Abstract
Background
Immunosuppressive calcineurin inhibitors, like cyclosporine (CsA), can be used for the clinical management of severe ulcerative colitis. However, patients treated with CsA are at a risk for developing kidney failure and may be more susceptible to colon cancer. Furthermore, severe neurotoxicity and hypertension are common problems. To avoid the side effects of CsA, new immunosuppressive drugs to treat colitis are needed. The aim of the present study was to test the immunosuppressive mammalian target of rapamycin inhibitor rapamycin in an experimental model of chronic colitis and to compare its effectiveness with CsA.
Methods
Chronic colitis was established in Balb/c mice after four feeding cycles of dextran sodium sulfate. Because leukocyte recruitment to sites of intestinal inflammation is crucial for the development of chronic colitis, intravital microscopy was used to study the effect of rapamycin and CsA on leukocyte–endothelium interactions and leukocyte extravasation. To assess the degree of colitis, histological sections were evaluated.
Results
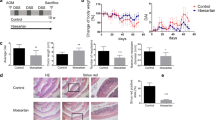
Both rapamycin and cyclosporine effectively reduced leukocyte sticking (>60%) in submucosal venules, as compared to controls. Furthermore, rapamycin, but not CsA, reduced (>35%) leukocyte extravasation in the mucosa. Both rapamycin and CsA treatments significantly improved the histologic inflammation score.
Conclusion
Our in vivo results demonstrate that rapamycin reduces leukocyte sticking and extravasation during chronic colitis induction and proves to be as effective as CsA at reducing experimental chronic colitis. These results support the use of rapamycin in clinical trials to avoid serious side effects of CsA therapy in chronic colitis patients.



Similar content being viewed by others
Abbreviations
- CsA:
-
Cyclosporine
- DSS:
-
Dextran sodium sulfate
References
Guslandi M (1999) Microcirculation in inflammatory bowel diseases. Int J Colorectal Dis 14:265
van Assche G, Rutgeerts P (2002) Antiadhesion molecule therapy in inflammatory bowel disease. Inflamm Bowel Dis 8:291–300
Spiik AK, Ridderstad A, Axelsson LG, Midtvedt T, Bjork L, Pettersson S (2002) Abrogated lymphocyte infiltration and lowered CD14 in dextran sulfate induced colitis in mice treated with p65 antisense oligonucleotides. Int J Colorectal Dis 17:223–232
Farkas S, Herfarth H, Rossle M, Schroeder J, Steinbauer M, Guba M, Beham A, Scholmerich J, Jauch KW, Anthuber M (2001) Quantification of mucosal leucocyte endothelial cell interaction by in vivo fluorescence microscopy in experimental colitis in mice. Clin Exp Immunol 126:250–258
Banic M, Anic B, Brkic T, Ljubicic N, Plesko S, Dohoczky C, Erceg D, Petrovecki M, Stipancic I, Rotkvic I (2002) Effect of cyclosporine in a murine model of experimental colitis. Dig Dis Sci 47:1362–1368
Lichtiger S, Present DH, Kornbluth A, Gelernt I, Bauer J, Galler G, Michelassi F, Hanauer S (1994) Cyclosporine in severe ulcerative colitis refractory to steroid therapy. N Engl J Med 330:1841–1845
D’Haens G, Lemmens L, Geboes K, Vandeputte L, Van Acker F, Mortelmans L, Peeters M, Vermeire S, Penninckx F, Nevens F, Hiele M, Rutgeerts P (2001) Intravenous cyclosporine versus intravenous corticosteroids as single therapy for severe attacks of ulcerative colitis. Gastroenterology 120:1323–1329
Loftus CG, Loftus EV Jr, Sandborn WJ (2003) Cyclosporin for refractory ulcerative colitis. Gut 52:172–173
Loftus CG, Egan LJ, Sandborn WJ (2004) Cyclosporine, tacrolimus, and mycophenolate mofetil in the treatment of inflammatory bowel disease. Gastroenterol Clin North Am 33:141–169, vii
Soriano-Izquierdo A, Gironella M, Massaguer A, Salas A, Gil F, Pique JM, Panes J (2004) Effect of cyclosporin A on cell adhesion molecules and leukocyte–endothelial cell interactions in experimental colitis. Inflamm Bowel Dis 10:789–800
Wijdicks EF, Wiesner RH, Krom RA (1995) Neurotoxicity in liver transplant recipients with cyclosporine immunosuppression. Neurology 45:1962–1964
de Mattos AM, Olyaei AJ, Bennett WM (2000) Nephrotoxicity of immunosuppressive drugs: long-term consequences and challenges for the future. Am J Kidney Dis 35:333–346
Olyaei AJ, de Mattos AM, Bennett WM (2001) Nephrotoxicity of immunosuppressive drugs: new insight and preventive strategies. Curr Opin Crit Care 7:384–389
Cohen RD, Stein R, Hanauer SB (1999) Intravenous cyclosporin in ulcerative colitis: a five-year experience. Am J Gastroenterol 94:1587–1592
Fernandez-Banares F, Bertran X, Esteve-Comas M, Cabre E, Menacho M, Humbert P, Planas R, Gassull MA (1996) Azathioprine is useful in maintaining long-term remission induced by intravenous cyclosporine in steroid-refractory severe ulcerative colitis. Am J Gastroenterol 91:2498–2499
Sehgal SN (1998) Rapamune (RAPA, rapamycin, sirolimus): mechanism of action immunosuppressive effect results from blockade of signal transduction and inhibition of cell cycle progression. Clin Biochem 31:335–340
Sehgal SN (2003) Sirolimus: its discovery, biological properties, and mechanism of action. Transplant Proc 35:7S–14S
Dumont FJ, Staruch MJ, Koprak SL, Melino MR, Sigal NH (1990) Distinct mechanisms of suppression of murine T cell activation by the related macrolides FK-506 and rapamycin. J Immunol 144:251–258
Kahan BD (2000) Efficacy of sirolimus compared with azathioprine for reduction of acute renal allograft rejection: a randomised multicentre study. The Rapamune US Study Group. Lancet 356:194–202
Shapiro AM, Lakey JR, Ryan EA, Korbutt GS, Toth E, Warnock GL, Kneteman NM, Rajotte RV (2000) Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N Engl J Med 343:230–238
Schreiber SL, Crabtree GR (1992) The mechanism of action of cyclosporin A and FK506. Immunol Today 13:136–142
Obermeier F, Dunger N, Strauch UG, Grunwald N, Herfarth H, Scholmerich J, Falk W (2003) Contrasting activity of cytosin–guanosin dinucleotide oligonucleotides in mice with experimental colitis. Clin Exp Immunol 134:217–224
Harris AG, Hecht R, Peer F, Nolte D, Messmer K (1997) An improved intravital microscopy system. Int J Microcirc Clin Exp 17:322–327
Menger MD, Steiner D, Messmer K (1992) Microvascular ischemia-reperfusion injury in striated muscle: significance of “no reflow”. Am J Physiol 263:H1892–H1900
Diekmann F, Waiser J, Fritsche L, Dragun D, Neumayer HH, Budde K (2001) Conversion to rapamycin in renal allograft recipients with biopsy-proven calcineurin inhibitor-induced nephrotoxicity. Transplant Proc 33:3234–3235
Citterlo F, Scata MC, Violi P, Romagnoli J, Pozzetto U, Nanni G, Castagneto M (2003) Rapid conversion to sirolimus for chronic progressive deterioration of the renal function in kidney allograft recipients. Transplant Proc 35:1292–1294
Egidi MF, Cowan PA, Naseer A, Gaber AO (2003) Conversion to sirolimus in solid organ transplantation: a single-center experience. Transplant Proc 35:131S–137S
Ciancio G, Burke GW, Gaynor JJ, Mattiazzi A, Roth D, Kupin W, Nicolas M, Ruiz P, Rosen A, Miller J (2004) A randomized long-term trial of tacrolimus/sirolimus versus tacrolimus/mycophenolate mofetil versus cyclosporine (NEORAL)/sirolimus in renal transplantation. II. Survival, function, and protocol compliance at 1 year. Transplantation 77:252–258
Chapman JR (2003) Optimizing the long-term outcome of renal transplants: opportunities created by sirolimus. Transplant Proc 35:67S–72S
Sato T, Inagaki A, Uchida K, Ueki T, Goto N, Matsuoka S, Katayama A, Haba T, Tominaga Y, Okajima Y, Ohta K, Suga H, Taguchi S, Kakiya S, Itatsu T, Kobayashi T, Nakao A (2003) Diabetes mellitus after transplant: relationship to pretransplant glucose metabolism and tacrolimus or cyclosporine A-based therapy. Transplantation 76:1320–1326
Miller LW (2002) Cardiovascular toxicities of immunosuppressive agents. Am J Transplant 2:807–818
Nishiyama A, Kobori H, Fukui T, Zhang GX, Yao L, Rahman M, Hitomi H, Kiyomoto H, Shokoji T, Kimura S, Kohno M, Abe Y (2003) Role of angiotensin II and reactive oxygen species in cyclosporine A-dependent hypertension. Hypertension 42:754–760
Morrisett JD, Abdel-Fattah G, Kahan BD (2003) Sirolimus changes lipid concentrations and lipoprotein metabolism in kidney transplant recipients. Transplant Proc 35:143S–150S
Kahan BD, Stepkowski SM, Napoli KL, Katz SM, Knight RJ, Van Buren C (2000) The development of sirolimus: The University of Texas-Houston experience. Clin Transpl 145–158
Kuypers DR (2005) Benefit–risk assessment of sirolimus in renal transplantation. Drug Safety 28:153–181
Budde K, Neumayer HH, Lehne G, Winkler M, Hauser IA, Lison A, Fritsche L, Soulillou JP, Fauchald P, Dantal J (2004) Tolerability and steady-state pharmacokinetics of everolimus in maintenance renal transplant patients. Nephrol Dial Transplant 19:2606–2614
Kojouharoff G, Hans W, Obermeier F, Mannel DN, Andus T, Scholmerich J, Gross V, Falk W (1997) Neutralization of tumour necrosis factor (TNF) but not of IL-1 reduces inflammation in chronic dextran sulfate sodium-induced colitis in mice. Clin Exp Immunol 107:353–358
Obermeier F, Kojouharoff G, Hans W, Scholmerich J, Gross V, Falk W (1999) Interferon-gamma (IFN-gamma)- and tumour necrosis factor (TNF)-induced nitric oxide as toxic effector molecule in chronic dextran sulfate sodium (DSS)-induced colitis in mice. Clin Exp Immunol 116:238–245
Guba M, von Breitenbuch P, Steinbauer M, Koehl G, Flegel S, Hornung M, Bruns CJ, Zuelke C, Farkas S, Anthuber M, Jauch KW, Geissler EK (2002) Rapamycin inhibits primary and metastatic tumor growth by antiangiogenesis: involvement of vascular endothelial growth factor. Nat Med 8:128–135
Munkholm P (2003) Review article: the incidence and prevalence of colorectal cancer in inflammatory bowel disease. Aliment Pharmacol Ther 18(Suppl 2):1–5
Eaden JA, Abrams KR, Mayberry JF (2001) The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut 48:526–535
Penn I (2000) Post-transplant malignancy: the role of immunosuppression. Drug Safety 23:101–113
Penn I (2000) Some problems with posttransplant lymphoproliferative disease. Transplantation 69:705–706
Garver RI Jr, Zorn GL, Wu X, McGiffin DC, Young KR Jr, Pinkard NB (1999) Recurrence of bronchioloalveolar carcinoma in transplanted lungs. N Engl J Med 340:1071–1074
Ondrus D, Pribylincova V, Breza J, Bujdak P, Miklosi M, Reznicek J, Zvara V (1999) The incidence of tumours in renal transplant recipients with long-term immunosuppressive therapy. Int Urol Nephrol 31:417–422
Herman M, Weinstein T, Korzets A, Chagnac A, Ori Y, Zevin D, Malachi T, Gafter U (2001) Effect of cyclosporin A on DNA repair and cancer incidence in kidney transplant recipients. J Lab Clin Med 137:14–20
Hojo M, Morimoto T, Maluccio M, Asano T, Morimoto K, Lagman M, Shimbo T, Suthanthiran M (1999) Cyclosporine induces cancer progression by a cell-autonomous mechanism. Nature 397:530–534
Seeliger H, Guba M, Koehl GE, Doenecke A, Steinbauer M, Bruns CJ, Wagner C, Frank E, Jauch KW, Geissler EK (2004) Blockage of 2-deoxy-d-ribose-induced angiogenesis with rapamycin counteracts a thymidine phosphorylase-based escape mechanism available for colon cancer under 5-fluorouracil therapy. Clin Cancer Res 10:1843–1852
Bruns CJ, Koehl GE, Guba M, Yezhelyev M, Steinbauer M, Seeliger H, Schwend A, Hoehn A, Jauch KW, Geissler EK (2004) Rapamycin-induced endothelial cell death and tumor vessel thrombosis potentiate cytotoxic therapy against pancreatic cancer. Clin Cancer Res 10:2109–2119
Acknowledgements
This work was supported by a grant from the Deutsche Forschungsgemeinschaft (AN334/1-1) to M.A. and S.F. and by a grant from Wyeth Pharma, Germany. The authors thank Gerhard Rogler for correction of the manuscript and Florian Obermeier for scoring the histologic sections.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farkas, S., Hornung, M., Sattler, C. et al. Rapamycin decreases leukocyte migration in vivo and effectively reduces experimentally induced chronic colitis. Int J Colorectal Dis 21, 747–753 (2006). https://doi.org/10.1007/s00384-005-0793-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-005-0793-7