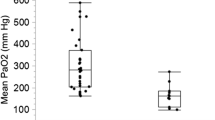
Abstract
Background
Hemodynamic response to successful extracorporeal cardiopulmonary resuscitation (eCPR) is not uniform. Pulse pressure (PP) as a correlate for myocardial damage or recovery from it, might be a valuable tool to estimate the outcome of these patients.
Methods
We report retrospective data of a single-centre registry of eCPR patients, treated at the Interdisciplinary Medical Intensive Care Unit at the Medical Centre, University of Freiburg, Germany, between 01/2017 and 01/2020. The association between PP of the first 10 days after eCPR and hospital survival was investigated. Moreover, patients were divided into three groups according to their PP [low (0–9 mmHg), mid (10–29 mmHg) and high (≥ 30 mmHg)] at each time point.
Results
One hundred forty-three patients (age 63 years, 74.1% male, 40% OHCA, average low flow time 49 min) were analysed. Overall hospital survival rate was 28%. A low PP both early after eCPR (after 1, 3, 6 and 12 h) and after day 1 to day 8 was associated with reduced hospital survival. At each time point (1 h to day 5) the classification of patients into a low, mid and high PP group was able to categorize the patients for a low (5–20%), moderate (20–40%) and high (50–70%) survival rate. A multivariable analysis showed that the mean PP of the first 24 h was an independent predictor for survival (p = 0.008).
Conclusion
In this analysis, PP occurred to be a valuable parameter to estimate survival and maybe support clinical decision making in the further course of patients after eCPR.
Graphic abstract

Similar content being viewed by others
Background
More than 500,000 patients per year suffer cardiac arrest in the US [1] and approximately 275,000 out of hospital cardiac arrests (OHCA) per year are reported for Europe [2]. In case of a refractory cardiac arrest, veno-arterial extracorporeal membrane oxygenation (VA ECMO) support can be considered as a rescue therapy in these patients [3, 4], referred as extracorporeal cardiopulmonary resuscitation (eCPR) [5, 6]. Both, the increasing availability of VA ECMO systems and the encouraging results of previous studies lead to increasing eCPR numbers [7, 8].
Chances of survival are higher in eCPR patients (28–38%) [4, 9,10,11,12] than in patients treated with conventional cardiopulmonary resuscitation (CPR) (11–14%) [13, 14]. However, mortality remains high in these very sick patients. Although eCPR represents a promising approach for patients with cardiac arrest, this highly invasive, costly and complex therapy raises many new medical and ethical questions in which effective prognostic assessment plays an important role. So far, superior survival has been demonstrated for an initially defibrillatable heart rhythm, a shorter low flow time as well as a higher pH and a lower serum lactate level [15, 16].
Multiple scores have been established to estimate the possibility of survival after eCPR. Most of these scores rely on parameters from a time point before ECMO initiation [17] and do include the above mentioned parameters.
VA ECMO therapy aims on delivering adequate organ perfusion preventing worsening the post resuscitation syndrome and progression to multi-organ failure. Several biomarkers (e.g. lactate, pH and standard bicarbonate concentration) reflecting hemodynamic stabilization have been shown to prognosticate survival early on the treatment course [18].
Another evident marker of changes in hemodynamic state is the pulse pressure (PP), reflecting the myocardial function and the level of afterload [19]. In several non-ECMO studies the level of PP was investigated with regards to patient’s outcome, obtaining very different results. For instance, a high PP was associated with lower survival rates in patients with cardiogenic shock in case of an acute coronary syndrome and after myocardial infarction as well as after coronary artery bypasses graft surgery [20,21,22]. On the other hand, in patients with decompensated heart failure a low PP was a strong predictor for reduced survival [23].
Although it has been shown that PP is able to predict cardiac output in VA ECMO patients [24], so far there is little evidence on the prognostic value of PP in eCPR patients, especially while ongoing VA ECMO support. Therefore, this retrospective study investigated the influence of PP on the successful ECMO weaning and hospital survival rate within the first 10 days after eCPR.
Methods
We report retrospective data of a single-centre registry of patients treated with eCPR. ECPR was defined as VA ECMO implantation during continuous resuscitation or within the first 20 min after return of spontaneous circulation (ROSC) with persistent hemodynamic instability [5]. Decision criteria that support or oppose the use of eCPR in the individual case and the further diagnostic and therapeutic strategy immediately after VA ECMO cannulation were performed in accordance with the German eCPR recommendations [6].
All patients treated at the Interdisciplinary Medical Intensive Care Unit at the Medical Centre, University of Freiburg, Germany, between January 2017 and January 2020 were analysed. Patient identity data derived from the registry were blinded and the study plan was approved by the local ethics committee (EK-Freiburg 151/14). The need for informed consent of the subjects was waived by the local ethics committee.
PP as well as all other analysed parameters were obtained from the medical patient records. Because of diagnostic transfers after VA ECMO implantation (CT scans or coronary angiography) immediately after eCPR, some PP values are missing (additional file 1, Table E7). Patients with missing values at a certain time point were not considered in the survival analysis. For visualization, the course of patients over time in relation to their corresponding pulse pressure PP was extrapolated using the next time-wise available value. After investigating the association between PP and hospital survival, patients were divided into three groups according to their median PP [low (0–9 mmHg), mid (10–29 mmHg) and high (≥ 30 mmHg)].
The primary endpoint was hospital survival. Moreover, successful VA ECMO weaning, 30 day survival and intensive care unit (ICU) survival were analysed. Successful ECMO weaning was defined as free from VA ECMO and alive for at least 48 h after decannulation. Unsuccessful weaning was defined as the inability to explant the ECMO device because of persistent cardiac failure or death during VA ECMO support or the need for re-cannulation within 48 h. To investigate pulse pressure independently from death due to anoxic brain injury, we also performed a survival analysis excluding these patients.
To compare the patient’s disease severity, the SAPS II score [25] at ICU admission was analysed.
Since, given the same level of diastolic blood pressure, there is collinearity for mean and systolic blood pressure as well as PP, only PP was considered in the multivariable analysis.
Study population
In this study, only patients with eCPR (VA ECMO cannulation in case of cardiac arrest or instable ROSC) were investigated. Patients with VA ECMO cannulation in case of cardiogenic shock were excluded. All patients admitted to our ICU after eCPR were included in this study. At our institution, patients with in hospital cardiac arrest (IHCA) without ROSC after 15 min were routinely accessed for extracorporeal resuscitation. By local standard, the presence of an unwitnessed cardiac arrest, prolonged duration of CPR without signs of life (breathing, swallowing etc.), a non-shockable initial rhythm and advanced age were considered relative contraindications for VA ECMO cannulation. Final decision to cannulate, however, was driven by a team decision at the bedside including at least two physicians, a perfusionist and two nurses. For OHCA, emergency medical services personal was encouraged to transport patients without ROSC with ongoing chest compressions to the hospital. Since 09/2018 patients with OHCA without ROSC were evaluated for prehospital eCPR. The same relative contra indications applied as for IHCA patients. Cannulation was performed immediately on-site. All patients with evidence of a cardiac cause of the cardiac arrest were primarily assessed by coronary angiography after eCPR and then received a CT scan. Patients with suggestive symptoms or findings of an extra-cardiac cause received a CT scan first. In this case coronary angiography was performed if the CT scan had not provided a sufficient explanation for the cardiac arrest.
ECMO centre and ECMO management
Our institution features a 24/7 ECMO-centre localized within a tertiary hospital with a 30-bed medical intensive care unit. Cannulations in our ECMO centre are performed by two experienced intensivist and a perfusionist in Seldinger’s technique without primary surgical cut down. All members of the ECMO team can be gathered within 30 min. Typical numbers for veno-arterial and veno-venous cannulation are 65 and 35 per year, respectively. There is a 24 h/7 days outreach team.
Either SCPC (Sorin centrifugal pump console, Livanova, London, United Kingdom) or Cardiohelp systems (Maquet Getinge Group, Rastatt, Germany) were used. Typical venous (draining) cannulas were 21–23 Fr (French = Charrière) in diameter and 55 cm of length while arterial (returning) cannulas were 15–17 Fr and 15–23 cm (both HLS cannula, Maquet Getinge Group, Rastatt, Germany). Since January 2019 decannulation is performed under fluoroscopic control using the MANTA Vascular Closure Device® [26], whenever possible.
For patients without life threatening bleeding, anticoagulation was provided by intravenous unfractionated heparin aiming at a partial thromboplastin time 1.5 times upper normal limit.
The management of vasopressors and fluid therapy was driven by clinical judgement of the ECMO experienced intensivist in charge and has been reported earlier [27, 28]. Within the first 48 h of ECMO support blood flow was kept the highest possible flow without the need for excessive volume substation. All patients received invasive blood pressure monitoring via the right radial artery and cerebral NIRS monitoring.
Moreover, within the first 48 h of ECMO support no inotropes were applied if there was detectable pulse pressure. Only in case of severe pulmonary edema or if echocardiography showed no opening of the aortic valve inotropes therapy was started. In particularly severe cases, in which despite these measures, severe pulmonary edema or insufficient opening of the aortic valve persisted, mechanical left heart decompression using the Impella® device was performed. The rationale of this algorithm is to avoid additional mechanical support to minimise additional complications. In one case intra-aortic balloon pump (IABP) was placed in addition to the VA ECMO therapy.
Treatment algorithms and standard operating procedures were subject to optimizations during the observational period, reflecting current state of the art recommendations and scientific knowledge.
Statistical analysis
Continuous variables are presented as median and interquartile range (IQR) and categorical variables as numbers and percentages. Mann–Whitney U test was used for the analysis of continuous variables, Pearson’s Chi-squared test or Fisher’s exact test for categorical variables. Kruskal–Wallis test was used for the analysis of continuous variables in more than two groups. Multivariable regression analysis was performed for univariate (dependent) predictors of hospital survival (threshold of p < 0.05). Results are given as odds ratio [(OR), 95% confidence interval (CI)]. 60-day survival was calculated using the Kaplan–Meier method. Analyses were exploratory in nature. As a result, there was no prespecified plan to adjust for multiple comparisons and inferences drawn from 95% confidence intervals or p values may not be reproducible. Statistical calculations were performed using IBM SPSS statistics 25.0 (Armonk, NY: IBM Corp, 2017). Survival analysis was conducted in R 32 and figures were produced using the package ggplot2 33.
Results
Patients
Two hundred thirty-two patients were treated with VA ECMO from 01/2017 to 01/2020. After exclusion of 89 patients treated for cardiogenic shock, 143 patients with eCPR (age 63 years, 74.1% male) could be analysed (Fig. 1). VA ECMO cannulation was performed in 135 patients while CPR and in 8 patients with unstable ROSC.
OHCA occurred in 39.9% of the patients and in 60.1% cardiac arrest had a coronary genesis (Table 1). Median no flow and low flow time were 0 min and 49 min, respectively. In 12 (8.4%) patients, eCPR was performed prehospital and 42% of all patients showed a shockable rhythm before VA ECMO implantation (additional file 1, table E1). SAPS II at ICU admission was 56.
Overall successful VA ECMO weaning and hospital survival rate were 35.7% and 28%, respectively (Table 2). Six (4.2%) patients received additional Impella® support and one (0.7%) patient additional IABP support.
Pulse pressure analysis
Throughout from 1 h to day 8 after eCPR, a low pulse pressure correlated with reduced hospital survival (additional file 1, table E2). A similar association was observed between low PP and reduced VA ECMO weaning rate (additional file 1, table E3). There was no difference in measured PP pre VA ECMO implantation (while CPR) in survivors and non-survivors (only applicable for patients with unstable ROSC).
The classification of patients into a low (0–9 mmHg), mid (10–29 mmHg) and high (≥ 30 mmHg) PP group showed a good discrimination between low, medium and high survival rates at each time point from 1 h until day 5 after eCPR (Table 3). The same association was shown in the subgroup of patients with ongoing VA ECMO only at each time point (additional file 1, table E4).
Although low or high PP at each single time point showed a strong association to increased mortality or survival rates, many patients switched between the different PP allocations in the first 24 h (Fig. 2). The Sankey chart visualizes the course of patients over the time in relation to their corresponding pulse pressure. Particularly, a large proportion of patients had alternating low or mid PP in the first 24 h. At each time point a high number of patients presenting with a low PP were dying, while mortality of patients with a high PP was very low. All over, the proportion of deceased patients and patients with a high PP was constantly increasing while the proportion of patients with a low or mid PP was decreasing.
Analysis of mean 24 h pulse pressure
There were no differences between the low, mid and high PP groups in sex and BMI, but patients with a low PP were younger (61 vs. 65 and 65 years, p = 0.040, Table 1). Moreover, patients with a low PP showed a higher rate of OHCA (55% vs. 35% and 25%, p = 0.013) compared to patients with a mid and high PP. The underlying cause of cardiac arrest was similar in all three groups, with 50–60% of patients showing a coronary cause. Low PP patients showed higher creatine kinase and creatine kinase-MB levels than patients with mid and high PP (p = 0.015 and p = 0.018).
There were no differences in the no flow time between the three groups, but patients with a low PP showed a numerically but not statistically significant higher low flow time (55 min vs. 44 min and 43 min). Moreover, SAPS II at hospital admission was higher in the low PP group. Interestingly, the rate of underlying coronary artery disease was higher in the mid and high PP group (p = 0.039). Furthermore, there was a trend for a higher rate of chronic renal failure in patients with a high PP (p = 0.052).
Successful VA ECMO weaning rate was higher for patients with a high PP (61%) than for patients with mid (39%) and low PP (15%, p < 0.001, Table 2). Hospital survival rates for patients with a mean low, mid and high PP in the first 24 h after eCPR were 5.7%, 31.5% and 55.6%, respectively (p < 0.001, Fig. 3). After exclusion of patients died due to anoxic brain injury or withdrawal of care in those circumstances, survival rates were 7.9%, 42.5% and 71.4%, respectively (p < 0.001). Survival rates of PP groups in patients without additional mechanical circulatory support (Impella® or IABP) were 6.4%, 31.4% and 55.6%, respectively (p < 0.001).
Analysis of pulse pressure as an independent predictor for survival
Univariate analysis showed a strong association of high mean PP of the first 24 h, high mean and systolic arterial pressure as well as a higher level of pH and standard bicarbonate, a lower level of potassium, glucose and lactate 24 h after eCPR with increased survival (additional file 1, table E5). Interestingly, diastolic arterial pressure was not associated with survival.
Multivariable analysis revealed mean PP of the first 24 h after eCPR [OR 3.2 (1.3–7.4), p = 0.008] and lactate [OR 0.6 (0.4–0.8), p = 0.003] as independent predictor for survival (Table 4).
Discussion
To our best knowledge, this is the first comprehensive analysis of PP and its meaning for the clinical course of eCPR patients. In this analysis low PP after eCPR was strongly associated with a reduced survival rate. The level of mean PP of the first 24 h corresponded to a survival rate of 6%, 32% and 56%, respectively. The discrimination between these three groups was present from the first hours after eCPR and lasted up to day 5.
The patients in this study showed similar baseline characteristics compared to previous reports from eCPR cohorts. This is especially true for important prognostic factors like low flow time, shockable heart rhythm and coronary genesis of cardiac arrest [10, 11, 15]. In addition, this analysis included patients with IHCA, OHCA and pre hospital VA ECMO cannulation, which covers the entire spectrum of eCPR applications. Furthermore, this cohort showed a rather high survival and VA ECMO weaning rate.
The allocation into a low (0–9 mmHg), mid (10–29 mmHg) and high (≥ 30 mmHg) PP group for each time point was based on clinical considerations and resulted in three risk groups. Patients with a low PP showed a survival rate of approx. 5–20% for each time point and patients with mid and high PP showed a survival rate of approx. 20–40% and 50–70%, respectively. Thus, not only an association between a very low PP and a high mortality could be shown, but also an association between a high PP and an above the average high survival rate compared to the overall cohort and other eCPR reports (30–40% survival rate) [4, 9,10,11]. In a subgroup analysis excluding patients who died of anoxic brain injury, these group differences were even more pronounced, suggesting a possible influence of PP on the physiology and organ function in these patients.
To extend the isolated observation of PP at each single time point, an analysis of the course of PP of all patients within the first days after eCPR was performed. The Sankey chart analysis reveals that patients do not remain permanently in a PP group, but change between the groups to a relevant extent, especially within the first 24 h. As the survival analysis of the three PP groups at each time point suggests, the rate of patients dying is highest in the low PP group. Furthermore, most of the changes between the groups were completed within the first 4 days, so that afterwards most of the patients already died or showed a recovery of the PP and thus changed into the high PP group.
Both from a clinical point of view and due to the high proportion of patients switching between PP groups within the first 24 h, a detailed analysis of the mean PP of the first 24 h was performed to examine the three PP groups with regard to their baseline characteristics and outcome.
There were several factors that were associated with a low PP level. First of all, patients with a low PP had a higher rate of OHCA compared to patients with mid and high PP. Corresponding to a higher rate of OHCA, the low PP group also showed an at least numerically higher low flow time (10 min longer than patients with mid and high PP). Moreover, patients with a low PP showed higher levels of creatine kinase and creatine kinase—MB. Therefore, the distribution of the duration of low flow time and creatine kinase might be appropriate to explain the level of PP as an indirect parameter for myocardial damage. On the other hand, PP is also determined by the level of arterial afterload. Therefore, it is important to note, that there were neither differences in the applied dose of the inotropic nor the vasoconstrictive therapy between the three PP groups.
In a few patients (6, 4.2%) with no opening of the aortic valve after inotropic stimulation or in case of severe pulmonary edema, the left heart decompression device Impella® or IABP were used additionally to the VA ECMO therapy. Even though there was no significant difference of left ventricular venting between the groups, this additional support may have led to a further reduced PP. These patients certainly represent a special cohort with an increased risk of mortality, but it seems reasonable to include these patients in this study as well, as it corresponds to real life experience and therapy. Moreover, even after excluding these patients from the analysis, the same association between PP and survival was observed.
Interestingly, there were no differences in no flow time, cause of cardiac arrest or underlying heart rhythm between the three groups. And paradoxically, there was a significantly higher rate of underlying coronary artery disease and a trend for a higher rate of chronic renal failure in patients from the high PP group, which might be explained by the higher rate of IHCA, as the patients were already hospitalised with corresponding diagnoses.
Beyond the extent of a strong association between the PP level and patient outcomes, the mean 24 h PP appeared as an independent predictor for survival besides the lactate level in a multivariable analysis of parameters available at bedside (vital parameters and blood gas analysis).
The results of this work thus represent a continuation of the findings of previous studies that examined the PP at the time before, during or immediately after VA ECMO cannulation.
Previously, it could be shown that PP before VA ECMO cannulation is an important prognostic parameter in patients with a cardiogenic shock. The survival after veno-arterial ECMO (SAVE) score includes a PP of ≤ 20 mmHg as a factor for increased in mortality [17]. Appropriately, the ECPR score by Park et al. included an initial PP immediately after VA ECMO cannulation of ≥ 24 mmHg as a factor for a higher survival rate [29]. These results are confirmed by a study of Ryu et al. in which an initial pulse pressure after eCPR of less than 25 mmHg was associated with inferior neurological outcome [30].
The only study that investigated the influence of the PP in the further course after eCPR, examined the period of the first 6 h after VA ECMO cannulation. In this mixed cohort (cardiogenic shock, respiratory failure and septic shock) of 69 patients, a high mean PP (≥ 30 mmHg) was associated with a superior survival and VA ECMO weaning rate [31].
The presented results show for the first time that the prognostic value of the PP is not only present at the time immediately before and after eCPR, but also at any other time within the next 5 days. In summary, we believe that the PP after eCPR, a parameter that is easy to assess at any time, has a high predictive value for survival. Although the PP should not be considered on its own and the shown results should be confirmed in larger trials, it certainly may help in clinical decisions making.
Limitations
This is a retrospective observational study and, therefore, contains the risk of selection and reporting bias.
Obviously, PP is not only driven by the patient’s capability to produce a PP but depends on medical or device treatment. In this analysis inotropic support was similar between the groups and only very few venting devices were used. For that, these confounders should be addressed adequately. However, the present observations can be transferred only to eCPR cohorts with similar treatment regimes.
Therefore, despite of using multivariable analysis to prove PP as an independent predictor for survival, there still might be remaining confounders. Moreover, this is a single-centre report and specific processes may influence the presented results. Together, due to these limitations, our findings about the PP after eCPR should be considered in the overall context of the patient's disease severity and should not prompt therapy limitations on its own.
Furthermore, for the results of the first 12 h it must be taken into account that due to patient transports and interventions some PP values were missing.
Conclusion
In this analysis, PP after eCPR was found to be a strong and independent predictor for survival, which could potentially be a useful parameter in daily clinical decision making.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- APACHE II score:
-
Acute physiology and chronic health evaluation
- BMI:
-
Body mass index
- BW:
-
Body weight
- CPR:
-
Cardiopulmonary resuscitation
- ECMO:
-
Extracorporeal membrane oxygenation
- eCPR:
-
Extracorporeal cardiopulmonary resuscitation
- IABP:
-
Intra-aortic balloon pump
- IHCA:
-
In hospital cardiac arrest
- ICU:
-
Intensive care unit
- OHCA:
-
Out of hospital cardiac arrest
- PP:
-
Pulse pressure
- PEA:
-
Pulseless electrical activity
- ROSC:
-
Return of spontaneous circulation
- SAPS II score:
-
Simplified Acute Physiology Score
- UHF:
-
University Hospital Freiburg
- VA:
-
Veno-arterial
References
Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Blaha MJ et al (2014) Executive summary: heart disease and stroke statistics-2014 update: a report from the American Heart Association. Circulation 129:399–410
Atwood C, Eisenberg MS, Herlitz J, Rea TD (2005) Incidence of EMS-treated out-of-hospital cardiac arrest in Europe. Resuscitation 67:75–80
Soar J, Nolan JP, Bottiger BW, Perkins GD, Lott C, Carli P et al (2015) European resuscitation council guidelines for resuscitation 2015: section 3. Adult advanced life support. Resuscitation 95:100–147
Kim SJ, Kim HJ, Lee HY, Ahn HS, Lee SW (2016) Comparing extracorporeal cardiopulmonary resuscitation with conventional cardiopulmonary resuscitation: a meta-analysis. Resuscitation 103:106–116
Jacobs I, Nadkarni V, Bahr J, Berg RA, Billi JE, Bossaert L et al (2004) Cardiac arrest and cardiopulmonary resuscitation outcome reports: update and simplification of the Utstein templates for resuscitation registries: a statement for healthcare professionals from a task force of the international liaison committee on resuscitation (American Heart Association, European Resuscitation Council, Australian Resuscitation Council, New Zealand Resuscitation Council, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Councils of Southern Africa). Circulation 110:3385–3397
Michels G, Wengenmayer T, Hagl C, Dohmen C, Bottiger BW, Bauersachs J et al (2019) Recommendations for extracorporeal cardiopulmonary resuscitation (eCPR): consensus statement of DGIIN, DGK, DGTHG, DGfK, DGNI, DGAI, DIVI and GRC. Clin Res Cardiol. 108:455–464
Extracorporeal Life Support Organization (ELSO) (2020): International summary of ECLS registry report, January 2020. https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwi7oO3IttfrAhXBy6QKHRiNDOAQFjAGegQIAxAB&url=https%3A%2F%2Fwww.elso.org%2FPortals%2F0%2FFiles%2FReports%2F2020_January%2FInternational%2520Summary%2520January%25202020_page1.pdf&usg=AOvVaw2rWvOUPpcX92qGjjCH-8f8. Accessed 07 Sep 2020
Stretch R, Sauer CM, Yuh DD, Bonde P (2014) National trends in the utilization of short-term mechanical circulatory support: incidence, outcomes, and cost analysis. J Am CollCardiol 64:1407–1415
Shin TG, Choi J-H, Jo IJ, Sim MS, Song HG, Jeong YK et al (2011) Extracorporeal cardiopulmonary resuscitation in patients with inhospital cardiac arrest: a comparison with conventional cardiopulmonary resuscitation. Crit Care Med 39:1–7
Kuroki N, Abe D, Iwama T, Suzuki K, Sugiyama K, Akashi A et al (2017) Association between delay to coronary reperfusion and outcome in patients with acute coronary syndrome undergoing extracorporeal cardiopulmonary resuscitation. Resuscitation 114:1–6
Wang CH, Chou NK, Becker LB, Lin JW, Yu HY, Chi NH et al (2014) Improved outcome of extracorporeal cardiopulmonary resuscitation for out-of-hospital cardiac arrest—a comparison with that for extracorporeal rescue for in-hospital cardiac arrest. Resuscitation 85:1219–1224
Chen YS, Lin JW, Yu HY, Ko WJ, Jerng JS, Chang WT et al (2008) Cardiopulmonary resuscitation with assisted extracorporeal life-support versus conventional cardiopulmonary resuscitation in adults with in-hospital cardiac arrest: an observational study and propensity analysis. Lancet 372:554–561
Reynolds JC, Frisch A, Rittenberger JC, Callaway CW (2013) Duration of resuscitation efforts and functional outcome after out-of-hospital cardiac arrest: when should we change to novel therapies? Circulation 128:2488–2494
Ouweneel DM, Schotborgh JV, Limpens J, Sjauw KD, Engstrom AE, Lagrand WK et al (2016) Extracorporeal life support during cardiac arrest and cardiogenic shock: a systematic review and meta-analysis. Intensiv Care Med 42:1922–1934
Wengenmayer T, Rombach S, Ramshorn F, Biever P, Bode C, Duerschmied D et al (2017) Influence of low-flow time on survival after extracorporeal cardiopulmonary resuscitation (eCPR). Crit Care 21:157
Debaty G, Babaz V, Durand M, Gaide-Chevronnay L, Fournel E, Blancher M et al (2017) Prognostic factors for extracorporeal cardiopulmonary resuscitation recipients following out-of-hospital refractory cardiac arrest. a systematic review and meta-analysis. Resuscitation 112:1–10
Schmidt M, Burrell A, Roberts L, Bailey M, Sheldrake J, Rycus PT et al (2015) Predicting survival after ECMO for refractory cardiogenic shock: the survival after veno-arterial-ECMO (SAVE)-score. Eur Heart J 36:2246–2256
Wengenmayer T, Duerschmied D, Graf E, Chiabudini M, Benk C, Muhlschlegel S et al (2019) Development and validation of a prognostic model for survival in patients treated with venoarterial extracorporeal membrane oxygenation: the PREDICT VA-ECMO score. Eur Heart J Acute Cardiovasc Care 8:350–359
Stevenson LW, Perloff JK (1989) The limited reliability of physical signs for estimating hemodynamics in chronic heart failure. JAMA 261:884–888
Harbaoui B, Nanchen D, Lantelme P, Gencer B, Heg D, Klingenberg R et al (2018) Prognostic value of pulse pressure after an acute coronary syndrome. Atherosclerosis 277:219–226
Mitchell GF, Moye LA, Braunwald E, Rouleau JL, Bernstein V, Geltman EM et al (1997) Sphygmomanometrically determined pulse pressure is a powerful independent predictor of recurrent events after myocardial infarction in patients with impaired left ventricular function. SAVE Investigators. Survival and Ventricular Enlargement. Circulation 96:4254–4260
Nikolov NM, Fontes ML, White WD, Aronson S, Bar-Yosef S, Gaca JG et al (2010) Pulse pressure and long-term survival after coronary artery bypass graft surgery. AnesthAnalg 110:335–340
Aronson D, Burger AJ (2004) Relation between pulse pressure and survival in patients with decompensated heart failure. Am J Cardiol 93:785–788
Mourad M, Eliet J, Zeroual N, Saour M, Sentenac P, Manna F et al (2020) Pulse pressure and end-tidal carbon dioxide for monitoring low native cardiac output during veno-arterial ECLS: a prospective observational study. Crit Care 24:569
Le Gall JR, Lemeshow S, Saulnier F (1993) A new simplified acute physiology score (SAPS II) based on a European/North American Multicenter Study. JAMA 270:2957–2963
Bemtgen X, Heidt T, Zotzmann V, Rilinger J, Wengenmayer T, Biever PM et al (2020) Venoarterial extracorporeal membrane oxygenation decannulation using the novel Manta vascular closure device. Eur Heart J Acute Cardiovasc Care. https://doi.org/10.1177/2048872620918707
Staudacher DL, Gold W, Biever PM, Bode C, Wengenmayer T (2017) Early fluid resuscitation and volume therapy in venoarterial extracorporeal membrane oxygenation. J Crit Care 37:130–135
Zotzmann V, Rilinger J, Lang CN, Duerschmied D, Benk C, Bode C et al (2019) Early full-body computed tomography in patients after extracorporeal cardiopulmonary resuscitation (eCPR). Resuscitation. https://doi.org/10.1016/j.resuscitation.2019.11.024
Park SB, Yang JH, Park TK, Cho YH, Sung K, Chung CR et al (2014) Developing a risk prediction model for survival to discharge in cardiac arrest patients who undergo extracorporeal membrane oxygenation. Int J Cardiol 177:1031–1035
Ryu JA, Chung CR, Cho YH, Sung K, Jeon K, Suh GY et al (2019) Neurologic outcomes in patients who undergo extracorporeal cardiopulmonary resuscitation. Ann ThoracSurg 108:749–755
Park BW, Seo DC, Moon IK, Chung JW, Bang DW, Hyon MS et al (2013) Pulse pressure as a prognostic marker in patients receiving extracorporeal life support. Resuscitation 84:1404–1408
R Core Team (2014). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/.
Wickham H (2016). ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag New York. ISBN 978-3-319-24277-4
Funding
Open Access funding enabled and organized by Projekt DEAL.. None.
Author information
Authors and Affiliations
Contributions
JR and TW contributed to the conception of the study. JR, AR and TW contributed to data collection. JR, AR, XB, MJ, VZ, PMB, DD, KK, CB, DLS and TW contributed to data analysis and interpretation. JR and TW drafted the manuscript. AR, XB, MJ, VZ, PMB, DD, CB, GT, CB and DLS revised the manuscript for important intellectual content. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
The protocol was approved by our institution’s ethical committee (EK-Freiburg 151/14).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rilinger, J., Riefler, A.M., Bemtgen, X. et al. Impact of pulse pressure on clinical outcome in extracorporeal cardiopulmonary resuscitation (eCPR) patients. Clin Res Cardiol 110, 1473–1483 (2021). https://doi.org/10.1007/s00392-021-01838-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-021-01838-7