Summary
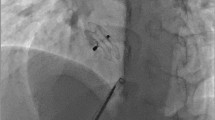
We report about our initial experience for the in 2005 modified Helex (Gore) device for closure of atrial septal defects (ASD) and persistent foramen ovale (PFO). Major changes were made at the delivery system for simplifying the Helex implantation procedure. We treated 11 patients, 8 children and 3 adults, with ages between 3 and 62 years. In 10 patients the diagnosis was a relevant ASD with volume overload of the right heart (Left to right shunts between 30 and 50%). One adult (age 58 years) have had a small left to right shunt with a PFO-like defect and the history of 2 neurologic embolic events. In 3 patients we found 2 defects. In all patients a Helex occluder was implanted successfully. The mean fluoroscopy time was 8,4 minutes. The immediate occlusion rate after 24 hours was 91%. In all cases there was a very good adaptation of the device to the anatomical structures. In this small series, the Helex occluder appears to offer a reliable system of occlusion for small and moderate ASDs and for PFO with minimal risk of major complications.
Similar content being viewed by others
References
Beitzke A, Schuchlenz H, Beitzke M, Gamillscheg A, Stein HI, Zartner P (2002) Interventional occlusion of foramen ovale and atrial septal defects after paradoxical embolism incidents. Z Kardiol 91(9):693–700
Billinger K et al (2006) Helex Septal Occluder for transcatheter closure of patent foramen ovale: multicenter experience. EuroInterv 1:465–471
Chan KC, Godman MJ, Walsh K, Wilson N, Redington A, Gibbs JL (1999) Transcatheter closure of atrial septal defect and interatrial communications with a new self expanding nitinol double disc device (Amplatzer septal occluder): multicentre UK experience. Heart 82(3):300–306
Gao W, Zhou AQ, Yu ZQ, Li F, Zhang YQ, Sun K, Zhong YM (2004) Transcatheter closure of secundum atrial septal defect in children. Zhonghua Er Ke Za Zhi 42(4):287–290
Ischinger TA, Kemkes B, Boosfeld C (2003) Partial malposition of PFO closure device: indication for elective surgical removal? Discussion of indications, procedural and anatomical aspects. Z Kardiol 92(2):188–192
Knirsch W, Dodge–Khatami A, Valsangiacomo– Buechel E, Weiss M, Berger F (2005) Challenges encountered during closure of atrial septal defects. Pediatr Cardiol 26(2):147–153
Krumsdorf U, Keppeler P, Horvath K, Zadan E, Schrader R, Sievert H (2001) Catheter closure of atrial septal defects and patent foramen ovale in patients with an atrial septal aneurysm using different devices. J Interv Cardiol 14(1):49–55
Krumsdorf U, Ostermayer S, Billinger K, Trepels T, Zadan E, Horvath K, Sievert H (2004) Incidence and clinical course of thrombus formation on atrial septal defect and patient foramen ovale closure devices in 1000 consecutive patients. J Am Coll Cardiol 43(2):302–309
Latson LA, Zahn EM, Wilson N (2000) Helex Septal Occluder for closure of atrial septal defects. Current Interventional Cardiology Reports 2:268–273
Latson LA, Wilson N (2000) A new transcatheter ASD closure device. Transcatheter closure of atrial–septal defects a safe, effective option. Cardiac Consult Magazine X(3):4
Pedra CA, Pedra SF, Esteves CA, Chamie F, Ramos S, Pontes SC Jr, Tress JC, Braga SL, Latson LA, Fontes VF (2003) Initial experience in Brazil with the Helex septal occluder for percutaneous occlusion of atrial septal defects. Arq Bras Cardiol 81(5):435–452
Pedra CA, Pedra SR, Esteves CA, Cassar R, Pontes SC Jr, Braga SL, Fontes VF (2004) Transcatheter closure of secundum atrial septal defects with complex anatomy. J Invasive Cardiol 16(3):117– 122
Peuster M, Reckers J, Fink C (2003) Secondary embolization of a Helex occluder implanted into a secundum atrial septal defect. Catheter Cardiovasc Interv 59(1):77–82
Post MC, Van Deyk K, Budts W (2005) Percutaneous closure of a patent foramen ovale: single–centre experience using different types of devices and mid–term outcome. Acta Cardiol 60(5):515–519
Sievert H, Horvath K, Zadan E et al. (2001) Patent foramen ovale closure in patients with transient ischemia attack/stroke. Journal of Interventional Cardiology 14(2):261–266
Sievert H, Krumsdorf U (2002) Transcatheter closure of intracardiac shunts. Z Kardiol 91 (Suppl 3):77–83
Skowasch D, Peuster M, Andrie R, Tiemann K, Luderitz B, Bauriedel G (2004) Transcatheter PFO closure with a prominent Eustachian valve. Z Kardiol 93(2):162–165
Vincent RN, Raviele AA, Diehl HJ (2003) Single–center experience with the HELEX septal occluder for closure of atrial septal defects in children. J Interv Cardiol 16(1):79–82
Zahn EM, Cheatham J, Latson LA, Wilson N (1999) Results of in vivo testing of a new Nitinol ePTFE septal occlusion device. Catheterization and Cardiovascular Diagnosis 47:124
Zahn EM, Wilson N, Cutright W, Latson LA (2001) Development and testing of the Helex Septal Occluder, a new expanded polytetrafluoro–ethylene atrial septal defect occlusion system. Circulation 104:711–716
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kozlik-Feldmann, R., Dalla Pozza, R., Römer, U. et al. First experience with the 2005 modified Gore Helex ASD occluder system. Clin Res Cardiol 95, 468–473 (2006). https://doi.org/10.1007/s00392-006-0413-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-006-0413-8