Abstract
Background
Inflammatory plays a key role in the development of coronary artery disease (CAD). Colchicine as an anti-inflammatory treatment for CAD has attracted much attention, its efficacy and safety are controversial and deserved further exploration.
Methods and results
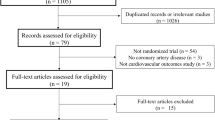
To evaluate the efficacy and safety of colchicine for patients with CAD, relevant randomized controlled trials (RCTs) were identified by searching several databases including PubMed, Web of Science, and EMBASE from January 1992 to May 2022. Fourteen eligible trials of colchicine therapy include populations with chronic coronary syndrome (CCS) (N = 2), acute coronary syndrome (ACS) (N = 5), and percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG) (N = 7), and involve a total of 13,235 patients which include 6654 subjects in colchicine group and 6581 subjects in the respective control arms. The outcome was reported as odds ratio (OR) and 95% confidence interval (CI), as the relative measure of association. Overall, the incidences of major adverse cardiovascular events (MACEs) (OR 0.65; 95% CI 0.54–0.77, p < 0.01), new ACS (OR 0.68; 95% CI 0.57–0.81, p < 0.01), coronary revascularization (OR 0.65; 95% CI 0.53–0.78, p < 0.01), and stroke (OR 0.51; 95% CI 0.32–0.82, p < 0.01), were lower in the colchicine group than in the placebo arm. We did not find a significant reduction in the incidence of atrial fibrillation (OR 0.84; 95% CI 0.68–1.04, p = 0.11), all-cause mortality (OR 1.06; 95% CI 0.83–1.35, p = 0.83), cardiovascular mortality (OR 0.77; 95% CI 0.52–1.15, p = 0.21). However, we found that colchicine did increase non-cardiovascular mortality (OR 1.44; 95% CI 1.04–2.01, p = 0.03). Although the incidence of gastrointestinal events in the colchicine treatment group was higher than that in the placebo arms (OR 2.08; 95% CI 1.39–3.12, p < 0.01), the symptoms disappeared rapidly after drug withdrawal and could be tolerated by most patients. Colchicine did not increase the incidence of infections (OR 1.42; 95% CI 0.82–2.46, p = 0.22), pneumonia (OR 1.55; 95% CI 0.58–4.18, p = 0.39), cancers (OR 0.98; 95% CI 0.79–1.22, p = 0.88), bleeding (OR 1.14; 95% CI 0.41–3.14, p = 0.80).
Conclusions
Colchicine is an effective, relatively safe drug that could be considered for the treatment of CAD. However, we need to pay attention to the increasing occurrence of non-cardiovascular mortality and infection especially pneumonia possibly caused by colchicine.
Graphical abstract
Efficacy and safety of colchicine for patients with CAD. CAD coronary artery disease; RCTs randomized controlled trials; OR odds ratio; MACEs major adverse cardiovascular events; ACS acute coronary syndrome; NNT number needed to treat; NNH number needed to harm








Similar content being viewed by others
Data availability
The present study is a meta-analysis of published randomized trials. All data used for analyses are presented in the manuscript.
References
Libby P, Theroux P (2005) Pathophysiology of coronary artery disease. Circulation 111:3481–3488
Knuuti J, Wijns W, Saraste A, Capodanno D, Barbato E, Funck-Brentano C et al (2020) ESC guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J 41:407–477
Collet JP, Thiele H, Barbato E, Barthelemy O, Bauersachs J, Bhatt DL et al (2021) 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Rev Esp Cardiol (Engl Ed) 74:544
O’Keefe JH, McCallister BD, Bateman TM, Kuhnlein DL, Ligon RW, Hartzler GO (1992) Ineffectiveness of colchicine for the prevention of restenosis after coronary angioplasty. J Am Coll Cardiol 19:1597–1600
Ridker PM, Libby P, MacFadyen JG, Thuren T, Ballantyne C, Fonseca F et al (2018) Modulation of the interleukin-6 signalling pathway and incidence rates of atherosclerotic events and all-cause mortality: analyses from the Canakinumab Anti-Inflammatory Thrombosis Outcomes Study (CANTOS). Eur Heart J 39:3499–3507
Ridker PM, Everett BM, Pradhan A, MacFadyen JG, Solomon DH, Zaharris E et al (2019) Low-dose methotrexate for the prevention of atherosclerotic events. N Engl J Med 380:752–762
Qaseem A, Harris RP, Forciea MA, Clinical Guidelines Committee of the American College of P, Denberg TD, Barry MJ et al (2017) Management of acute and recurrent gout: a clinical practice guideline from the American College of Physicians. Ann Intern Med 166:58–68
Ozen S, Demirkaya E, Erer B, Livneh A, Ben-Chetrit E, Giancane G et al (2016) EULAR recommendations for the management of familial Mediterranean fever. Ann Rheum Dis 75:644–651
Imazio M, Gaita F, LeWinter M (2015) Evaluation and treatment of pericarditis: a systematic review. JAMA 314:1498–1506
Chaldakov GN (1982) Antitubulins—a new therapeutic approach for atherosclerosis? Atherocslerosis 44:385–390
Chaldakov GN (2017) Colchicine, inflammation and fibrosis in cardiovascular disease—merging three classical tales. Biomed Rev 28:110–115
Tun B, Frishman WH (2018) Effects of anti-inflammatory medications in patients with coronary artery disease: a focus on losmapimod. Cardiol Rev 26:152–156
Martinez GJ, Celermajer DS, Patel S (2018) The NLRP3 inflammasome and the emerging role of colchicine to inhibit atherosclerosis-associated inflammation. Atherosclerosis 269:262–271
Cronstein BN, Molad Y, Reibman J, Balakhane E, Levin RI, Weissmann G (1995) Colchicine alters the quantitative and qualitative display of selectins on endothelial cells and neutrophils. J Clin Invest 96:994–1002
Leung YY, Yao Hui LL, Kraus VB (2015) Colchicine—update on mechanisms of action and therapeutic uses. Semin Arthritis Rheum 45:341–350
Shah B, Pillinger M, Zhong H, Cronstein B, Xia Y, Lorin JD et al (2020) Effects of acute colchicine administration prior to percutaneous coronary intervention: COLCHICINE-PCI randomized trial. Circ Cardiovasc Interv 13:e008717
Nidorf SM, Eikelboom JW, Budgeon CA, Thompson PL (2013) Low-dose colchicine for secondary prevention of cardiovascular disease. J Am Coll Cardiol 61:404–410
Tardif JC, Kouz S, Waters DD, Bertrand OF, Diaz R, Maggioni AP et al (2019) Efficacy and safety of low-dose colchicine after myocardial infarction. N Engl J Med 381:2497–2505
Nidorf SM, Fiolet ATL, Mosterd A, Eikelboom JW, Schut A, Opstal TSJ et al (2020) Colchicine in patients with chronic coronary disease. N Engl J Med 383:1838–1847
Tong DC, Quinn S, Nasis A, Hiew C, Roberts-Thomson P, Adams H et al (2020) Colchicine in patients with acute coronary syndrome: the Australian COPS randomized clinical trial. Circulation 142:1890–1900
Galli M, Princi G, Crea F, D’Amario D (2021) Colchicine and risk of non-cardiovascular death in patients with coronary artery disease: a pooled analysis underling possible safety concerns. Eur Heart J Cardiovasc Pharmacother 7:e18–e19
Deftereos S, Giannopoulos G, Angelidis C, Alexopoulos N, Filippatos G, Papoutsidakis N et al (2015) Anti-inflammatory treatment with colchicine in acute myocardial infarction: a pilot study. Circulation 132:1395–1403
Silvis MJM, Fiolet ATL, Opstal TSJ, Dekker M, Suquilanda D, Zivkovic M et al (2021) Colchicine reduces extracellular vesicle NLRP3 inflammasome protein levels in chronic coronary disease: A LoDoCo2 biomarker substudy. Atherosclerosis 334:93–100
Vaidya K, Arnott C, Martinez GJ, Ng B, McCormack S, Sullivan DR et al (2018) Colchicine therapy and plaque stabilization in patients with acute coronary syndrome: a CT coronary angiography study. JACC Cardiovasc Imaging 11:305–316
Hennessy T, Soh L, Bowman M, Kurup R, Schultz C, Patel S et al (2019) The Low dose colchicine after myocardial infarction (LoDoCo-MI) study: a pilot randomized placebo controlled trial of colchicine following acute myocardial infarction. Am Heart J 215:62–69
Kajikawa M, Higashi Y, Tomiyama H, Maruhashi T, Kurisu S, Kihara Y et al (2019) Effect of short-term colchicine treatment on endothelial function in patients with coronary artery disease. Int J Cardiol 281:35–39
Tsai TL, Wei JC, Wu YT, Ku YH, Lu KL, Wang YH et al (2019) The association between usage of colchicine and pneumonia: a nationwide, population-based cohort study. Front Pharmacol 10:908
Chia EW, Grainger R, Harper JL (2008) Colchicine suppresses neutrophil superoxide production in a murine model of gouty arthritis: a rationale for use of low-dose colchicine. Br J Pharmacol 153:1288–1295
Hiroshi Asako PK, Baethge BA, Wolf RE, Neil Granger D (1992) Colchicine and methotrexate reduce leukocyte adherence and emigration in rat mesenteric venules. Inflammation 6:45–56
Torres A, Cilloniz C, Niederman MS, Menendez R, Chalmers JD, Wunderink RG et al (2021) Pneumonia. Nat Rev Dis Primers 7:25
Bacmeister L, Schwarzl M, Warnke S, Stoffers B, Blankenberg S, Westermann D et al (2019) Inflammation and fibrosis in murine models of heart failure. Basic Res Cardiol 114:19
Shen S, Duan J, Hu J, Qi Y, Kang L, Wang K et al (2022) Colchicine alleviates inflammation and improves diastolic dysfunction in heart failure rats with preserved ejection fraction. Eur J Pharmacol 929:175126
Ujihara Y, Kanagawa M, Mohri S, Takatsu S, Kobayashi K, Toda T et al (2019) Elimination of fukutin reveals cellular and molecular pathomechanisms in muscular dystrophy-associated heart failure. Nat Commun 10:5754
Zhang C, Chen B, Guo A, Zhu Y, Miller JD, Gao S et al (2014) Microtubule-mediated defects in junctophilin-2 trafficking contribute to myocyte transverse-tubule remodeling and Ca2+ handling dysfunction in heart failure. Circulation 129:1742–1750
Roth ME, Chinn ME, Dunn SP, Bilchick KC, Mazimba S (2022) Association of colchicine use for acute gout with clinical outcomes in acute decompensated heart failure. Clin Cardiol 45:733–741
Deftereos S, Giannopoulos G, Panagopoulou V, Bouras G, Raisakis K, Kossyvakis C et al (2014) Anti-inflammatory treatment with colchicine in stable chronic heart failure: a prospective, randomized study. JACC Heart Fail 2:131–137
Funding
This study was supported by the Liaoning Province Central Guiding Local Science and Technology Foundation (No. 2019JH6/10400005) and the National Natural Science Foundation of China (No. 8217021483).
Author information
Authors and Affiliations
Contributions
All authors listed have made a substantial, direct, and intellectual contribution to the work, and approved it for publication.
Corresponding author
Ethics declarations
Conflict of interest
All the authors have no conflicts of interest to declare.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, T., Liu, G. & Yu, B. A meta-analysis evaluating efficacy and safety of colchicine for prevention of major cardiovascular events in patients with coronary artery disease. Clin Res Cardiol 112, 1487–1505 (2023). https://doi.org/10.1007/s00392-023-02254-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-023-02254-9