Abstract
Objective
The aim in this study was to determine whether solute carrier family 22, member 4 (SLC22A4), and runt-related transcription factor 1 (RUNX1) polymorphisms are associated with susceptibility to rheumatoid arthritis (RA) in populations of different ethnicities.
Methods
We conducted a literature search using the MEDLINE and EMBASE, and performed a meta-analysis using a fixed or random effects model.
Results
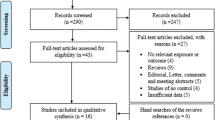
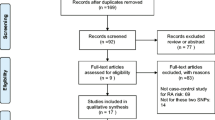
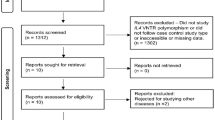
A total of 26 comparative studies from 14 articles met the study inclusion criteria. Studies on the SLC22A4 polymorphism involved 12,458 RA patients and 9283 controls, and studies on the RUNX1 polymorphism involved 3958 RA patients and 3773 controls. The meta-analysis showed no association between the 22 + 21 genotype of the SLC22A4 F1 and RA in overall group [odds ratio (OR) 1.074, 95 % confidence interval (CI) 0.952–1.212, p = 0.245]. After stratification by ethnicity, the meta-analysis indicated that the 22 + 21 genotype of the SLC22A4 F1 was associated significantly with RA in the East Asian population, but not in the European population (OR 1.124, 95 % CI 1.018–1.240, p = 0.021; OR 0.981, 95 % CI 0.773–1.243, p = 0.871).
Conclusion
This meta-analysis demonstrates that the SLC22A4 F1 polymorphism is associated with susceptibility to RA in East Asians, but not in Europeans.
Zusammenfassung
Ziel
Ziel der Studie war es zu ermitteln, ob Polymorphismen des 4. Mitglieds der SLC(solute carrier“)-Familie 22 (SLC22A4) und des „runt-related“ Transkriptionsfaktors 1 (RUNX1) in verschiedenen Ethnien mit der Suszeptibilität für rheumatoide Arthritis assoziiert sind.
Methoden
Nach Publikationen wurde in den Datenbanken MEDLINE und EMBASE gesucht, die Metaanalyse wurde nach einem „fixed or random effects“ Modell durchgeführt.
Ergebnisse
Insgesamt 26 vergleichende Studien aus 14 Publikationen erfüllten die Einschlusskriterien. In die Studien zum SLC22A4-Polymorphismus waren 12,458 RA-Patienten und 9283 Kontrollpersonen eingeschlossen, in die zum RUNX1-Polymorphismen 3958 bzw. 3773. In der Gruppe insgesamt ergab die Metaanalyse keine Assoziation zwischen dem 22 + 21-Genotyp von SLC22A4 F1 und RA (Odds Ratio, OR, 1,074, 95 %-Konfidenzintervall, KI, 0,952–1,212, p = 0,245). Bei Stratifikation nach ethnischer Zugehörigkeit zeigte sich eine statistisch signifikante Assoziation zwischen dem 22 + 21-Genotyp von SLC22A4 F1 und RA im ostasiatischen Kollektiv (OR 1,124, 95 %-KI 1,018–1,240, p = 0,021), nicht dagegen im europäischen Kollektiv (OR 0,981, 95 %-KI 0,773–1,243, p = 0,871).
Fazit
Die Metaanalyse weist eine Assoziation zwischen dem SLC22A4-F1-Polymorphismus und der Suszeptibilität für RA bei Ostasiaten nach, nicht dagegen bei Europäern.



Similar content being viewed by others
References
Choi SJ, Rho YH, Ji JD et al (2006) Genome scan meta-analysis of rheumatoid arthritis. Rheumatology (Oxford) 45:166–170
Cantagrel A, Navaux F, Loubet-Lescoulie P et al (1999) Interleukin-1beta, interleukin-1 receptor antagonist, interleukin-4, and interleukin-10 gene polymorphisms: relationship to occurrence and severity of rheumatoid arthritis. Arthritis Rheum 42:1093–1100
Tokuhiro S, Yamada R, Chang X et al (2003) An intronic SNP in a RUNX1 binding site of SLC22A4, encoding an organic cation transporter, is associated with rheumatoid arthritis. Nat Genet 35:341–348
Alarcon-Riquelme ME (2003) A RUNX trio with a taste for autoimmunity. Nat Genet 35:299–300
Ren TL, Han ZJ, Yang CJ et al (2014) Association of SLC22A4 gene polymorphism with rheumatoid arthritis in the chinese population. J Biochem Mol Toxicol 28:206–210
Plant D, Barton A, Thomson W et al (2009) A re-evaluation of three putative functional single nucleotide polymorphisms in rheumatoid arthritis. Ann Rheum Dis 68:1373–1375
Watanabe Y, Nunokawa A, Kaneko N et al (2009) Two-stage case-control association study of polymorphisms in rheumatoid arthritis susceptibility genes with schizophrenia. J Hum Genet 54:62–65
Okada Y, Mori M, Yamada R et al (2008) SLC22A4 polymorphism and rheumatoid arthritis susceptibility: a replication study in a Japanese population and a metaanalysis. J Rheumatol 35:1723–1728
Komlosi K, Talian GC, Farago B et al (2008) No influence of SLC22A4 C6607T and RUNX1 G24658C genotypic variants on the circulating carnitine ester profile in patients with rheumatoid arthritis. Clin Exp Rheumatol 26:61–66
Takata Y, Inoue H, Sato A et al (2008) Replication of reported genetic associations of PADI4, FCRL3, SLC22A4 and RUNX1 genes with rheumatoid arthritis: results of an independent Japanese population and evidence from meta-analysis of east Asian studies. J Hum Genet 53:163–173
Orozco G, Sanchez E, Gonzalez-Gay MA et al (2006) SLC22A4, RUNX1, and SUMO4 polymorphisms are not associated with rheumatoid arthritis: a case-control study in a Spanish population. J Rheumatol 33:1235–1239
Martinez A, Valdivia A, Pascual-Salcedo D et al (2006) Role of SLC22A4, SLC22A5, and RUNX1 genes in rheumatoid arthritis. J Rheumatol 33:842–846
Kuwahara M, Ikari K, Momohara S et al (2005) Failure to confirm association between SLC22A4 polymorphism and rheumatoid arthritis in a Japanese population. Arthritis Rheum 52:2947–2948
Barton A, Eyre S, Bowes J et al (2005) Investigation of the SLC22A4 gene (associated with rheumatoid arthritis in a Japanese population) in a United Kingdom population of rheumatoid arthritis patients. Arthritis Rheum 52:752–758
Newman B, Wintle RF, Oene M van et al (2005) SLC22A4 polymorphisms implicated in rheumatoid arthritis and Crohn’s disease are not associated with rheumatoid arthritis in a Canadian Caucasian population. Arthritis Rheum 52:425–429
Wesoly J, Toes RE, Slagboom PE, Huizinga TW (2005) RUNX1 intronic SNP is not associated with rheumatoid arthritis susceptibility in Dutch Caucasians. Rheumatology (Oxford) 44:1196
Plenge RM, Padyukov L, Remmers EF et al (2005) Replication of putative candidate-gene associations with rheumatoid arthritis in > 4,000 samples from North America and Sweden: association of susceptibility with PTPN22, CTLA4, and PADI4. Am J Hum Genet 77:1044–1060
Lee YH, Rho YH, Choi SJ et al (2007) PADI4 polymorphisms and rheumatoid arthritis susceptibility: a meta-analysis. Rheumatol Int 27:827–833
Lee YH, Rho YH, Choi SJ et al (2007) The PTPN22 C1858T functional polymorphism and autoimmune diseases—a meta-analysis. Rheumatology (Oxford) 46:49–56
Lee YH, Kim JH, Song GG (2014) Chemokine receptor 5 Delta32 polymorphism and systemic lupus erythematosus, vasculitis, and primary Sjogren’s syndrome: meta-analysis of possible associations. Z Rheumatol (in press)
Yamada R, Tokuhiro S, Chang X, Yamamoto K (2004) SLC22A4 and RUNX1: identification of RA susceptible genes. J Mol Med (Berl) 82:558–564
Chatzikyriakidou A, Voulgari PV, Lambropoulos A, Drosos AA (2013) Genetics in rheumatoid arthritis beyond HLA genes: what meta-analyses have shown? Semin Arthritis Rheum 43:29–38
Gu E, Lu J, Xing D et al (2014) Rs7574865 polymorphism in signal transducers and activators of transcription 4 gene and rheumatoid arthritis: an updated meta-analysis of 28 case-control comparisons. Int J Rheum Dis. doi:10.1111/1756-185X.12363 (in press)
Acknowledgments
This study was supported in part by a grant of the Korea Healthcare Technology R&D Project, Ministry for Health & Welfare, Republic of Korea (HI12C1834).
Compliance with ethical guidelines
Conflict of interest. Y.H. Lee, S.-C. Bae, J.-H. Kim, Y.H. Seo, S.J. Choi, J.D. Ji, and G.G. Song state that there are no conflicts of interest. The accompanying manuscript does not include studies on humans or animals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, Y., Bae, SC., Kim, JH. et al. Meta-analysis of SLC22A4 and RUNX1 polymorphisms. Z Rheumatol 74, 351–358 (2015). https://doi.org/10.1007/s00393-014-1447-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00393-014-1447-3