Abstract
Introduction
Hypoperfusion of the bowel is a risk factor for anastomotic failure. Electrical field stimulation has been shown to improve repair in ischemic tissue, but its influence in hypoperfused colon has not been investigated. The hypothesis of this experimental animal study was that electrical field stimulation improves anastomotic healing in ischemic bowel.
Materials and methods
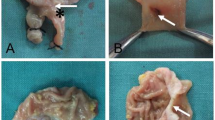
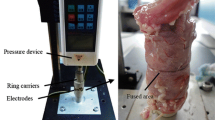
Thirty rats were divided evenly into three groups: control, ischemia/placebo, and ischemia/test group. Ischemia was induced by ligation of the arterial supply to the proximal colon. The watershed area was identified and transected. Field stimulation was achieved by application of negatively charged diethylaminoethyl Sephadex beads in methylcellulose gel to the colonic epithelium prior to anastomosis. The placebo group had methylcellulose gel only applied and control animals had anastomosis only. Anastomotic strength was measured using anastomotic bursting pressure and hydroxyproline content. Systemic effect was investigated via interleukin-6 and vascular endothelial growth factor assay.
Results
The ischemia/electrical field stimulation (EFS) group had significantly increased bursting pressure and hydroxyproline content in comparison with the placebo group (P < 0.001). Serum cytokine levels were unaffected.
Conclusion
Negatively charged EFS improves anastomotic healing in hypoperfused colon without induction of systemic cytokines and has potential as a local treatment in high-risk bowel anastomosis.






Similar content being viewed by others
References
McArdle CS, McMillan DC, Hole DJ (2005) Impact of anastomotic leakage on long-term survival of patients undergoing curative resection for colorectal cancer. Br J Surg 92(9):1150–1154
Law WL, Choi HK, Lee YM, Ho JW, Seto CL (2007) Anastomotic leakage is associated with poor long-term outcome in patients after curative colorectal resection for malignancy. J Gastrointest Surg 11(1):8–15
Walker KG, Bell SW, Rickard MJ, Mehanna D, Dent OF, Chapuis PH, Bokey EL (2004) Anastomotic leakage is predictive of diminished survival after potentially curative resection for colorectal cancer. Ann Surg 240(2):255–259
Montesani C, De MR, Chiappalone S, Narilli P, D’Amato A, Ribotta G (1992) Critical evaluation of the anastomoses in large bowel surgery: experience gained in 533 cases. Hepatogastroenterology 39(4):304–308
Hyman N, Manchester TL, Osler T, Burns B, Cataldo PA (2007) Anastomotic leaks after intestinal anastomosis: it’s later than you think. Ann Surg 245(2):254–258
Pronio A, Di FA, Narilli P, Mancini B, Caporilli D, Piroli S, Vestri A, Montesani C (2007) Anastomotic dehiscence in colorectal surgery. Analysis of 1290 patients. Chir Ital 59(5):599–609
Matthiessen P, Hallbook O, Rutegard J, Sjodahl R (2004) Intraoperative adverse events and outcome after anterior resection of the rectum. Br J Surg 91(12):1608–1612
Zhao M, Song B, Pu J, Wada T, Reid B, Tai G, Wang F, Guo A, Walczysko P, Gu Y, Sasaki T, Suzuki A, Forrester JV, Bourne HR, Devreotes PN, McCaig CD, Penninger JM (2006) Electrical signals control wound healing through phosphatidylinositol-3-OH kinase-gamma and PTEN. Nature 442(7101):457–460
Guler M, Kologlu M, Kama NA, Renda N, Gozalan U, Yuksek YN, Daglar G (2002) Effect of topically applied charged particles on healing of colonic anastomoses. Arch Surg 137(7):813–817
Reddy GK, Enwemeka CS (1996) A simplified method for the analysis of hydroxyproline in biological tissues. Clin Biochem 29(3):225–229
Grzelak I, Olszewski WL, Zaleska M, Durlik M, Lagiewska B, Muszynski M, Rowinski W (1996) Blood cytokine levels rise even after minor surgical trauma. J Clin Immunol 16(3):159–164
Seyhan N, Guler G (2006) Review of in vivo static and ELF electric fields studies performed at Gazi Biophysics Department. Electromagn Biol Med 25(4):307–323
Krukowski M, Simmons DJ, Summerfield A, Osdoby P (1988) Charged beads: generation of bone and giant cells. J Bone Miner Res 3(2):165–171
De MM, Gagliano N, Moscheni C, Dellavia C, Calastrini C, Pellati A, Gioia M, Caruso A, Stabellini G (2005) Changes in polyamines, c-myc and c-fos gene expression in osteoblast-like cells exposed to pulsed electromagnetic fields. Bioelectromagnetics 26(3):207–214
Haddad JB, Obolensky AG, Shinnick P (2007) The biologic effects and the therapeutic mechanism of action of electric and electromagnetic field stimulation on bone and cartilage: new findings and a review of earlier work. J Altern Complement Med 13(5):485–490
Foulds IS, Barker AT (1983) Human skin battery potentials and their possible role in wound healing. Br J Dermatol 109(5):515–522
Kanof NM (1964) Gold leaf in the treatment of cutaneous ulcers. J Invest Dermatol 43:441–442
McGinnis ME, Vanable JW Jr (1986) Voltage gradients in newt limb stumps. Prog Clin Biol Res 210:231–238
Sta Iglesia DD, Vanable JW Jr (1998) Endogenous lateral electric fields around bovine corneal lesions are necessary for and can enhance normal rates of wound healing. Wound Repair Regen 6(6):531–542
Cheng K, Goldman RJ (1998) Electric fields and proliferation in a dermal wound model: cell cycle kinetics. Bioelectromagnetics 19(2):68–74
Sun S, Cho M (2004) Human fibroblast migration in three-dimensional collagen gel in response to noninvasive electrical stimulus. II. Identification of electrocoupling molecular mechanisms. Tissue Eng 10(9–10):1558–1565
Chao PH, Lu HH, Hung CT, Nicoll SB, Bulinski JC (2007) Effects of applied DC electric field on ligament fibroblast migration and wound healing. Connect Tissue Res 48(4):188–197
Zhao M, Bai H, Wang E, Forrester JV, McCaig CD (2004) Electrical stimulation directly induces pre-angiogenic responses in vascular endothelial cells by signaling through VEGF receptors. J Cell Sci 117(Pt 3):397–405
Tepper OM, Callaghan MJ, Chang EI, Galiano RD, Bhatt KA, Baharestani S, Gan J, Simon B, Hopper RA, Levine JP, Gurtner GC (2004) Electromagnetic fields increase in vitro and in vivo angiogenesis through endothelial release of FGF-2. FASEB J 18(11):1231–1233
Collins D, Ridgway PF, Winter DC, Fennelly D, Evoy D (2009) Gastrointestinal perforation in metastatic carcinoma: a complication of bevacizumab therapy. Eur J Surg Oncol 35:444–446
August DA, Serrano D, Poplin E (2008) “Spontaneous,” delayed colon and rectal anastomotic complications associated with bevacizumab therapy. J Surg Oncol 97(2):180–185
Lipska MA, Bissett IP, Parry BR, Merrie AE (2006) Anastomotic leakage after lower gastrointestinal anastomosis: men are at a higher risk. ANZ J Surg 76(7):579–585
Alberts JC, Parvaiz A, Moran BJ (2003) Predicting risk and diminishing the consequences of anastomotic dehiscence following rectal resection. Colorectal Dis 5(5):478–482
Ba ZF, Yokoyama Y, Toth B, Rue LW III, Bland KI, Chaudry IH (2004) Gender differences in small intestinal endothelial function: inhibitory role of androgens. Am J Physiol Gastrointest Liver Physiol 286(3):G452–G457
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kennelly, R., Conneely, J.B., Bouchier-Hayes, D.J. et al. Electrical field stimulation promotes anastomotic healing in poorly perfused rat colon. Int J Colorectal Dis 26, 339–344 (2011). https://doi.org/10.1007/s00384-010-1053-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-010-1053-z