, Xiao GG, Liu Y, Chen X, Li L, Wu S, Chen Y, Jiang H, Tan L, Xie J, Zhu X, Liang S, Deng H (2008) How is mRNA expression predictive for protein expression? A correlation study on human circulating monocytes. Acta Biochim Biophys Sin 40:426–436
Zhang CM, Chi X, Wang B, Zhang M, Ni YH, Chen RH, Li XN, Guo XR (2008) Downregulation of STEAP4, a highly-expressed TNF-α-inducible gene in adipose tissue, is associated with obesity in humans. Acta Pharmacol Sin 29:587–592
Moreno-Navarrete JM, Ortega F, Serrano M, Pérez-Pérez R, Sabater M, Ricart W, Tinahones F, Peral B, Fernández-Real JM (2011) Decreased STAMP2 expression in association with visceral adipose tissue dysfunction. J Clin Endocrinol Metab 2011:E1816–1825
Zhang J, Wu Y, Zhang Y, Leroith D, Bernlohr DA, Chen X (2008) The role of lipocalin 2 in the regulation of inflammation in adipocytes and macrophages. Mol Endocrinol 22:1416–1426
Cheng HL, Bryant C, Cook R, O’Connor H, Rooney K, Steinbeck K (2012) The relationship between obesity and hypoferraemia in adults: a systematic review. Obes Rev 13:150–161
Harrison PM, Arosio P (1996) The ferritins: molecular properties, iron storage function and cellular regulation. Biochim Biophys Acta 1275:161–203
Knutson MD (2007) Steap proteins: implications for iron and copper metabolism. Nutr Rev 65:335–340
Anderson GJ, Vulpe CD (2009) Mammalian iron transport. Cell Mol Life Sci 66:3241–3261
Theil EC, Goss DJ (2009) Living with iron (and oxygen): questions and answers about iron homeostasis. Chem Rev 109:4568–4579
ten Freyhaus H, Calay ES, Yalcin A, Vallerie SN, Yang L, Calay ZZ, Saatcioglu F, Hotamisligil GS (2012) Stamp2 controls macrophage inflammation through nicotinamide adenine dinucleotide phosphate homeostasis and protects against atherosclerosis. Cell Metab 16:81–89
Guo H, Jin D, Zhang Y, Wright W, Bazuine M, Brockman DA, Bernlohr DA, Chen X (2010) Lipocalin-2 deficiency impairs thermogenesis and potentiates diet-induced insulin resistance in mice. Diabetes 59:1376–1385
Law IK, Xu A, Lam KS, Berger T, Mak TW, Vanhoutte PM, Liu JT, Sweeney G, Zhou M, Yang B, Wang Y (2010) Lipocalin-2 deficiency attenuates insulin resistance associated with aging and obesity. Diabetes 59:872–882
Acknowledgments
This work was supported by Fondo de Investigacion Sanitaria (FIS) PI09/02330, PI08/1146, PI11/02681 from the Spanish Instituto de Salud Carlos III, by the Department of Health (48/2011 and 58/2011) of the Gobierno de Navarra of Spain and by Plan de Investigacion de la Universidad de Navarra (PIUNA) (2009–2011).
The authors gratefully acknowledge the valuable collaboration of all the members of the Multidisciplinary Obesity Team, Clínica Universidad de Navarra, Pamplona, Spain. CIBER de Fisiopatología de la Obesidad y Nutrición (CIBEROBN) is an initiative of the Instituto de Salud Carlos III, Spain.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Catalán, V., Gómez-Ambrosi, J., Rodríguez, A. et al. Six-transmembrane epithelial antigen of prostate 4 and neutrophil gelatinase-associated lipocalin expression in visceral adipose tissue is related to iron status and inflammation in human obesity. Eur J Nutr 52, 1587–1595 (2013). https://doi.org/10.1007/s00394-012-0464-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-012-0464-8