Abstract
Objects
Epigenetic alterations, known as epimutations, act by deregulating gene expression. These epimutations are reversible through the action of chromatin modifiers such as DNA methylation (DNA-met) and histone deacetylases (HDAC) inhibitors. The present study evaluated the effect of 5-azacitidine (5-aza) and sodium butyrate (NaBu) as inhibitors of DNA-met and HDAC, respectively, in the expression of genes involved in apoptosis.
Methods
D54-MG, U373-MG, and T98G cell lines were exposed to 8 mM of NaBu and 12 μM of 5-aza, as well as a combination of both, for 24 h. The expression of the Bcl-2, Bak-1, Bax, Caspase-3, and Caspase-9 genes was assessed by RT-PCR.
Results
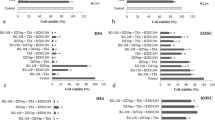
They show that the Bcl-2, Caspase-3, and Caspase-9 genes were not expressed by the U373-MG and T98G lines, and that the D54-MG line did not express Bak-1. After treatment, however, these cell lines expressed all of the genes due to the effect of 5-aza on Bak-1 in D54-MG and Caspase-9 in T98G, which suggests repression by DNA-met. Meanwhile, Bcl-2, Caspase-3, and Caspase-9 were in the U373-MG and T98G lines expressed after NaBu treatment. The effect of 5-aza induced an increase in the expression of Bax and Bcl-2, while NaBu produced a similar effect on the Bak-1 and Bax genes.
Conclusions
Results reveal that histone deacetylation is the principle mechanism for repressing these genes and that their basal expression is regulated primarily by this form of histone modification.

Similar content being viewed by others
References
Arenas-Huertero F, Recillas-Targa F (2002) Modificaciones epigenéticas de la cromatina en la generación de cáncer. Gac Méd Méx 138:547–555
Chu SH, Ma YB, Feng DF, Zhang H, Qiu JH, Zhu ZA (2012) Effect of 5-Aza-2′-deoxycytidine on SLC22A18 in glioma U251 cells. Mol Med Report 5:138–141
Fukuda H, Sano N, Muto S, Horikoshi M (2006) Simple histone acetylation plays a complex role in the regulation of gene expression. Brief Funct Gen Prot 5:190–208
Langley B, Gensert JM, Beal MF, Ratan RR (2005) Remodeling chromatin and stress resistance in the central nervous system: histone deacetylase inhibitors as novel and broadly effective neuroprotective agents. Curr Drug Targets—CNS Neurol Dis 4:41–51
Lopez-Sierra L, Esteller M (2008) Proteins that bind methylated DNA and human cancer: reading the wrong words. Br J Canc 98:1881–1885
Ma WW, Adjei AA (2009) Novel agents on the horizon for cancer therapy. Folia Histochem Cytobiol 44:111–137
Bogdanovic D, Mishra A, Medrano EE (2009) DNA methylation and methyl-CpG binding proteins: developmental requirements and function. Chromosoma 118:549–656
Kim TY, Bang YJ, Robertson KD (2006) Histone deacetylase inhibitor for cancer therapy. Epigenetics 1:14–23
Issa JPJ (2008) DNA methylation as a therapeutic target in cancer. Mutat Res 647:77–85
Kim T-Y, Zhong S, Fields CR, Hoon J, Robertson KD (2006) Epigenomic profiling reveals novel and frequent targets of aberrant DNA methylation-mediated silencing in malignant glioma. Cancer Res 66:7490–7501
Ashley DM, Riffkin CD, Muscat AM, Knight MK, Kaye AH, Novak U, Hwkins CJ (2005) Caspase 8 is absent or low in many exvivo gliomas. Cancer 104:1487–1496
Shundong C, Yuehua M, Delong L (2009) New clinical developments in histone deacetylase inhibitors for epigenetic therapy of cancer. J Hemat Oncol 2:1–11
Leone G, Dáló F, Zardo G, Voso MT, Nervi C (2008) Epigenetic treatment of myelodysplastic syndromes and acute myeloid leukemias. Curr Med Chem 15:1274–1287
Stresemann C, Brueckner B, Musch T, Stopper H, Lyko F (2006) Functional diversity of DNA methyltransferase inhibitors in human cancer cell lines. Cancer Res 66:2794–2800
Egler V, Korur S, Failly M, Boulay JL, Imber R, Lino MM, Merlo A (2008) Histone deacetylase inhibition and blockade of the glycolytic pathway synergistically induce glioblastoma cell death. Clin Cancer Res 14:3132–3140
Senese S, Zaragoza K, Minardi S, Muradore I, Ronzoni S, Passafaro A, Bernard L, Draetta GF, Alcalay M, Seiser C, Chiocca S (2007) Role for histone deacetylase 1 in human tumor cell proliferation. Mol Cell Biol 27:4784–4790
Joachimiak R, Kaźnica A, Drewa T (2007) Influence of sodium butyrate on hepatocellular carcinoma (hepG2) and glioblastoma (C6) cell lines in vitro. Acta Pol Pharm 64:561–563
Kim EH, Kim HS, Kim SU, Noh EJ, Lee JS, Choi KS (2005) Sodium butyrate sensitizes human glioma cells to TRAIL-mediated apoptosis through inhibition of Cdc2 and the subsequent downregulation of survivin and XIAP. Oncogene 24:6877–6889
Sawa H, Murakami H, Ohshima Y, Sugino T, Nakajyo T, Kisanuki T, Tamura Y, Satone A, Ide W, Hashimoto I, Kamada H (2001) Histone deacetylase inhibitors such as sodium butyrate and trichostatin A induce apoptosis through an increase of the bcl-2-related protein Bad. Brain Tumor Pathol 18:109–114
Eramo A, Pallini R, Lotti F, Sette G, Patti M, Bartucci M, Ricci-Viani SM, Stassi G, Larocca LM, Crino L, Paschle C, De Maria R (2005) Inhibition of DNA methylation sensitizes glioblastoma for tumor necrosis factor -related apoptosis-induced ligand- mediated destruction. Cancer Res 65:11469–11477
Martinez R, Setien F, Voelter C, Casado S, Quesada MP, Schackert G, Esteller M (2007) CpG island promoter hypermethylation of the pro-apoptotic gene caspase-8 is a common hallmark of relapsed glioblastoma multiforme. Carcinogenesis 28:1264–1268
Karagiannis TC, El-Osta A (2006) Clinical potential of histone deacetylase inhibitors as stand-alone therapeutics and in combination with other chemotherapeutics or radiotherapy for cancer. Epigenetics 1:121–126
Acknowledgments
The authors thank the Dirección de Investigación at the Hospital Infantil de México Federico Gómez for its support (HIM/2007/020); as well as Mexico’s National Science and Technology Council (CONACyT), through FOSISS SALUD-2012-01-181456; and Sistema Nacional de Investigadores (SNI) (Francisco Arenas-Huertero).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Solís-Paredes, M., Eguía-Aguilar, P., Chico-Ponce de León, F. et al. Epigenetic modifications in cell lines of human astrocytoma differentially regulate expression of apoptotic genes. Childs Nerv Syst 30, 123–129 (2014). https://doi.org/10.1007/s00381-013-2258-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-013-2258-6