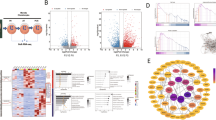
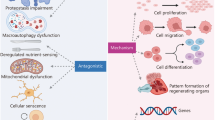
Abstract
The precise molecular mechanisms associated with osteoarthritis (OA), the most common musculoskeletal disorder, are poorly understood. There are currently no effective treatments to prevent the initiation and progression of the disease. In recent years, the development of mRNAome has made it possible to identify new mechanisms and therapeutic targets. However, the differentially expressed genes screened by different microarrays are not completely the same. In order to avoid this shortcoming, we integrate the different genes from different tissues and data sets, and select the commonly expressed genes for further studies.
Zusammenfassung
Bisher sind die genauen molekularen Mechanismen, die mit Arthrose, der häufigsten mukuloskeletalen Erkrankung, einhergehen, nur unzureichend verstanden. Derzeit stehen keine wirksamen Therapien zu Verfügung, um den Beginn und das Fortschreiten der Krankheit zu verhindern. In den letzten Jahren ist es durch die Entwicklung des mRNAoms möglich geworden, neue Mechanismen und therapeutische Ziele zu identifizieren. Aber die unterschiedlich exprimierten Gene, die mittels verschiedener Microarrays untersucht werden, sind nicht völlig identisch. Um diesem Mangel abzuhelfen, werden die unterschiedlichen Gene aus verschiedenen Geweben und Datensätzen integriert und die häufig exprimierten Gene für weitere Studien ausgewählt.





Similar content being viewed by others
References
Dahaghin S, Bierma-Zeinstra SM, Ginai AZ, Pols HA, Hazes JM, Koes BW (2005) Prevalence and pattern of radiographic hand osteoarthritis and association with pain and disability (the Rotterdam study). Ann Rheum Dis 64(5):682–687
Oliveria SA, Felson DT, Reed JI, Cirillo PA, Walker AM (1995) Incidence of symptomatic hand, hip, and knee osteoarthritis among patients in a health maintenance organization. Arthritis Rheum 38(8):1134–1141
Dillon CF, Rasch EK, Gu Q, Hirsch R (2006) Prevalence of knee osteoarthritis in the United States: arthritis data from the third national health and nutrition examination survey 1991–94. J Rheumatol 33(11):2271–2279
Xia B, Di C, Zhang J, Hu S, Jin H, Tong P (2014) Osteoarthritis pathogenesis: a review of molecular mechanisms. Calcif Tissue Int 95(6):495–505
Felson DT (2006) Clinical practice. Osteoarthritis of the knee. N Engl J Med 354(8):841–848
Hunter DJ, March L, Sambrook PN (2002) Knee osteoarthritis: the influence of environmental factors. Clin Exp Rheumatol 20(1):93–100
Findlay DM, Atkins GJ (2014) Osteoblast-chondrocyte interactions in osteoarthritis. Curr Osteoporos Rep 12(1):127–134
Zhong L, Huang X, Karperien M, Post JN (2015) The regulatory role of signaling crosstalk in hypertrophy of MSCs and human articular chondrocytes. Int J Mol Sci 16(8):19225–19247
Chen YJ, Chang WA, Wu LY, Hsu YL, Chen CH, Kuo PL (2018) Systematic analysis of transcriptomic profile of chondrocytes in osteoarthritic knee using next-generation sequencing and bioinformatics. J Clin Med 7(12):535. https://doi.org/10.3390/jcm7120535
Scanzello CR, Goldring SR (2012) The role of synovitis in osteoarthritis pathogenesis. Bone 51(2):249–257
Griffin TM, Scanzello CR (2019) Innate inflammation and synovial macrophages in osteoarthritis pathophysiology. Clin Exp Rheumatol 37(120):57–63
Kraus VB, McDaniel G, Huebner JL, Stabler TV, Pieper CF, Shipes SW et al (2016) Direct in vivo evidence of activated macrophages in human osteoarthritis. Osteoarthr Cartil 24(9):1613–1621
Zhang H, Cai D, Bai X (2020) Macrophages regulate the progression of osteoarthritis. Osteoarthr Cartil 28(5):555–561
Zhou Y, Wang Z, Chen X, Zhang J, Yang L, Liu S et al (2020) Identification of differentially expressed miRNAs and mRNAs in synovial of osteoarthritis via RNA-sequencing. BMC Med Genet 21(1):46
Zhang H, Chen C, Cui Y, Li Y, Wang Z, Mao X et al (2019) lnc-SAMD14‑4 can regulate expression of the COL1A1 and COL1A2 in human chondrocytes. PeerJ 7:e7491
Fisch KM, Gamini R, Alvarez-Garcia O, Akagi R, Saito M, Muramatsu Y et al (2018) Identification of transcription factors responsible for dysregulated networks in human osteoarthritis cartilage by global gene expression analysis. Osteoarthr Cartil 26(11):1531–1538
Brophy RH, Zhang B, Cai L, Wright RW, Sandell LJ, Rai MF (2018) Transcriptome comparison of meniscus from patients with and without osteoarthritis. Osteoarthr Cartil 26(3):422–432
Chou CH, Wu CC, Song IW, Chuang HP, Lu LS, Chang JH et al (2013) Genome-wide expression profiles of subchondral bone in osteoarthritis. Arthritis Res Ther 15(6):R190
Ramos YF, Bos SD, Lakenberg N, Bohringer S, den Hollander WJ, Kloppenburg M et al (2014) Genes expressed in blood link osteoarthritis with apoptotic pathways. Ann Rheum Dis 73(10):1844–1853
Hu SI, Carozza M, Klein M, Nantermet P, Luk D, Crowl RM (1998) Human HtrA, an evolutionarily conserved serine protease identified as a differentially expressed gene product in osteoarthritic cartilage. J Biol Chem 273(51):34406–34412
Hou Y, Lin H, Zhu L, Liu Z, Hu F, Shi J et al (2014) The inhibitory effect of IFN-gamma on protease HTRA1 expression in rheumatoid arthritis. J Immunol 193(1):130–138
Tsuchiya A, Yano M, Tocharus J, Kojima H, Fukumoto M, Kawaichi M et al (2005) Expression of mouse HtrA1 serine protease in normal bone and cartilage and its upregulation in joint cartilage damaged by experimental arthritis. Bone 37(3):323–336
Song EK, Jeon J, Jang DG, Kim HE, Sim HJ, Kwon KY et al (2018) ITGBL1 modulates integrin activity to promote cartilage formation and protect against arthritis. Sci Transl Med 10(462):eaam7486. https://doi.org/10.1126/scitranslmed.aam7486
Murayama MA, Kakuta S, Maruhashi T, Shimizu K, Seno A, Kubo S et al (2014) CTRP3 plays an important role in the development of collagen-induced arthritis in mice. Biochem Biophys Res Commun 443(1):42–48
Huang Y, Wan G, Tao J (2017) C1q/TNF-related protein‑3 exerts the chondroprotective effects in IL-1beta-treated SW1353 cells by regulating the FGFR1 signaling. Biomed Pharmacother 85:41–46
Liu R, Shuai Y, Luo J, Zhang Z (2019) SEMA3C promotes cervical cancer growth and is associated with poor prognosis. Front Oncol 9:1035
Zhang H, Ma H, Zhang W, Duan D, Zhu G, Cao W et al (2020) Increased expression of Sema3C indicates a poor prognosis and is regulated by miR-142-5p in glioma. Biol Pharm Bull 43(4):639–648
Yin L, Li J, Wang J, Pu T, Wei J, Li Q et al (2021) MAOA promotes prostate cancer cell perineural invasion through SEMA3C/PlexinA2/NRP1-cMET signaling. Oncogene 40(7):1362–1374
Tang MW, Malvar Fernandez B, Newsom SP, van Buul JD, Radstake T, Baeten DL et al (2018) Class 3 semaphorins modulate the invasive capacity of rheumatoid arthritis fibroblast-like synoviocytes. Baillieres Clin Rheumatol 57(5):909–920
Nanthapisal S, Eleftheriou D, Gilmour K, Leone V, Ramnath R, Omoyinmi E et al (2018) Cutaneous vasculitis and recurrent infection caused by deficiency in complement factor I. Front Immunol 9:735
Tseng MH, Fan WL, Liu H, Yang CY, Ding JJ, Lee HJ et al (2020) Complement factor I mutation may contribute to development of thrombotic microangiopathy in lupus nephritis. Front Med 7:621609
Servais A, Noel LH, Roumenina LT, Le Quintrec M, Ngo S, Dragon-Durey MA et al (2012) Acquired and genetic complement abnormalities play a critical role in dense deposit disease and other C3 glomerulopathies. Kidney Int 82(4):454–464
Tseng MH, Lin SH, Wu CY, Chien HP, Yang HY, Chen YC et al (2018) Serum complement factor I is associated with disease activity of systemic lupus erythematosus. Oncotarget 9(9):8502–8511
Uzquiano A, Cifuentes-Diaz C, Jabali A, Romero DM, Houllier A, Dingli F et al (2019) Mutations in the heterotopia gene Eml1/EML1 severely disrupt the formation of primary cilia. Cell Rep 28(6):1596–1611.e10
Li S, Zhao W, Sun M (2020) An analysis regarding the association between the ISLR gene and gastric carcinogenesisa. Front Genet 11:620
Kobayashi H, Gieniec KA, Wright JA, Wang T, Asai N, Mizutani Y et al (2021) The balance of stromal BMP signaling mediated by GREM1 and ISLR drives colorectal carcinogenesis. Gastroenterology 160(4):1224–1239.e30
Cui C, Han S, Shen X, He H, Chen Y, Zhao J et al (2020) ISLR regulates skeletal muscle atrophy via IGF1-PI3K/Akt-Foxo signaling pathway. Cell Tissue Res 381(3):479–492
de Rotte MC, Bulatovic M, Heijstek MW, Jansen G, Heil SG, van Schaik RH et al (2012) ABCB1 and ABCC3 gene polymorphisms are associated with first-year response to methotrexate in juvenile idiopathic arthritis. J Rheumatol 39(10):2032–2040
de Rotte M, Pluijm SMF, de Jong PHP, Bulatovic Calasan M, Wulffraat NM, Weel A et al (2018) Development and validation of a prognostic multivariable model to predict insufficient clinical response to methotrexate in rheumatoid arthritis. PLoS ONE 13(12):e208534
Xiao C, Lv C, Sun S, Zhao H, Ling H, Li M et al (2021) TSP1 is the essential domain of SEMA5A involved in pannus formation in rheumatoid arthritis. Baillieres Clin Rheumatol 60(12):5833–5842
Gras C, Eiz-Vesper B, Jaimes Y, Immenschuh S, Jacobs R, Witte T et al (2014) Secreted semaphorin 5A activates immune effector cells and is a biomarker for rheumatoid arthritis. Arthritis Rheumatol 66(6):1461–1471
Du Y, Wu X, Chen M, Wang W, Xv W, Ye L et al (2017) Elevated semaphorin5A in systemic lupus erythematosus is in association with disease activity and lupus nephritis. Clin Exp Immunol 188(2):234–242
Yuan L, Le Bras A, Sacharidou A, Itagaki K, Zhan Y, Kondo M et al (2012) ETS-related gene (ERG) controls endothelial cell permeability via transcriptional regulation of the claudin 5 (CLDN5) gene. J Biol Chem 287(9):6582–6591
Cong X, Zhang XM, Zhang Y, Wei T, He QH, Zhang LW et al (2018) Disruption of endothelial barrier function is linked with hyposecretion and lymphocytic infiltration in salivary glands of Sjogren’s syndrome. Biochim Biophys Acta Mol Basis Dis 1864(10):3154–3163
Aho TL, Lund RJ, Ylikoski EK, Matikainen S, Lahesmaa R, Koskinen PJ (2005) Expression of human pim family genes is selectively up-regulated by cytokines promoting T helper type 1, but not T helper type 2, cell differentiation. Immunology 116(1):82–88
Kim K, Kim JH, Youn BU, Jin HM, Kim N (2010) Pim‑1 regulates RANKL-induced osteoclastogenesis via NF-kappaB activation and NFATc1 induction. J Immunol 185(12):7460–7466
Ha YJ, Choi YS, Han DW, Kang EH, Yoo IS, Kim JH et al (2019) PIM‑1 kinase is a novel regulator of proinflammatory cytokine-mediated responses in rheumatoid arthritis fibroblast-like synoviocytes. Baillieres Clin Rheumatol 58(1):154–164
Ma D, Yu T, Peng L, Wang L, Liao Z, Xu W (2019) PIM1, CYP1B1, and HSPA2 targeted by quercetin play important roles in osteoarthritis treatment by achyranthes bidentata. Evid Based Complement Alternat Med 2019:1205942
Fu R, Xia Y, Li M, Mao R, Guo C, Zhou M et al (2019) Pim‑1 as a therapeutic target in lupus nephritis. Arthritis Rheumatol 71(8):1308–1318
Woodell-May JE, Sommerfeld SD (2020) Role of inflammation and the immune system in the progression of osteoarthritis. J Orthop Res 38(2):253–257
Estrada McDermott J, Pezzanite L, Goodrich L, Santangelo K, Chow L, Dow S et al (2021) Role of innate immunity in initiation and progression of osteoarthritis, with emphasis on horses. Animals 11(11):3247
Barreto G, Manninen M, Eklund KK (2020) Osteoarthritis and toll-like receptors: when innate immunity meets chondrocyte apoptosis. Biology 9(4):65
Edilova MI, Akram A, Abdul-Sater AA (2021) Innate immunity drives pathogenesis of rheumatoid arthritis. Biomed J 44(2):172–182
McInnes IB, Schett G (2011) The pathogenesis of rheumatoid arthritis. N Engl J Med 365(23):2205–2219
Funding
This work was supported by grants from the National Natural Science Foundation of China (81901668) and Beijing Jishuitan Hospital Nova Program (XKXX-202112).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S. Gao and H. Song declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies performed were in accordance with the ethical standards indicated in each case.
Additional information
Redaktion
Ulf Müller-Ladner, Bad Nauheim
Uwe Lange, Bad Nauheim

Scan QR code & read article online
Rights and permissions
About this article
Cite this article
Gao, S., Song, H. Integrated comparison of the mRNAome in cartilage, synovium, and macrophages in osteoarthritis. Z Rheumatol 83 (Suppl 1), 62–70 (2024). https://doi.org/10.1007/s00393-022-01171-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00393-022-01171-y