Abstract
Daytime variation affects the tolerance of cardiomyocytes to ischemia–reperfusion injury (IRI). This study aims to evaluate the impact of time-of-day reperfusion on clinical outcomes of remote ischemic conditioning (RIC) as an adjuvant to primary percutaneous coronary intervention(PPCI) in ST-elevation myocardial infarction(STEMI) patients. A post-hoc analysis of a prospective, single-center parallel 1:1 randomized trial (RIC-STEMI) was performed. This analysis included 448 STEMI patients previously randomized to either PPCI alone (PPCI group) (n = 217) or RIC as an adjuvant to PPCI (RIC + PPCI group) (n = 231). Moreover, the sample was divided according to the time of PPCI: night-morning (22 h-11h59min) (n = 216) or afternoon (12 h-21h59min) (n = 232) groups. The primary follow-up endpoint was a composite of cardiac death and hospitalization due to heart failure. There were no significant differences in the clinical characteristics and the follow-up outcomes between groups. The afternoon period (HR = 0.474; 95% CI 0.230–0.977; p = 0.043) and RIC (HR = 0.423; 95% CI 0.195–0.917; p = 0.029) were independent predictors of the primary follow-up endpoint. An univariate analysis showed a lower frequency of primary follow-up endpoint, just in the afternoon period (10.3%vs0.9%; p = 0.002), in the RIC + PPCI group. A multivariate analysis revealed that RIC was an independent predictor of the primary follow-up endpoint in the afternoon group (HR = 0.098; 95% CI 0.012–0.785; p = 0.029), but not in the night-morning group. In addition, the afternoon period was not an independent predictor of the primary follow-up endpoint when the multivariate analysis was performed in the PPCI group. In conclusion, this study showed an important cardioprotective effect of RIC, namely in the afternoon period, suggesting that the afternoon period enhances the cardioprotection induced by RIC.
Similar content being viewed by others
Introduction
Ischemic heart disease, mainly myocardial infarction (MI), remains a leading cause of death worldwide [1]. Nowadays, reperfusion strategies promptly restore blood flow and allow the reduction of infarct size and mortality [2,3,4]. However, abrupt reperfusion can, paradoxically, cause additional damage, called ischemia–reperfusion injury (IRI), which may be responsible for up to 50% of final infarct size and contribute to heart failure (HF) development, partially compromising the beneficial effect of reperfusion [5, 6].
Remote ischemic conditioning (RIC) is a cardioprotective strategy, in which brief cycles of non-lethal ischemia and reperfusion are applied to a distant organ before, during or after a long period of myocardial ischemia [7]. The rational of RIC has been extensively reviewed and established in basic research but the results of the translational investigation are controversial and largely disappointing.
Sloth et al. (2014), enrolled 251 ST-elevation myocardial infarction (STEMI) patients and revealed a 51% decrease in all-cause mortality, non-fatal MI, stroke and HF in patients who underwent RIC [8]. White et al. (2015), included 197 STEMI patients and showed a 27% reduction in infarct size and a 19% reduction of myocardial edema, assessed by cardiac magnetic resonance (CMR), in patients who underwent RIC [9]. Eitel et al. (2015), in the LIPSIA CONDITIONING study involving 696 STEMI patients, revealed a decrease in infarct size in patients who underwent combined intrahospital RIC and post-conditioning when compared with conventional primary percutaneous coronary intervention (PPCI) group [10]. Gaspar et al. [11], in a prospective single-center randomized trial with 448 STEMI patients, showed improvement in outcomes (cardiac death or HF hospitalization) in patients undergoing RIC as an adjunct to PPCI, over a mean follow-up period of 2.1 years [11]. Nevertheless, Hausenloy et al. [12], 13), in the largest prospective multicenter study (CONDI2/ERICPPCI) with 5401 patients, did not show evidence that RIC reduced cardiac death, HF hospitalization, improved left ventricle ejection fraction (EF) [12] or reduced infarct size at six months by CMR [13].
Therefore, it remains unclear whether RIC, restricted to patients with higher risk and greater susceptibility, could be cardioprotective.
Cardiovascular diseases show diurnal variation, with a higher incidence of STEMI in the morning. Recently, Montaigne et al. (2018), revealed the influence of the daytime variation on tolerance to IRI in patients undergoing aortic valve replacement surgery. This study concluded that surgeries performed in the afternoon were associated with better clinical outcomes compared to those carried out in the morning [14].
Up till now, the influence of daytime variation in IRI and in clinical results of RIC has never been raised. Therefore, our aim was to assess whether RIC, as an adjuvant to PPCI, performed in STEMI patients in the afternoon period had different clinical results.
Methods
Study design
This study consists of a post-hoc analysis of the RIC-STEMI study (NCT02313961) [11], a single center, open label, parallel 1:1 randomized controlled trial.
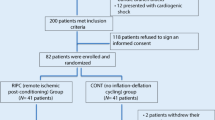
This trial aimed to assess the superiority of RIC (3 cycles of inflation and deflation of a left lower limb cuff, for 5 min each) over PPCI in all-comer patients presenting with STEMI between March 2013 and December 2015. Eligible patients were at least 18 years old and were admitted to the emergency department of Braga Hospital with putative STEMI [15]. Exclusion criteria were cardiogenic shock, defined by a systolic blood pressure lower than 90 mmHg and evidence of tissue hypoperfusion; post-cardiac arrest status; the need for mechanical ventilation; known peripheral arterial disease; evidence of lower limb ischemia and recent MI (within the last 30 days).
To perform this study the authors consulted the database of RIC-STEMI study. This analysis included 448 STEMI patients previously randomized to either PPCI alone (PPCI group) (n = 217) or RIC as an adjuvant to PPCI (RIC + PPCI group) (n = 231). To assess the effect of daytime variation on RIC clinical results, the sample was divided according to the time of PPCI in night-morning (22 h–11 h59 min) or afternoon (12 h–21 h59 min) periods.
The cardioprotective effect of RIC was compared within isolated PPCI, according to the period of the day in which it was performed. Figure 1 shows the study flow diagram.
The admission echocardiography was performed in the first 24 h of hospitalization. The median month to measure the follow-up ejection fraction was 10 (IQR 1.8) months.
Study endpoints
The primary follow-up endpoint was a composite of cardiac mortality and hospitalization due to HF.
The minimum predefined follow-up time was 12 months.
Secondary endpoints were follow-up EF (estimated by Simpson’s biplane method), major adverse cardiovascular and cerebrovascular events on follow-up (MACCE), hospitalization due to HF, all-cause mortality, and cardiac mortality.
Hospitalization due to HF included readmissions due to acute or chronically decompensated HF, planned implantation of cardioverter-defibrillator (ICD) or cardiac resynchronization therapy device (CRT).
Cardiac death was defined as natural death due to cardiac causes and required evidence of acutely decompensated HF or sudden cardiac arrest. Patients found dead at home were not considered.
MACCE consisted of the composite of total death, MI, stroke (persistent neurological deficit) and target vessel revascularization.
Ethical procedures
This study was approved by Braga Hospital Ethical Committee and met the criteria established by the Declaration of Helsinki 1964 as revised in 2013 and the International Conference of Harmonisation Guidelines for Good Clinical Practice. All patients enrolled gave witness oral consent and written informed consent after clinical stabilization.
Statistical analysis
Statistical analysis was performed using IBM® SPSS®, version 27.
The normality of the distribution of continuous variables was evaluated through the Shapiro–Wilk’s test and histogram analysis. Continuous variables were described by median (Mdn) and interquartile range (IQ) since normality was not found in any of them. Categorical variables were described by absolute and relative frequencies (%).
The Mann–Whitney test was performed to compare continuous variables. The Chi-square test was used to compare categorical variables, although if the percentage of cells with < 5 expected counts were greater than 20%, Fisher’s exact test was preferred. Results with statistical significance were considered whenever the p value < 0.05.
If two variables were dichotomous, Phi (φ) was presented, as measured of effect size, considering the small, medium, or large effect for absolute values close to 0.10, 0.30 and 0.50, respectively.
Cox’s proportional hazards model with forward’s method was employed to evaluate the impact of RIC on the primary follow-up endpoint according to the period of the day in which PPCI was performed. Results are reported as hazard ratio (HR) and with a 95% of confidence interval (CI).
Results
The RIC-STEMI study enrolled 448 STEMI patients, 217 of which were randomly allocated to the PPCI group and 231 to the RIC + PPCI group. The baseline characteristics and clinical presentation of both groups were previously published [11]. To highlight that a higher concentration of haemoglobin in the RIC + PPCI group (RIC + PPCI group 14.2 [13–15.4] g/dl vs PPCI group:13.9 [12.8–15] g/dL; p = 0.035) was the only statistical difference between groups.
Regarding the time of PPCI, no significant differences were detected. PPCI was performed in the afternoon period in 53% (n = 116) of the PPCI group and 50% (n = 116) of the RIC + PPCI group.
In this post-hoc analysis of the RIC-STEMI study, the study population was divided into 2 groups, according to the time of PPCI, and Table 1 shows baseline characteristics as well as clinical presentation of night-morning and afternoon groups.
In addition to the balanced number of patients randomized to perform RIC, both groups had statistically similar characteristics, except for gender distribution, with a higher frequency of female gender in the afternoon group (24% vs 16%, p = 0.04).
During hospitalization, no divergence was found for 48-h troponin I level area under the curve (AUC) (trapezoid rule) between groups (Night-morning group: 43 [22–77] ng/mL vs afternoon group: 38 [17–73] ng/mL; p = 0.12). Regarding clinical presentation, anterior MI was reported in 44% of both groups, admission EF lower than 35% was observed in 19% of both groups and 77% of the night-morning group and in 79% of the afternoon group had occluded artery (TIMI 0) at the time of PPCI.
After confirming the homogeneity of groups, we evaluated the relation of the time of PPCI with follow-up results and no statistical difference was found (Table 2). There was no difference in follow-up EF (night-morning group: 54 [11] % vs afternoon group 55 [11] %, p = 0.26) between both groups.
Nevertheless, a survival analysis was performed using the Cox regression model by the forward method, to verify whether the afternoon period is an independent predictor of the primary follow-up endpoint.
Table 3 shows the regression model. The last stage of the model was statistically significant [X2 (6) = 85,688; p < 0,001] and revealed that the afternoon period was an independent predictor of the lower primary follow-up endpoint (HR = 0.474; 95% CI 0.230–0.977; p = 0.043). In addition, admission EF less than 35%, HF during hospitalization, 48 h Troponin I level AUC, admission creatinine and RIC were also independent predictors.
Finally, in this post-hoc analysis, there was a statistically lower frequency of hospitalization due to HF (afternoon group: 0.9% vs 9.5%, p = 0.003) and primary follow-up endpoint (afternoon group: 0.9% vs 10.3%, p = 0.002) in the RIC + PPCI group only in the afternoon period (Table 4).
Considering secondary follow-up endpoints, hospitalizations due to HF were also less frequent in the RIC + PPCI group only in the afternoon period (afternoon group: 0.9% vs 10%, p = 0.003).
There were no significant differences between PPCI and RIC + PPCI group, in both periods, regarding MACCE, MI, cardiac mortality and total mortality.
A survival analysis of the afternoon period (Table 5) was performed using the Cox regression model through the forward method and confirmed, in the regression model [X2(5) = 51,555; p < 0,001], that RIC remains a statistically significant independent predictor of primary follow-up endpoint (HR = 0.098; 95% CI 0.012–0.785; p = 0.029).
However, in the survival analysis of the night-morning group (Table 6), RIC was not an independent predictor of the primary follow-up endpoint.
These results were supported by the Kaplan Meier curves which showed that RIC had a significant impact on time without a primary follow-up endpoint in STEMI patients included in RIC-STEMI study (Fig. 2A) (p value LogRank test = 0,08) and in our afternoon group (Fig. 2B) (p value LogRank test = 0,002), but not in a night-morning group (Fig. 2C).
Finally, a survival analysis of the PPCI group was performed and the afternoon period was not an independent predictor of the primary follow-up endpoint (Table 7).
Discussion
In opposition to previous studies [8,9,10,11], Hausenloy et al. in CONDI2/ERICPPCI [12], the largest prospective multicenter study, did not reveal beneficial effects of RIC in reducing cardiac mortality or hospitalization due to HF. Despite its great value, this study had some particularities, which may have influenced the results. To point out: a short follow-up time (12 months), inadequate for the detection of HF events secondary to ventricular remodeling and a median ischemia time of less than 3 h. Considering that patients with anterior MI and a total ischemia time between 3 to 8 h benefited the most from RIC [16], the shorter time may have compromised the RIC impact. At the very least, these results reinforce an already known truth, that RIC is not effective under all circumstances [17]. Several factors influence the benefit of RIC, namely: age, history of ischemic coronary disease, total ischemia time, infarct size and localization, occluded artery at the time of RIC, comedication and comorbidity [16, 18]. There may be even other factors that have not been described yet, with a possible impact on the response to RIC.
This study is a post-hoc analysis of the RIC-STEMI study [11], a single-center randomized controlled trial. A previous pre-specified sub-analysis had already exposed that RIC only had a benefit in populations with anterior MI, total ischemia time greater than 3 h and occluded artery at the time of RIC. Our study had similar groups with well-balanced characteristics, except for a higher proportion of women in the afternoon group; however, no justification was discovered to explain this finding. No differences were found regarding age, anterior MI, total ischemia time and the proportion of occluded artery at the time of RIC. The analysis of the time-of-day reperfusion impact on the clinical results in STEMI patients revealed a much lower frequency of primary follow-up endpoint when RIC was performed as an adjuvant to PPCI in the afternoon. These results occurred without differences in 48 h troponin I levels AUC. Indeed, Gaspar et al. (2018) and Sloth et al. (2014) already failed to show reduced infarct size through troponin levels despite improvement in clinical results [8, 11]. Perhaps this biomarker is not the most accurate way to access infarct size, and an imagological evaluation should be preferred as in LIPSIA CONDITIONING STUDY [10]. Regarding the lack of differences in follow-up ejection fraction, this could be due to the fact that some participants died before doing the follow-up echocardiogram which could have had an impact on the study results.
A relation between MI and day-time variation was previously reported considering the higher incidence and larger dimensions of MI in the early morning [19,20,21,22,23]. These findings could be explained by the endogenous circadian fluctuation of thrombolytic activity, platelet aggregation and adrenergic activation, which some studies describe as being increased in the early morning [24,25,26]. In addition, Montaigne et al., also showed a relation between daytime variation and cardiomyocyte tolerance to IRI, higher in the afternoon, in patients who underwent aortic valve replacement surgery. This finding was concomitant with transcriptional alterations in the expression of the circadian gene of Rev-Erbα [14]. Furthermore, in two analyses of STEMI patients that underwent manual thrombus aspiration [21] and PPCI [27] smaller infarct size and better clinical results were found when symptoms began in the afternoon.
Our study points to the existence of a biorhythm for the cardioprotective effect of RIC since RIC was an independent predictor of the primary follow-up endpoint just in the afternoon group (HR = 0.098; 95% CI 0.012–0.785; p = 0.029). The afternoon period was not an independent predictor of the primary follow-up endpoint in the PPCI group.
Accordingly, our study revealed an important cardioprotective effect of RIC, namely in the afternoon period, suggesting that the afternoon period enhances the cardioprotection induced by RIC.
The authors consider that is still unclear whether RIC, restricted to high-risk patients, is cardioprotective and future clinical investigation should take into consideration the previously pointed features.
Therefore, larger studies are necessary to confirm these results and it would also be interesting to perform this sub-analysis in the studies previously published.
Limitations
This study has limitations related to the fact that it was a post-hoc analysis of a single-center randomized controlled trial with a limited sample, which compromises the subgroup analysis. In this line, the number of events evaluated in the follow-up was also reduced, despite a mean follow-up of 2.1 years.
Abbreviations
- ACEi/ARB:
-
Angiotensin converter enzyme Inhibitor/Angiotensin II receptor blocker
- AUC:
-
Area under curve
- CABG:
-
Coronary artery bypass grafting
- CI:
-
Confidence interval
- CMR:
-
Cardiac magnetic resonance
- CRT:
-
Cardiac resynchronization therapy device
- EF:
-
Ejection fraction
- HF:
-
Heart failure
- HR:
-
Hazard ratio
- ICD:
-
Cardioverter-defibrillator
- IR:
-
Interquartile range
- IRI:
-
Ischemia–reperfusion injury
- MACCE:
-
Major adverse cardiovascular and cerebrovascular events on follow-up
- Mdn:
-
Median
- MI:
-
Myocardial infarction
- PCI:
-
Percutaneous coronary intervention
- PPCI:
-
Primary percutaneous coronary intervention
- RIC:
-
Remote ischemic conditioning
- STEMI:
-
ST-elevation myocardial infarction
References
Townsend N, Nichols M, Scarborough P, Rayner M (2015) Cardiovascular disease in Europe-epidemiological update. Eur Hear J 36:2696–2705
De Luca G, Suryapranata H, Ottervanger JP, Antman EM (2004) Time delay to treatment and mortality in primary angioplasty for acute myocardial infarction: every minute of delay counts. Circulation 109:1223–1225
Heusch G, Gersh BJ (2017) The pathophysiology of acute myocardial infarction and strategies of protection beyond reperfusion: a continual challenge. Eur Hear J 38:774–784
Jernberg T, Johanson P, Held C, Svennblad B, Lindbäck J, Wallentin L (2011) Association between adoption of evidence-based treatment and survival for patients with ST-elevation myocardial infarction. JAMA - J Am Med Assoc 305:1677–1684
Yellon DM, Hausenloy DJ (2007) Myocardial reperfusion injury. N Engl J Med 357:1121–1135
Hausenloy DJ, Yellon DM (2013) Myocardial ischemia-reperfusion injury: a neglected therapeutic target. J Clin Invest 123:92–100
Chong J, Bulluck H, Yap EP, Ho AF, Boisvert WA, Hausenloy DJ (2018) Remote ischemic conditioning in ST-segment elevation myocardial infarction–an update. Cond Med 1:13–22
Sloth AD, Schmidt MR, Munk K, Kharbanda RK, Redington AN, Schmidt M, Pedersen L, Sorensen HT, BOTKER HE, (2014) Improved long-term clinical outcomes in patients with ST-elevation myocardial infarction undergoing remote ischaemic conditioning as an adjunct to primary percutaneous coronary intervention. Eur Heart J 35:168–175
White SK, Frohlich GM, Sado DM, Maestrini V, Fontana M, Treibel TA, Tehrani S, Flett AS, Meier P, Ariti C, Davies JR, Moon JC, Yellon DM, Hausenloy H (2015) Remote ischemic conditioning reduces myocardial infarct size and edema in patients with ST-segment elevation myocardial infarction. JACC Cardiovasc Interv 8:178–188
Eitel I, Stiermaier T, Rommel KP, Fuernau G, Sandri M, Mangner N, Linke A, Erbs S, Lurz P, Boudriot E, Meinhard M, Desch SGA, Thiele H (2015) Cardioprotection by combined intrahospital remote ischaemic perconditioning and postconditioning in ST-elevation myocardial infarction: the randomized LIPSIA CONDITIONING trial. Eur Heart J 36:3049–3057
Gaspar A, Lourenço AP, Pereira MÁ, Azevedo P, Roncon-Albuquerque R, Marques J, Leite-Moreira AF (2018) Randomized controlled trial of remote ischaemic conditioning in ST-elevation myocardial infarction as adjuvant to primary angioplasty (RIC-STEMI). Basic Res Cardiol 113:14
Hausenloy DJ, Kharbanda RK, Møller UK, Ramlall M, Aarøe J, Butler R, Bulluck H, Clayton T, Dana A, Dodd M, Engstrom T, Evans R, Lassen JF, Christensen EF, Garcia-Ruiz JM, Gorog DA, Hjort J, Houghton RF, Ibanez B, Knight R, Lippert FK, Lønborg JT, Maeng M, Milasinovic D, More R, Nicholas JM, Jensen LO, Radovanovic N, Rakhit RD, Ravkilde J, Ryding AD, Schmidt MR, Riddervold IS, Sørensen HT, Stankovic G, Varma M, Webb I, Terkelsen CJ, Greenwood JP, Yellon DM, Bøtker HE, on behalf of theCONDI-2/ERIC-PPCI Investigators (2019) Effect of remote ischaemic conditioning on clinical outcomes in patients with acute myocardial infarction (CONDI-2/ERIC-PPCI): a single-blind randomised controlled trial. Lancet 394:1415–1424
Francis R, Chong J, Ramlall M, Bucciarelli-Ducci C, Clayton T, Dodd M, Engstrøm T, Evans R, Ferreira V, Fontana M, Greenwood JP, Kharbanda RK, Kim WY, Kotecha T, Lønborg JT, Mathur A, Møller UK, Moon J, Perkins A, Rakhit RD, Yellon DM, Bøtker HE, Bulluck H, Hausenloy DJ (2021) Effect of remote ischaemic conditioning on infarct size and remodelling in ST-segment elevation myocardial infarction patients: The CONDI-2/ERIC-PPCI CMR substudy. Basic Res Cardiol 116:59
Montaigne D, Marechal X, Modine T, Coisne A, Mouton S, Fayad G, Ninni S, Klein C, Ortmans S, Seunes C, Potelle C, Berthier A, Gheeraert C, Piveteau C, Deprez R, Eeckhoute J, Duez H, Lacroix D, Deprez B, Jegou B, Koussa M, Edme JL, Lefebvre P, Staels B (2018) Daytime variation of perioperative myocardial injury in cardiac surgery and its prevention by Rev-Erbα antagonism: a single-centre propensity-matched cohort study and a randomised study. Lancet 391:59–69
Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, Caforio ALP, Crea P, Goudevenos JA, Halvorsen S, Hindricks G, Kastrati A, Lenzen MJ, Prescott E, Roffi M, Valgimigli M, Varenhorst C, Vranckx P, Widimský P (2018) 2017 ESC guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J 39:119–177
Bell RM, Bøtker HE, Carr RD, Davidson SM, Downey JM, Dutka DP, Heusch G, Ibanez B, Macallister R, Stoppe C, Ovize M, Redington A, Walker JM, Yellon DM (2016) 9th Hatter Biannual Meeting: position document on ischaemia/reperfusion injury, conditioning and the ten commandments of cardioprotection. Basic Res Cardiol 111:41
Hausenloy D, Ntsekhe M, Yellon DM (2020) A future for remote ischaemic conditioning in high risk patients. Basic Res Cardiol 115:35
Ferdinandy P, Hausenloy DJ, Heusch G, Baxter GF, Schulz R (2014) Interaction of risk factors, comorbidities, and comedications with ischemia/reperfusion injury and cardioprotection by preconditioning, postconditioning, and remote conditioning. Pharmacol rev 66:1142–1174
Suárez-Barrientos A, López-Romero P, Vivas D, Castro-Ferreira F, Núñez-Gil I, Franco E, Núñez-Gil I, Franco E, Ruiz-Mateos B, García-Rubira JC, Fernández-Ortiz A, Macaya C, Ibanez B (2011) Circadian variations of infarct size in acute myocardial infarction. Heart 97:970–976
Fournier S, Eeckhout E, Mangiacapra F, Trana C, Lauriers N, Beggah AT, Monney P, Cook S, Bardy D, Vogt P, Muller O (2012) Circadian variations of ischemic burden among patients with myocardial infarction undergoing primary percutaneous coronary intervention. Am Heart J 163:208–213
Fournier S, Muller O, Benedetto U, Roffi M, Pilgrim T, Eberli FR, Rickli H, Radovanovic D, Erne P, Cook S, Noble S, Fesselet R, Zuffi A, Degrauwe S, Masci P, Windecker S, Eeckhout E, Iglesias JF (2018) Circadian dependence of manual thrombus aspiration benefit in patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Clin Res Cardiol 107:338–346
Reiter R, Swingen C, Moore L, Henry TD, Traverse JH (2012) Circadian dependence of infarct size and left ventricular function after ST elevation myocardial infarction. Circ Res 110:105–110
Bulluck H, Nicholas J, Crimi G, White SK, Ludman AJ, Pica S, Raineri C, Cabrera-Fuentes HA, Yellon D, Rodriguez-Palomares J, Garcia-Dorado D, Hausenloy DJ (2017) Circadian variation in acute myocardial infarct size assessed by cardiovascular magnetic resonance in reperfused STEMI patients. Int J Cardiol 230:149–154
Tofler GH, Brezinski D, Schafer AI, Czeisler CA, Ritherford JD (1987) Concurrent morning increase in platelet aggregability and the risk of myocardial infarction and sudden cardiac death. N Engl J Med 316:1514–1518
Petralito A, Mangiafico R, Gibiino S, Cuffari MF, Fiore C (1982) Daily modifications of plasma fibrinogen platelets aggregation, Howell’s time, PTT, TT, and antithrombin II in normal subjects and in patients with vascular disease. Chronobiologia 9:195–201
Scheer FAJL, Michelson AD, Frelinger AL, Evoniuk H, Kelly EE, McCarthy M, Doamekpor LA, Barnard MR, Shea SA (2011) The human endogenous circadian system causes greatest platelet activation during the biological morning independent of behaviors. PLoS ONE 6:e24549
De Luca G, Suryapranata H, Ottervanger JP, Vant Hof AWJ, Hoorntje JCA, Gosselink ATM, Dambriknk JHE, Zijlstra F, Boer MJ (2005) Circadian variation in myocardial perfusion and mortality in patients with ST-segment elevation myocardial infarction treated by primary angioplasty. Am Heart J 150:1185–1189
Acknowledgements
We thank all participants, all nurses and interventional cardiologists engaged in this study for their invaluable assistance.
Funding
Open access funding provided by FCT|FCCN (b-on). The RIC-STEMI study was supported by the Portuguese Foundation for Science and Technology (Projects PEst-C/SAU/UI0051/2011 and EXCL/BIM-MEC/0055/2012) through the Cardiovascular R&D Unit, by the European Commission Grant FP7-Health-2010 (MEDIA- 261,409) and by Project DOCnet (NORTE-01-0145-FEDER-000003), supported by Norte Portugal Regional Operational Programme (NORTE 2020), under the PORTUGAL 2020 Partnership Agreement, through the European Regional Development Fund (ERDF). European Structural and Investment Funds (ESIF), under Lisbon Portugal Regional Operational Programme and National Funds through Foundation for Science and Technology under project POCI-01-0145-FEDER-016385. The RIC-STEMI was also supported by a grant from “José de Mello Saúde”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest or disclosure.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pires, C.M., Lamas, D., Gaspar, A. et al. The impact of time-of-day reperfusion on remote ischemic conditioning in ST-elevation myocardial infarction: a RIC-STEMI substudy. Heart Vessels 38, 909–918 (2023). https://doi.org/10.1007/s00380-023-02247-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-023-02247-8