Abstract
Purpose
γ-Catenin is a protein closely related to β-catenin. While the overexpression of β-catenin has been linked with impaired prognosis and survival in various malignancies, both oncogenic and tumor suppressor functions have been described for γ-catenin. Thus, its role in cancer remains controversial. In this study, we examined the impact of γ-catenin expression on the malignant potential of colorectal cancer cells.
Methods
γ-Catenin was knocked down by short interfering RNA in the γ-catenin-proficient DLD-1 cell line and stably overexpressed in the γ-catenin-deficient cell line RKO. The effects of these molecular manipulations on the malignant potential of the cell lines were tested in vitro and in vivo in a xenograft tumor model.
Results
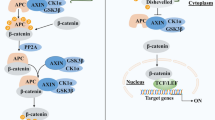
γ-Catenin contributed to Wnt signaling independent of the cellular context. Unlike its sister molecule β-catenin, γ-catenin inhibited cellular invasion and anoikis in cells endogenously expressing γ-catenin. In line with this tumor suppressor function, its de novo expression in RKO cells inhibited proliferation via cell cycle arrest. In a xenograft tumor model, overexpression of γ-catenin starkly reduced tumor growth in vivo.
Conclusions
This is the first report demonstrating a tumor-suppressive effect of γ-catenin in colorectal cancer both in vitro and in vivo. Detailed in vitro analysis revealed that effects of γ-catenin differ in γ-catenin proficient and deficient cells, indicating that its function in colorectal cancer is dependent on the cellular context. This finding adds to our understanding of γ-catenin and may have implications for future studies of catenin/Wnt targeted cancer therapies.




Similar content being viewed by others
References
Zhurinsky J, Shtutman M, Ben-Ze’ev A (2000) Plakoglobin and beta-catenin: protein interactions, regulation and biological roles. J Cell Sci 113(Pt 18):3127–3139
Ben-Ze’ev A, Geiger B (1998) Differential molecular interactions of beta-catenin and plakoglobin in adhesion, signaling and cancer. Curr Opin Cell Biol 10(5):629–639
Kolligs FT, Bommer G, Goke B (2002) Wnt/beta-catenin/tcf signaling: a critical pathway in gastrointestinal tumorigenesis. Digestion, 66 (3):131–144. doi:10.1159/000066755
Williams BO, Barish GD, Klymkowsky MW, Varmus HE (2000) A comparative evaluation of beta-catenin and plakoglobin signaling activity. Oncogene 19(50):5720–5728. doi:10.1038/sj.onc.1203921
Maeda O, Usami N, Kondo M, Takahashi M, Goto H, Shimokata K, Kusugami K, Sekido Y (2004) Plakoglobin (gamma-catenin) has TCF/LEF family-dependent transcriptional activity in beta-catenin-deficient cell line. Oncogene 23(4):964–972. doi:10.1038/sj.onc.1207254
Miravet S, Piedra J, Miro F, Itarte E, Garcia de Herreros A, Dunach M (2002) The transcriptional factor Tcf-4 contains different binding sites for beta-catenin and plakoglobin. J Biol Chem 277(3):1884–1891. doi:10.1074/jbc.M110248200
Simcha I, Shtutman M, Salomon D, Zhurinsky J, Sadot E, Geiger B, Ben-Ze’ev A (1998) Differential nuclear translocation and transactivation potential of beta-catenin and plakoglobin. J Cell Biol 141(6):1433–1448
Kolligs FT, Hu G, Dang CV, Fearon ER (1999) Neoplastic transformation of RK3E by mutant beta-catenin requires deregulation of Tcf/Lef transcription but not activation of c-myc expression. Mol Cell Biol 19(8):5696–5706
Kolligs FT, Kolligs B, Hajra KM, Hu G, Tani M, Cho KR, Fearon ER (2000) gamma-catenin is regulated by the APC tumor suppressor and its oncogenic activity is distinct from that of beta-catenin. Genes Dev 14(11):1319–1331
Caca K, Kolligs FT, Ji X, Hayes M, Qian J, Yahanda A, Rimm DL, Costa J, Fearon ER (1999) Beta- and gamma-catenin mutations, but not E-cadherin inactivation, underlie T-cell factor/lymphoid enhancer factor transcriptional deregulation in gastric and pancreatic cancer. Cell Growth Differ 10(6):369–376
Shiina H, Breault JE, Basset WW, Enokida H, Urakami S, Li LC, Okino ST, Deguchi M, Kaneuchi M, Terashima M, Yoneda T, Shigeno K, Carroll PR, Igawa M, Dahiya R (2005) Functional loss of the gamma-catenin gene through epigenetic and genetic pathways in human prostate cancer. Cancer Res 65(6):2130–2138. doi:10.1158/0008-5472.CAN-04-3398
Skotheim RI, Abeler VM, Nesland JM, Fossa SD, Holm R, Wagner U, Florenes VA, Aass N, Kallioniemi OP, Lothe RA (2003) Candidate genes for testicular cancer evaluated by in situ protein expression analyses on tissue microarrays. Neoplasia 5(5):397–404
Varis A, Wolf M, Monni O, Vakkari ML, Kokkola A, Moskaluk C, Frierson H Jr, Powell SM, Knuutila S, Kallioniemi A, El-Rifai W (2002) Targets of gene amplification and overexpression at 17q in gastric cancer. Cancer Res 62(9):2625–2629
Nagel JM, Kriegl L, Horst D, Engel J, Gautam S, Mantzoros CS, Kirchner T, Goke B, Kolligs FT (2010) gamma-Catenin is an independent prognostic marker in early stage colorectal cancer. Int J Color Dis 25(11):1301–1309. doi:10.1007/s00384-010-1046-y
Parker HR, Li Z, Sheinin H, Lauzon G, Pasdar M (1998) Plakoglobin induces desmosome formation and epidermoid phenotype in N-cadherin-expressing squamous carcinoma cells deficient in plakoglobin and E-cadherin. Cell Motil Cytoskeleton 40(1):87–100. doi:10.1002/(SICI)1097-0169(1998)40:1<87::AID-CM8>3.0.CO;2-C
Shafiei F, Rahnama F, Pawella L, Mitchell MD, Gluckman PD, Lobie PE (2008) DNMT3A and DNMT3B mediate autocrine hGH repression of plakoglobin gene transcription and consequent phenotypic conversion of mammary carcinoma cells. Oncogene 27(18):2602–2612. doi:10.1038/sj.onc.1210917
Winn RA, Bremnes RM, Bemis L, Franklin WA, Miller YE, Cool C, Heasley LE (2002) gamma-Catenin expression is reduced or absent in a subset of human lung cancers and re-expression inhibits transformed cell growth. Oncogene 21(49):7497–7506. doi:10.1038/sj.onc.1205963
Rieger-Christ KM, Ng L, Hanley RS, Durrani O, Ma H, Yee AS, Libertino JA, Summerhayes IC (2005) Restoration of plakoglobin expression in bladder carcinoma cell lines suppresses cell migration and tumorigenic potential. Br J Cancer 92(12):2153–2159. doi:10.1038/sj.bjc.6602651
Weng Z, Xin M, Pablo L, Grueneberg D, Hagel M, Bain G, Muller T, Papkoff J (2002) Protection against anoikis and down-regulation of cadherin expression by a regulatable beta-catenin protein. J Biol Chem 277(21):18677–18686. doi:10.1074/jbc.M105331200
Hakimelahi S, Parker HR, Gilchrist AJ, Barry M, Li Z, Bleackley RC, Pasdar M (2000) Plakoglobin regulates the expression of the anti-apoptotic protein BCL-2. J Biol Chem 275(15):10905–10911
Nagashima H, Okada M, Hidai C, Hosoda S, Kasanuki H, Kawana M (1997) The role of cadherin-catenin-cytoskeleton complex in angiogenesis: antisense oligonucleotide of plakoglobin promotes angiogenesis in vitro, and protein kinase C (PKC) enhances angiogenesis through the plakoglobin signaling pathway. Heart Vessels Suppl 12:110–112
Mukhina S, Mertani HC, Guo K, Lee KO, Gluckman PD, Lobie PE (2004) Phenotypic conversion of human mammary carcinoma cells by autocrine human growth hormone. Proc Natl Acad Sci U S A 101(42):15166–15171. doi:10.1073/pnas.0405881101
Simcha I, Geiger B, Yehuda-Levenberg S, Salomon D, Ben-Ze’ev A (1996) Suppression of tumorigenicity by plakoglobin: an augmenting effect of N-cadherin. J Cell Biol 133(1):199–209
Aceto N, Bardia A, Miyamoto DT, Donaldson MC, Wittner BS, Spencer JA, Yu M, Pely A, Engstrom A, Zhu H, Brannigan BW, Kapur R, Stott SL, Shioda T, Ramaswamy S, Ting DT, Lin CP, Toner M, Haber DA, Maheswaran S (2014) Circulating tumor cell clusters are oligoclonal precursors of breast cancer metastasis. Cell 158(5):1110–1122. doi:10.1016/j.cell.2014.07.013
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Animal handling and experimentation were performed in accordance with German Animal Protection Law and approved by the Animal Care and Use Committee of Bavaria (AZ 55.2-1-54-2531-49/06).
Electronic supplementary material
Supplementary Figure 1.
Efficient siRNA knock-down of ß-catenin in DLD-1 cells as shown by Western Blot and Immunofluorescence. a DLD-1 cells were transfected with siRNA against ß-catenin. 48h after transfection, cells were harvested and probed with antibodies against ß-catenin by Western Blotting. Expression of β-actin served as a control for equal loading. b Control and ß-catenin siRNA transfected DLD-1 cells were stained with ß-catenin antibodies by immunofluorescence. (PPTX 397 kb)
Rights and permissions
About this article
Cite this article
Nagel, J.M., Lahm, H., Ofner, A. et al. γ-Catenin acts as a tumor suppressor through context-dependent mechanisms in colorectal cancer. Int J Colorectal Dis 32, 1243–1251 (2017). https://doi.org/10.1007/s00384-017-2846-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-017-2846-0