Abstract
Purpose
Maternal folate intake has reduced the incidence of human neural tube defects by 60–70 %. However, 30–40 % of cases remain nonresponsive to folate intake. The main purpose of this study was to understand the molecular mechanism of folate nonresponsiveness in a mouse model of neural tube defect.
Methods
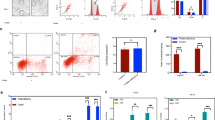
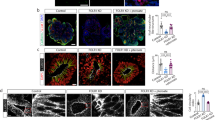
We used a folate-nonresponsive Fkbp8 knockout mouse model to elucidate the molecular mechanism(s) of folate nonresponsiveness. Neurospheres were grown from neural stem cells isolated from the lumbar neural tube of E9.5 Fkbp8 −/− and wild-type embryos. Immunostaining was used to determine the protein levels of oligodendrocyte transcription factor 2 (Olig2), Nkx6.1, class III beta-tubulin (TuJ1), O4, glial fibrillary acidic protein (GFAP), histone H3 Lys27 trimethylation (H3K27me3), ubiquitously transcribed tetratricopeptide repeat (UTX), and Msx2, and quantitative real-time (RT)-PCR was used to determine the message levels of Olig2, Nkx6.1, Msx2, and noggin in neural stem cells differentiated in the presence and absence of folic acid.
Results
Fkbp8 −/−-derived neural stem cells showed (i) increased noggin expression; (ii) decreased Msx2 expression; (iii) premature differentiation—neurogenesis, oligodendrogenesis (Olig2 expression), and gliogenesis (GFAP expression); and (iv) increased UTX expression and decreased H3K27me3 polycomb modification. Exogenous folic acid did not reverse these markers.
Conclusions
Folate nonresponsiveness could be attributed in part to increased noggin expression in Fkbp8 −/− embryos, resulting in decreased Msx2 expression. Folate treatment further increases Olig2 and noggin expression, thereby exacerbating ventralization.






Similar content being viewed by others
References
Czeizel AE, Dudas I (1992) Prevention of the first occurrence of neural-tube defects by periconceptional vitamin supplementation. N Engl J Med 327:1832–1835
Berry RJ, Li Z, Erickson JD, Li S, Moore CA, Wang H, Mulinare J, Zhao P, Wong LY, Gindler J, Hong SX, Correa A (1999) Prevention of neural-tube defects with folic acid in China. China-U.S. Collaborative Project for Neural Tube Defect Prevention. N Engl J Med 341:1485–1490
Berry RJ, Li Z (2002) Folic acid alone prevents neural tube defects: evidence from the China study. Epidemiology 13:114–116
Wald N, Sneddon J, Densen J, Frost C, Stone R (1991) Prevention of neural tube defects: results of the Medical Research Council Vitamin Study. MRC Vitamin Study Research Group. Lancet 338:131–137
Blom HJ, Shaw GM, den Heijer M, Finnell RH (2006) Neural tube defects and folate: case far from closed. Nat Rev Neurosci 7:724–731
Mosley BS, Cleves MA, Siega-Riz AM, Shaw GM, Canfield MA, Waller DK, Werler MM, Hobbs CA, National Birth Defects Prevention Study (2009) Neural tube defects and maternal folate intake among pregnancies conceived after folic acid fortification in the United States. Am J Epidemiol 169:9–17
Shookhoff JM, Gallicano GI (2010) A new perspective on neural tube defects: folic acid and microRNA misexpression. Genesis 48:282–294
Ichi S, Costa FF, Bischof JM, Nakazaki H, Shen YW, Boshnjaku V, Sharma S, Mania-Farnell B, McLone DG, Tomita T, Soares MB, Mayanil CS (2010) Folic acid remodels chromatin on Hes1 and Neurog2 promoters during caudal neural tube development. J Biol Chem 285:36922–36932
Ichi S, Nakazaki H, Boshnjaku V, Singh RM, Mania-Farnell B, Xi G, McLone DG, Tomita T, Mayanil CS (2012) Fetal neural tube stem cells from Pax3 mutant mice proliferate, differentiate, and form synaptic connections when stimulated with folic acid. Stem Cells Dev 21:321–330
Nakazaki H, Reddy AC, Mania-Farnell B, Shen YW, Ichi S, McCabe C, George D, McLone DG, Tomita T, Mayanil CS (2008) Key basic helix-loop-helix transcription factor genes Hes1 and Ngn2 are regulated by Pax3 during mouse embryonic development. Dev Biol 316:510–523
Mayanil CS, Ichi S, Mania-Farnell B, Boshnjaku V, Tomita T, McLone DG (2011) Maternal intake of folic acid and neural crest stem cells. In: Stem cell regulators (vitamins and hormones), vol. 87. Academic, New York, p 143-173
Boshnjaku V, Shim K-W, Tsurubuchi T, Ichi S, Xi G, Mania-Farnell B, McLone DG, Tomita T, Mayanil CS (2012) Nuclear localization of folate receptor alpha: a new role as a transcription factor. Sci Rep 2:980
Leung KY, De Castro SC, Savery D, Copp AJ, Greene ND (2013) Nucleotide precursors prevent folic acid-resistant neural tube defects in the mouse. Brain 136:2836–2841
Wong RL, Wlodarczyk BJ, Min KS, Scott ML, Kartiko S, Yu W, Merriweather MY, Vogel P, Zambrowicz BP, Finnell RH (2008) Mouse Fkbp8 activity is required to inhibit cell death and establish dorso-ventral patterning in the posterior neural tube. Hum Mol Genet 17:587–601
Lam E, Martin M, Wiederrecht G (1995) Isolation of a cDNA encoding a novel human FK506-binding protein homolog containing leucine zipper and tetratricopeptide repeat motifs. Gene 160:297–302
Pedersen KM, Finsen B, Celis JE, Jensen NA (1999) muFKBP38: a novel murine immunophilin homolog differentially expressed in Schwannoma cells and central nervous system neurons in vivo. Electrophoresis 20:249–255
Edlich F, Lucke C (2011) From cell death to viral replication: the diverse functions of the membrane-associated FKBP38. Curr Opin Pharmacol 11:348–353
Fong S, Mounkes L, Liu Y, Maibaum M, Alonzo E, Desprez PY, Thor AD, Kashani-Sabet M, Debs RJ (2003) Functional identification of distinct sets of antitumor activities mediated by the FKBP gene family. Proc Natl Acad Sci U S A 100:14253–14258
Cho A, Ko HW, Eggenschwiler JT (2008) FKBP8 cell-autonomously controls neural tube patterning through a Gli2- and Kif3a-dependent mechanism. Dev Biol 321:27–39
Bulgakov OV, Eggenschwiler JT, Hong DH, Anderson KV, Li T (2004) FKBP8 is a negative regulator of mouse sonic hedgehog signaling in neural tissues. Development 131:2149–2159
Shirane M, Ogawa M, Motoyama J, Nakayama KI (2008) Regulation of apoptosis and neurite extension by FKBP38 is required for neural tube formation in the mouse. Genes Cells 13:635–651
Murdoch JN, Copp AJ (2010) The relationship between sonic Hedgehog signaling, cilia, and neural tube defects. Birth Defects Res A Clin Mol Teratol 88:633–652
Anderson RM, Stottmann RW, Choi M, Klingensmith J (2006) Endogenous bone morphogenetic protein antagonists regulate mammalian neural crest generation and survival. Dev Dyn 235:2507–2520
Copp AJ, Greene ND, Murdoch JN (2003) The genetic basis of mammalian neurulation. Nat Rev Genet 4:784–793
Woo WM, Zhen HH, Oro AE (2012) Shh maintains dermal papilla identity and hair morphogenesis via a Noggin-Shh regulatory loop. Genes Dev 26:1235–1246
McMahon JA, Takada S, Zimmerman LB, Fan CM, Harland RM, McMahon AP (1998) Noggin-mediated antagonism of BMP signaling is required for growth and patterning of the neural tube and somite. Genes Dev 12:1438–1452
Richter A, Valdimarsdottir L, Hrafnkelsdottir HE, Runarsson JF, Omarsdottir AR, Ward-van Oostwaard D, Mummery C, Valdimarsdottir G (2014) BMP4 promotes EMT and mesodermal commitment in human embryonic stem cells via SLUG and MSX2. Stem Cells 32:636–648
Ishii M, Han J, Yen HY, Sucov HM, Chai Y, Maxson RE Jr (2005) Combined deficiencies of Msx1 and Msx2 cause impaired patterning and survival of the cranial neural crest. Development 132:4937–4950
Akizu N, Estarás C, Guerrero L, Martí E, Martínez-Balbás MA (2010) H3K27me3 regulates BMP activity in developing spinal cord. Development 137:2915–2925
Wallingford JB, Niswander LA, Shaw GM, Finnell RH (2013) The continuing challenge of understanding, preventing, and treating neural tube defects. Science 339:1222002-1–1222002-1
Harris MJ, Juriloff DM (2007) Mouse mutants with neural tube closure defects and their role in understanding human neural tube defects. Birth Defects Res A Clin Mol Teratol 79:187–210
Harris MJ, Juriloff DM (2010) An update to the list of mouse mutants with neural tube closure defects and advances toward a complete genetic perspective of neural tube closure. Birth Defects Res Part A Clin Mol Teratol 88:653–669
Eggenschwiler JT, Espinoza E, Anderson KV (2001) Rab23 is an essential negative regulator of the mouse Sonic hedgehog signalling pathway. Nature 412:194–198
Reiter JF, Skarnes WC (2006) Tectonic, a novel regulator of the Hedgehog pathway required for both activation and inhibition. Genes Dev 20:22–27
Patterson VL, Damrau C, Paudyal A, Reeve B, Grimes DT, Stewart ME, Williams DJ, Siggers P, Greenfield A, Murdoch JN (2009) Mouse hitchhiker mutants have spina bifida, dorso-ventral patterning defects and polydactyly: identification of Tulp3 as a novel negative regulator of the Sonic hedgehog pathway. Hum Mol Genet 18:1719–1739
Ybot-Gonzalez P, Gaston-Massuet C, Girdler G, Klingensmith J, Arkell R, Greene ND, Copp AJ (2007) Neural plate morphogenesis during mouse neurulation is regulated by antagonism of Bmp signaling. Development 134:3203–3211
Johnson RL, Laufer E, Riddle RD, Tabin C (1994) Ectopic expression of Sonic hedgehog alters dorsal-ventral patterning of somites. Cell 79:1165–1173
Fan CM, Tessier-Lavigne M (1994) Patterning of mammalian somites by surface ectoderm and notochord: evidence for sclerotome induction by a hedgehog homolog. Cell 79:1175–1186
Fan CM, Porter JA, Chiang C, Chang DT, Beachy PA, Tessier-Lavigne M (1995) Long-range sclerotome induction by sonic hedgehog: direct role of the amino-terminal cleavage product and modulation by the cyclic AMP signaling pathway. Cell 81:457–465
Marcelle C, Stark MR, Bronner-Fraser M (1997) Coordinate actions of BMPs, Wnts, Shh and noggin mediate patterning of the dorsal somite. Development 124:3955–3963
Ingram WJ, McCue KI, Tran TH, Hallahan AR, Wainwright BJ (2008) Sonic Hedgehog regulates Hes1 through a novel mechanism that is independent of canonical Notch pathway signaling. Oncogene 27:1489–1500
Briscoe J, Pierani A, Jessell TM, Ericson J (2000) A homeodomain protein code specifies progenitor cell identity and neuronal fate in the ventral neural tube. Cell 101:435–445
Briscoe J, Sussel L, Serup P, Hartigan-O'Connor D, Jessell TM, Rubenstein JL, Ericson J (1999) Homeobox gene Nkx2.2 and specification of neuronal identity by graded Sonic hedgehog signaling. Nature 398:622–627
Ericson J, Rashbass P, Schedl A, Brenner-Morton S, Kawakami A, van Heyningen V, Jessell TM, Briscoe J (1997) Pax6 controls progenitor cell identity and neuronal fate in response to graded Shh signaling. Cell 90:169–180
Fuccillo M, Joyner AL, Fishell G (2006) Morphogen to mitogen: the multiple roles of hedgehog signaling in vertebrate neural development. Nat Rev Neurosci 7:772–783
Lan F, Bayliss PE, Rinn JL, Whetstine JR, Wang JK, Chen S, Iwase S, Alpatov R, Issaeva I, Canaani E, Roberts TM, Chang HY, Shi Y (2007) A histone H3 lysine 27 demethylase regulates animal posterior development. Nature 449:689–694
Acknowledgments
The authors wish to thank Bill Goossens, Microscope facility at Lurie Children’s Hospital of Chicago Research Center. This work was supported by the State of Illinois Excellence in Academic Medicine award (to CSM), a Grant from the Spastic Paralysis Research Foundation of Illinois-Eastern Iowa District of Kiwanis (to CSM and DGM), and the Spina Bifida Association and Ann and Robert H. Lurie Children’s Hospital of Chicago Research Center Pilot Grant award (to CSM), and Eleanor Clarke Distinguished Developmental Neurobiology Research Scholar endowment (CSM) and in part by NIH grant HD067244 (RHF).
Conflict of interest
We declare no conflict of interests.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Tsurubuchi, T., Allender, E.V., Siddiqui, M.R. et al. A critical role of noggin in developing folate-nonresponsive NTD in Fkbp8 −/− embryos. Childs Nerv Syst 30, 1343–1353 (2014). https://doi.org/10.1007/s00381-014-2428-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-014-2428-1