Abstract
Aims
The aim of this study was to assess the usefulness of PAS, β-catenin and Ki-67 in categorising hepatoblastomas (HBs) and their significance in predicting prognosis. In addition, we have also documented the various chemotherapy induced histologic changes in HBs.
Method
In this retrospective observational study of 29 cases of hepatoblastomas, 27 cases were considered for statistical analysis, excluding two cases of diagnostic core biopsies. All clinicopathological parameters and follow up data were collected. All HB cases including the mixed epithelial and mesenchymal HBs were classified into two groups: fetal predominant and embryonal predominant type according to the predominant epithelial component. PAS, β-catenin and Ki-67 staining were done and their correlation with histologic subtypes was assessed. Kaplan–Meier survival analysis was performed in relation to histology, PAS, β-catenin and Ki-67 staining characteristics.
Result
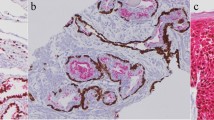
Diffuse nuclear staining of β-catenin was significantly more common in embryonal predominant type (p = 0.036), whereas strong PAS positivity was significantly associated with fetal predominant type (p = 0.002). But no significant correlation was seen between histologic type and Ki-67 staining (p = 0.42). Survival analysis showed cases with diffuse PAS positivity, focal nuclear β-catenin staining and low Ki-67 LI had better survival.
Conclusion
PAS is a simple stain and can be utilised in histological categorisation of HB and also predicting its outcome. Nuclear β-catenin staining which is significantly common in embryonal elements in HB predicts shorter survival.







Similar content being viewed by others

References
Lopez-Terrada D, Zimmermann A (2012) Current issues and controversies in the classification of pediatric hepatocellular tumors. Pediatr Blood Cancer 59:780–784
Chopra A, Iyer VK, Agarwala S, Mathur SR, Aron M, Gupta SD et al (2010) Apoptotic protein expression, glycogen content, DNA ploidy and cell proliferation in hepatoblastoma subtyping and their role in prognostication. Pediatr Surg Int 26:1173–1178
Haas JE, Feusner JH, Finegold MJ (2001) Small cell undifferentiated histology in hepatoblastoma may be unfavorable. Cancer 92:3130–3134
Malogolowkin MH, Katzenstein HM, Meyers RL et al (2011) Complete surgical resection is curative for children with hepatoblastoma with pure fetal histology: a report from the Children’s Oncology Group. J Clin Oncol 29:3301–3306
Kasai M, Watanabe I (1970) Histologic classification of liver-cell carcinoma in infancy and childhood and its clinical evaluation. Cancer 25:552–563
Wei Y, Fabre M, Branchereau S, Gauthier F, Perilongo G, Buendia MA (2000) Activation of β-catenin in epithelial and mesenchymalhepatoblastomas. Oncogene 19(4):498–504
Ranganathan S, Tan X, Monga SP (2005) Β-catenin and met deregulation in childhood Hepatoblastomas. Pediatr Dev Pathol 8(4):435–447
Park WS, Oh RR, Park JY, Kim PJ, Shin MS, Lee JH et al (2001) Nuclear localization of β-catenin is an important prognostic factor in hepatoblastoma. J Pathol 193(4):483–490
Gupta K, Rane S, Das A, Marwaha RK, Menon P, Rao K (2012) Relationship of β-catenin and postchemotherapy histopathologic changes with overall survival in patients with hepatoblastoma. J Pediatr Hematol Oncol 34:e320–e328
Ara T, Fukuzawa M, Oue T, Komoto Y, Kusafuka T, Imura K et al (1997) Immunohistochemical Assessment of the MIB-1 labeling index in human hepatoblastoma and its prognostic relevance. J Pediatr Surg 32:1690–1694
Purcell R, Childs M, Maibach R, Miles C, Turner C, Zimmermann A et al (2011) HGF/c-Met related activation of bcatenin in hepatoblastoma. J Exp Clin Cancer Res 30:96
Spector LG, Birch J (2012) The epidemiology of hepatoblastoma. Pediatr Blood Cancer 59:776–779
Ikeda H, Matsuyama S, Tanimura M (1997) Association between hepatoblastoma and very low birth weight: a trend or a chance? J Pediatr 130:557–560
Weksberg R, Shuman C, Beckwith JB (2009) Beckwith–Wiedemann syndrome. Eur J Hum Genet 18(1):8–14
Hirschman BA, Pollock BH, Tomlinson GE (2005) The spectrum of APC mutations in children with hepatoblastoma from familial adenomatous polyposis kindreds. J Pediatr 147:263–266
von Schweinitz D, Hecker H, Schmidt-von-Arndt G et al (1997) Prognostic factors and staging systems in childhood hepatoblastoma. Int J Cancer 74:593–599
Douglass EC, Reynolds M, Finegold M et al (1993) Cisplatin, vincristine and fluorouracil therapy for hepatoblastoma: a pediatric oncology group study. J Clin Oncol 11:96–99
Brown J, Perilongo G, Shafford E et al (2000) Pretreatment prognostic factors for children with hepatoblastoma—results from the International Society of Pediatric Oncology (SIOP) Study SIOPEL-1. Eur J Cancer 36:1418–1425
Vos A (1995) Primary liver tumors in children. Eur J Surg Cancer 21:101–105
Iyer VK, Kapila K, Agarwala S, Verma K (2005) Fine needle aspiration cytology of hepatoblastoma. Recognition of subtypes on cytomorphology. Acta Cytol 49:355–364
Bera G, Das RN, Islam N, Roy P, Mishra PK, Datta C et al (2017) FNAC aided diagnosis and categorization of hepatoblastoma: a report of three cases. Diagn Cytopathol 45:77–82
Inagawa S, Itabashi M, Adachi S, Kawamoto T, Hori M, Shimazaki J et al (2002) Expression and prognostic roles of β-catenin in hepatocellular carcinoma: correlation with tumor progression and postoperative survival. Clin Cancer Res 8(2):450–456
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declared that they have no potential conflict of interest.
Rights and permissions
About this article
Cite this article
Bera, G., Das, R.N., Roy, P. et al. Utility of PAS and β-catenin staining in histological categorisation and prediction of prognosis of hepatoblastomas. Pediatr Surg Int 33, 961–970 (2017). https://doi.org/10.1007/s00383-017-4115-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-017-4115-2