Abstract
Background/Purpose
In a multi-project research line, we are currently testing whether a morphologically and functionally near normal epidermis can be cultured from human sweat gland (SG) cells and be used as a skin substitute. The present study focuses on the stratum corneum of the epidermis that assumes a vital barrier function for the skin. The main process in the formation of the cornified cell envelope in human epidermis, i.e. crosslinking of proteins and lipids, is catalyzed by several transglutaminases (TG). Therefore, we compared the expression patterns of various TG and their substrates in SG-derived versus keratinocyte-derived epidermal substitutes.
Methods
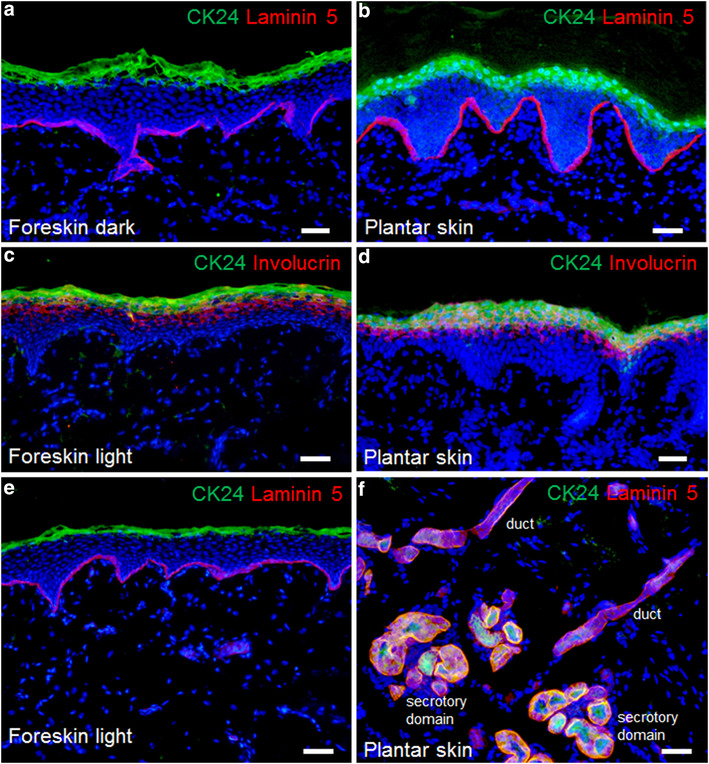
Sweat gland cells, keratinocytes, and fibroblasts were isolated from human skin samples and cultivated separately to generate epidermal substitutes. These were transplanted onto the back of athymic rats. After 2 weeks, the transplants were excised and analyzed histologically as well as by indirect immunofluorescence. We looked at the expression of TG1, 3, 5, and their substrates involucrin and loricrin (=markers of epidermal differentiation) in SG-derived and keratinocyte-derived skin substitutes as well as in normal skin.
Results
The SG cell-derived epidermis was near normal anatomically, formed a cornified cell envelope and demonstrated TG1, 3, and 5 as well as involucrin and loricrin expression patterns similar to those found in keratinocyte-derived epidermis and normal control skin.
Conclusion
These findings support the thesis that SG cells have the potential to form a near normal stratified epidermal analog that might be used as a skin substitute. The expression of TG1 and 3, not normally expressed in human SG, suggests the presence of re-programmed SG cells and/or stem cells capable of both de novo generating and maintaining an epidermis.



Similar content being viewed by others
References
Sheridan RL, Tompkins RG (1995) Cultured autologous epithelium in patients with burns of ninety percent or more of the body surface. J Trauma 38:48–50
Sheridan RL et al (2001) Initial experience with a composite autologous skin substitute. Burns 27:421–424
Matsumura H, Meyer NA, Mann R, Heimbach DM (1998) Melting graft-wound syndrome. J Burn Care Rehabil 19:292–295
Meuli M, Raghunath M (1997) Burns (Part 2): tops and flops using cultured epithelial autografts in children. Pediatr Surg Int 12(7):471–474
Raghunath M, Meuli M (1997) Cultured epithelial autografts: diving from surgery into matrix biology. Pediatr Surg Int 12(7):478–483
Gobet R, Raghunath M, Altermatt S, Meuli-Simmen C, Benathan M, Dietl A, Meuli M (1997) Efficacy of cultured epithelial autografts in pediatric burns and reconstructive surgery. Surgery 121(6):654–661
Raghunath M, Höpfner B, Aeschlimann D, Lüthi U, Meuli M, Altermatt S, Gobet R, Bruckner-Tuderman L, Steinmann B (1996) Cross-linking of the dermo-epidermal junction of skin regenerating from keratinocyte autografts: anchoring fibrils are a target for tissue transglutaminase. J Clin Invest 98(5):1174–1184
Candi E, Schmidt R, Melino G (2005) The cornified envelope: a model of cell death in the skin. Nat Rev Mol Cell Biol 6:328–340
Nemes Z, Steinert PM (1999) Bricks and mortar of the epidermal barrier. Exp Mol Med 31:5–19
Candi E et al (2001) Transglutaminase 5 cross-links loricrin, involucrin, and small proline-rich proteins in vitro. J Biol Chem 276:35014–35023
Steven AC, Steinert PM (1994) Protein composition of cornified cell envelopes of epidermal keratinocytes. J Cell Sci 107(Pt 2):693–700
Steinert PM, Marekov LN (1999) Initiation of assembly of the cell envelope barrier structure of stratified squamous epithelia. Mol Biol Cell 10:4247–4261
Eckert RL, Sturniolo MT, Broome AM, Ruse M, Rorke EA (2005) Transglutaminase function in epidermis. J Invest Dermatol 124:481–492
Hitomi K et al (2003) Analysis of epidermal-type transglutaminase (transglutaminase 3) in human stratified epithelia and cultured keratinocytes using monoclonal antibodies. J Invest Dermatol 32:95–103
Candi E et al (2002) Expression of transglutaminase 5 in normal and pathologic human epidermis. J Invest Dermatol 119:670–677
Pontiggia L, Biedermann T, Meuli M, Widmer D, Böttcher-Haberzeth S, Schiestl C, Schneider J, Braziulis E, Montaño I, Meuli-Simmen C, Reichmann E (2009) Markers to evaluate the quality and self-renewing potential of engineered human skin substitutes in vitro and after transplantation. J Invest Dermatol 129:480–490
Schon M, Benwood J, O’Connell-Willstaedt T, Rheinwald JG (1999) Human sweat gland myoepithelial cells express a unique set of cytokeratins and reveal the potential for alternative epithelial and mesenchymal differentiation states in culture. J Cell Sci 112(Pt 12):1925–1936
Costea DE, Loro LL, Dimba EA, Vintermyr OK, Johannessen AC (2003) Crucial effects of fibroblasts and keratinocyte growth factor on morphogenesis of reconstituted human oral epithelium. J Invest Dermatol 121:1479–1486
Brouard M, Barrandon Y (2003) Controlling skin morphogenesis: hope and despair. Curr Opin Biotechnol 14:520–525
Inada R et al (2000) Facilitated wound healing by activation of the transglutaminase 1 gene. Am J Pathol 157:1875–1882
Stark HJ, Baur M, Breitkreutz D, Mirancea N, Fusenig NE (1999) Organotypic keratinocyte co-cultures in defined medium with regular epidermal morphogenesis and differentiation. J Invest Dermatol 112:681–691
Acknowledgments
This work was supported by a grant from the European Union (EuroSTEC: LSHB-CT-2006-037409) and by the University of Zurich. We are particularly grateful to the Fondation Gaydoul and the sponsors of “DonaTissue” (Thérèse Meier, Robert Zingg, the Vontobel Foundation, and the Werner Spross Foundation) for their generous financial support and interest in our work.
Conflict of interest statement
The authors declare that none of them have any conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tharakan, S., Pontiggia, L., Biedermann, T. et al. Transglutaminases, involucrin, and loricrin as markers of epidermal differentiation in skin substitutes derived from human sweat gland cells. Pediatr Surg Int 26, 71–77 (2010). https://doi.org/10.1007/s00383-009-2517-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-009-2517-5