Abstract
Purpose
Presently, no markers exist to predict metachronous metastasis at the time a primary colorectal cancer is diagnosed. While aneuploidy indicates poor survival prognosis and elevated carcinoembryonic antigen (CEA) levels the presence of recurrent disease, the predictive value of both markers regarding imminent metachronous metastases is unclear.
Methods
Sixty patients with distant recurrence throughout a 5-year follow-up (TM+) were randomly chosen and 60 patients without metastasis matched to this cohort (TM−). In addition, an enlarged collective (n = 217; n TM+ = 85, n TM- = 132) with median follow-up of 79.2 months was assessed by logistic regression regarding metachronous metastases. Univariate and stepwise regression analyses included clinicopathological characteristics, preoperative CEA levels and aneuploidy assessed by DNA image cytometry.
Results
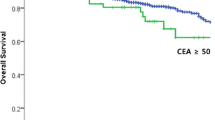
The matched-pair collective showed aneuploidy in 71.1 % (TM−) and 85.0 % (TM+; p = 0.076), and elevated CEA in 24.5 % (TM−) and 52.2 % [TM+; odds ratio (OR), 3.414; p = 0.007]. The enlarged collective presented aneuploidy in 71.2 % (TM−) and 83.5 % (TM+; OR 2.050, p = 0.038), and elevated CEA in 28.6 % (TM−) and 48.9 % (TM+; OR 2.391, p = 0.020). Elevated CEA and aneuploidy did not show any association (p = 0.919). In contrast, logistic regression analyses demonstrated that besides increased T category (OR 1.745, p = 0.019), both elevated CEA level (OR 2.633, p = 0.015) and aneuploidy (OR 1.929, p = 0.058) were independent predictive markers for metachronous metastasis.
Conclusions
Our data show that aneuploidy and elevated CEA levels besides increased T category could serve for individual risk assessment to predict metachronous metastases. The fact that still aneuploidy missed the significance level by a small margin emphasizes the need for larger validation studies.


Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, Thun MJ (2008) Cancer statistics, 2008. CA Cancer J Clin 58(2):71–96. doi:10.3322/CA.2007.0010
Laubert T, Habermann JK, Hemmelmann C, Kleemann M, Oevermann E, Bouchard R, Hildebrand P, Jungbluth T, Burk C, Esnaashari H, Schloricke E, Hoffmann M, Ziegler A, Bruch HP, Roblick UJ (2012) Metachronous metastasis- and survival-analysis show prognostic importance of lymphadenectomy for colon carcinomas. BMC Gastroenterol 12:24. doi:10.1186/1471-230X-12-24
Chuang SC, Su YC, Lu CY, Hsu HT, Sun LC, Shih YL, Ker CG, Hsieh JS, Lee KT, Wang JY (2011) Risk factors for the development of metachronous liver metastasis in colorectal cancer patients after curative resection. World J Surg 35(2):424–429. doi:10.1007/s00268-010-0881-x
Stein U, Walther W, Arlt F, Schwabe H, Smith J, Fichtner I, Birchmeier W, Schlag PM (2009) MACC1, a newly identified key regulator of HGF-MET signaling, predicts colon cancer metastasis. Nat Med 15(1):59–67. doi:10.1038/nm.1889
Schmiegel W, Reinacher-Schick A, Arnold D, Graeven U, Heinemann V, Porschen R, Riemann J, Rodel C, Sauer R, Wieser M, Schmitt W, Schmoll HJ, Seufferlein T, Kopp I, Pox C (2008) Update S3-guideline "colorectal cancer" 2008. Z Gastroenterol 46(8):799–840. doi:10.1055/s-2008-1027726
Wiratkapun S, Kraemer M, Seow-Choen F, Ho YH, Eu KW (2001) High preoperative serum carcinoembryonic antigen predicts metastatic recurrence in potentially curative colonic cancer: results of a five-year study. Dis Colon Rectum 44(2):231–235
Park IJ, Choi GS, Lim KH, Kang BM, Jun SH (2009) Serum carcinoembryonic antigen monitoring after curative resection for colorectal cancer: clinical significance of the preoperative level. Ann Surg Oncol 16(11):3087–3093. doi:10.1245/s10434-009-0625-z
Araujo SE, Bernardo WM, Habr-Gama A, Kiss DR, Cecconello I (2007) DNA ploidy status and prognosis in colorectal cancer: a meta-analysis of published data. Dis Colon Rectum 50(11):1800–1810. doi:10.1007/s10350-007-9013-6
Geigl JB, Obenauf AC, Schwarzbraun T, Speicher MR (2008) Defining 'chromosomal instability'. Trends Genet 24(2):64–69. doi:10.1016/j.tig.2007.11.006
Pino MS, Chung DC (2010) The chromosomal instability pathway in colon cancer. Gastroenterology 138(6):2059–2072. doi:10.1053/j.gastro.2009.12.065
Witzig TE, Loprinzi CL, Gonchoroff NJ, Reiman HM, Cha SS, Wieand HS, Katzmann JA, Paulsen JK, Moertel CG (1991) DNA ploidy and cell kinetic measurements as predictors of recurrence and survival in stages B2 and C colorectal adenocarcinoma. Cancer 68(4):879–888
Gerling M, Meyer KF, Fuchs K, Igl BW, Fritzsche B, Ziegler A, Bader F, Kujath P, Schimmelpenning H, Bruch HP, Roblick UJ, Habermann JK (2010) High frequency of aneuploidy defines ulcerative colitis-associated carcinomas: a comparative prognostic study to sporadic colorectal carcinomas. Ann Surg. doi:10.1097/SLA.0b013e3181deb664
Lin JK, Chang SC, Yang SH, Jiang JK, Chen WC, Lin TC (2003) Prognostic value of DNA ploidy patterns of colorectal adenocarcinoma. Hepatogastroenterology 50(54):1927–1932
Deans GT, Williamson K, Hamilton P, Heatley M, Arthurs K, Patterson CC, Rowlands BJ, Parks TG, Spence RA (1993) DNA densitometry of colorectal cancer. Gut 34(11):1566–1571
Walther A, Houlston R, Tomlinson I (2008) Association between chromosomal instability and prognosis in colorectal cancer: a meta-analysis. Gut 57(7):941–950. doi:10.1136/gut.2007.135004
Sun XF (2006) Clinicopathological and biological features of DNA tetraploid colorectal cancers. Cancer J 12(6):501–506
Bendardaf R, Lamlum H, Ristamaki R, Algars A, Collan Y, Pyrhonen S (2004) Response to chemotherapy (irinotecan plus 5-fluorouracil) in colorectal carcinoma can be predicted by tumour DNA content. Oncology 66(1):46–52. doi:10.1159/000076334
Armitage NC, Ballantyne KC, Sheffield JP, Clarke P, Evans DF, Hardcastle JD (1991) A prospective evaluation of the effect of tumor cell DNA content on recurrence in colorectal cancer. Cancer 67(10):2599–2604
Tang R, Ho YS, Chen HH, See LC, Wang JY (1998) Different prognostic effect of postoperative chemoradiation therapy on diploid and nondiploid high-risk rectal cancers. Dis Colon Rectum 41(12):1494–1499
Fleming I, Cooper J, Henson DE et al (1997) AJCC Cancer Staging Manual, 5th edn. Lippincot-Raven, Philadelphia
Auer GU, Caspersson TO, Wallgren AS (1980) DNA content and survival in mammary carcinoma. Anal Quant Cytol 2(3):161–165
Risques RA, Moreno V, Ribas M, Marcuello E, Capella G, Peinado MA (2003) Genetic pathways and genome-wide determinants of clinical outcome in colorectal cancer. Cancer Res 63(21):7206–7214
Salazar R, Roepman P, Capella G, Moreno V, Simon I, Dreezen C, Lopez-Doriga A, Santos C, Marijnen C, Westerga J, Bruin S, Kerr D, Kuppen P, van de Velde C, Morreau H, Van Velthuysen L, Glas AM, Van’t Veer LJ, Tollenaar R (2011) Gene expression signature to improve prognosis prediction of stage II and III colorectal cancer. J Clin Oncol 29(1):17–24. doi:10.1200/JCO.2010.30.1077
Smith JJ, Deane NG, Wu F, Merchant NB, Zhang B, Jiang A, Lu P, Johnson JC, Schmidt C, Bailey CE, Eschrich S, Kis C, Levy S, Washington MK, Heslin MJ, Coffey RJ, Yeatman TJ, Shyr Y, Beauchamp RD (2010) Experimentally derived metastasis gene expression profile predicts recurrence and death in patients with colon cancer. Gastroenterology 138(3):958–968. doi:10.1053/j.gastro.2009.11.005
Heinzerling JH, Anthony T, Livingston EH, Huerta S (2007) Predictors of distant metastasis and mortality in patients with stage II colorectal cancer. Am Surg 73(3):230–238
Kerr D, Gray R, Quirke P, Watson D, Yothers G, Lavery IC, Lee M, O’Connell MJ, Shak S, Wolmark N (2009) A quantitative multigene RT-PCR assay for prediction of recurrence in stage II colon cancer: selection of the genes in four large studies and results of the independent, prospectively designed QUASAR validation study. J Clin Oncol 27(15s):suppl; abstr 4000
Soreide K, Slewa A, Stokkeland PJ, van Diermen B, Janssen EA, Soreide JA, Baak JP, Korner H (2009) Microsatellite instability and DNA ploidy in colorectal cancer: potential implications for patients undergoing systematic surveillance after resection. Cancer 115(2):271–282. doi:10.1002/cncr.24024
Canna K, McArdle PA, McMillan DC, McNicol AM, Smith GW, McKee RF, McArdle CS (2005) The relationship between tumour T-lymphocyte infiltration, the systemic inflammatory response and survival in patients undergoing curative resection for colorectal cancer. Br J Cancer 92(4):651–654. doi:10.1038/sj.bjc.6602419
Takagawa R, Fujii S, Ohta M, Nagano Y, Kunisaki C, Yamagishi S, Osada S, Ichikawa Y, Shimada H (2008) Preoperative serum carcinoembryonic antigen level as a predictive factor of recurrence after curative resection of colorectal cancer. Ann Surg Oncol 15(12):3433–3439. doi:10.1245/s10434-008-0168-8
Acknowledgments
Grants from the Ad Infinitum Foundation, the Werner & Clara Kreitz Foundation, and the H.W. & J. Hector Foundation are gratefully acknowledged. This study was performed in collaboration with the “North German Tumorbank of Colorectal Cancer” (DKH #108446) and the “Surgical Center for Translational Oncology–Lübeck” (SCTO-L).
Conflict of interest
All authors declare that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Laubert, T., Bente, V., Freitag-Wolf, S. et al. Aneuploidy and elevated CEA indicate an increased risk for metachronous metastasis in colorectal cancer. Int J Colorectal Dis 28, 767–775 (2013). https://doi.org/10.1007/s00384-012-1625-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-012-1625-1