Abstract
Background
Total anomalous pulmonary venous connection (TAPVC) is a rare congenital heart disease of newborns characterized by impaired left ventricle growth and diastolic dysfunction. We hypothesized that the patients with TAPVC reduced blood flow into the left heart prenatally could affect left atrium (LA) not just growth but function. We compared the age-related changes in LA deformation using two-dimensional speckle-tracking echocardiography (2DSTE) in Patients with TAPVC.
Method
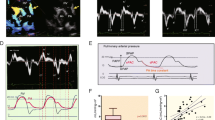
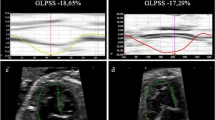
This single-center, retrospective cohort study was conducted on consecutive isolated TAPVC patients who underwent neonatal surgery between January 1, 2009 and January 1, 2022. The LA datasets in TAPVC patients were analyzed before surgery (n = 28) and follow-ups at 1−2 (n = 24) and 5−7 years of age (n = 13) and compared with those of age-matched healthy controls (January 2009−2022). The LA strain (ε), indicating LA function, was analyzed using QLAB represented by reservoir (εR), conduit (εCD), and contractile (εCT) strains. LA pressure was evaluated by periodic follow-up catheterization after repair.
Results
Compared to the controls, the TAPVC patients had significantly smaller LA maximum volume preoperatively, and with age, the LA maximal volumes reached normal levels, while the LA minimal volumes were larger. All 2DSTE-determined LA strains showed significant reductions at all time points in the TAPVC group compared to those in the control (median εR, εCD, and εCT; before surgery: 17.0% vs. 26.0%, 12.9% vs. 15.9%, and 6.3% vs. 10.4%; follow-up at 1−2 years: 30.0% vs. 45.7%, 23.2% vs. 29.6%, and 6.1% vs. 16.3%; follow-up at 5−7 years: 31.2% vs. 43.1%, 25.0% vs. 31.2%, and 5.2% vs. 10.8%, respectively; p < 0.05). Only εCT did not represented a significant change over time even though after correction of blood flow (median εCT: 6.0% → 5.9%). Patients with pulmonary venous obstruction (PVO) at birth showed significantly decreased εR and εCD and higher LA pressure compared to those without PVO.
Conclusion
This study showed that nevertheless maximum volume of LA was recovered within the normal range, reduced LA strains, especially contractile function lasted from birth even after repair in Patients with TAPVC.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, Dr Kiyohiro Takigiku, upon reasonable request.
Abbreviations
- TAPVC:
-
Total anomalous pulmonary venous connection
- LA:
-
Left atrial; 2DSTE, two-dimensional speckle-tracking echocardiography
- εR:
-
Reservoir function; εCD, conduit function
- εCT:
-
Contractile function
- PVO:
-
Pulmonary venous obstruction
- LV:
-
Left ventricle
- HLHS:
-
Hypoplastic left heart syndrome
- 2DE:
-
Two-dimensional echocardiography
- LVEF:
-
Left ventricle ejection fraction
- E:
-
Peak early diastolic mitral inflow velocity
- A:
-
Peak atrial filling velocity during late diastole
- IVS:
-
Inter-ventricular septum
- e’:
-
Peak early diastolic tissue Doppler velocity at the mitral annulus
- BSA:
-
Body surface area
- RT3DE:
-
Real-time three-dimensional echocardiography
- ICC:
-
Intraclass correlation coefficient
- AF:
-
Atrial fibrillation
- HFpEF:
-
Heart failure with preserved ejection fraction
- EACVI:
-
European Association of Cardiovascular Imaging
References
Karamlou T, Gurofsky R, Sukhni EA, Coles JG, Williams WG, Caldarone CA, Arsdell GSV, McCrindle BW (2007) Factors associated with mortality and reoperation in 377 children with total anomalous pulmonary venous connection. Circulation 115(12):1591–1598
Talwar S, Arora Y, Gupta SK, Kothari SS, Ramakrishnan S, Saxena A, Choudhary SK (2019) Total anomalous pulmonary venous connection beyond the first decade of life. World J Pediatr Congenit Heart Surg 10(2):185–191
McBride MG, Kirshbom PM, Gaynor JW, Ittenbach RF, Wernovsky G, Clancy RR, Flynn TB, Hartman DM, Spray TL, Tanel RE, Santiago MC, Paridon SM (2007) Late cardiopulmonary and musculoskeletal exercise performance after repair for total anomalous pulmonary venous connection during infancy. J Thorac Cardiovasc Surg 133(6):1533–1539
Paridon SM, Sullivan NM, Schneider J, Pinsky WW (1993) Cardiopulmonary performance at rest and exercise after repair of total anomalous pulmonary venous connection. Am J Cardiol 72(18):1444–1447
Rosenquist GC, Kelly JL, Chandra R, Ruckman RN, Galioto FM, Midgley FM, Scott LP (1985) Small left atrium and change in contour of the ventricular septum in total anomalous pulmonary venous connection: a morphometric analysis of 22 infant hearts. Am J Cardiol 55(6):777–782
Mathew R, Thilenius OG, Replogle RL, Arcilla RA (1977) Cardiac function in total anomalous pulmonary venous return before and after surgery. Circulation 55(2):361–370
Bălgrădean M, Cinteză E, Cîrstoveanu C, Enculescu A, Pleşca D (2013) Abnormalities in embryological development in total anomalous pulmonary venous connection. A case report. Rom J Morphol Embryol 54(3):635–637
Lima CO, Valdes-Cruz LM, Allen HD, Horowitz S, Sahn DJ, Goldberg SJ, Barron JV, Grenadier E (1983) Prognostic value of left ventricular size measured by echocardiography in infants with total anomalous pulmonary venous drainage. Am J Cardiol 51(7):1155–1159
Gobergs R, Salputra E, Lubaua I (2016) Hypoplastic left heart syndrome: a review. Acta Med Litu 23(2):86–98
Marcondes LD, Galati JC, Jones BO, Konstantinov IE, d’Udekem Y, Brizard CP, Cheung MM (2014) Abnormal left ventricular diastolic function at late follow-up after repair of total anomalous pulmonary venous drainage: the impact of altered ventricular loading in utero. J Thorac Cardiovasc Surg 148(1):238–244
Nakamura Y, Hoashi T, Nakata T, Shimada M, Ozawa H, Kurosaki K, Ichikawa H (2019) Left ventricular function after repair of totally anomalous pulmonary venous connection. Ann Thorac Surg 107(1):151–156
Sun BJ, Park J-H (2021) Echocardiographic measurement of left atrial strain- a key requirement in clinical practice. Circ J 86(1):6–13
Kutty S, Padiyath A, Li L, Peng Q, Rangamani S, Schuster A, Danford DA (2013) Functional maturation of left and right atrial systolic and diastolic performance in infants, children, and adolescents. J Am Soc Echocardiogr 26(4):398-409.e392
Ghelani SJ, Brown DW, Kuebler JD, Perrin D, Shakti D, Williams DN, Marx GR, Colan SD, Geva T, Harrild DM (2018) Left Atrial Volumes and strain in healthy children measured by three-dimensional echocardiography: normal values and maturational changes. J Am Soc Echocardiogr 31(2):187-193.e181
Cantinotti M, Scalese M, Giordano R, Franchi E, Assanta N, Molinaro S, Iervasi G, Santoro G, Koestenberger M, Kutty S (2019) Left and right atrial strain in healthy caucasian children by two-dimensional speckle-tracking echocardiography. J Am Soc Echocardiogr 32(1):165-168.e3
White BR, Ho DY, Faerber JA, Katcoff H, Glatz AC, Mascio CE, Stephens P Jr, Cohen MS (2019) Repair of total anomalous pulmonary venous connection: risk factors for postoperative obstruction. Ann Thorac Surg 108(1):122–129
Haycock GB, Schwartz GJ, Wisotsky DH (1978) Geometric method for measuring body surface area: a height-weight formula validated in infants, children, and adults. J Pediatr 93(1):62–66
Lopez L, Colan SD, Frommelt PC, Ensing GJ, Kendall K, Younoszai AK, Lai WW, Geva T (2010) Recommendations for quantification methods during the performance of a pediatric echocardiogram: a report from the pediatric measurements writing group of the American society of echocardiography pediatric and congenital heart disease council. J Am Soc Echocardiogr 23(5):465–495
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, Flachskampf FA, Foster E, Goldstein SA, Kuznetsova T, Lancellotti P, Muraru D, Picard MH, Rietzschel ER, Rudski L, Spencer KT, Tsang W, Voigt JU (2015) Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American society of echocardiography and the European association of cardiovascular imaging. J Am Soc Echocardiogr 28(1):1-39.e14
Kanda Y (2013) Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 48(3):452–458
Grotenhuis HB, Cifra B, Mertens LL, Riessenkampff E, Manlhiot C, Seed M, Yoo S-J, Grosse-Wortmann L (2018) Left ventricular remodelling in long-term survivors after the arterial switch operation for transposition of the great arteries. Eur Heart J Cardiovasc Imaging 20(1):101–107
Mondillo S, Cameli M, Caputo ML, Lisi M, Palmerini E, Padeletti M, Ballo P (2011) Early detection of left atrial strain abnormalities by speckle-tracking in hypertensive and diabetic patients with normal left atrial size. J Am Soc Echocardiogr 24(8):898–908
Cameli M, Lisi M, Focardi M, Reccia R, Natali BM, Sparla S, Mondillo S (2012) Left atrial deformation analysis by speckle tracking echocardiography for prediction of cardiovascular outcomes. Am J Cardiol 110(2):264–269
Smiseth OA, Baron T, Marino PN, Marwick TH, Flachskampf FA (2021) Imaging of the left atrium: pathophysiology insights and clinical utility. Eur Heart J Cardiovasc Imaging 23(1):2–13
Wakami K, Ohte N, Asada K, Fukuta H, Goto T, Mukai S, Narita H, Kimura G (2009) Correlation between left ventricular end-diastolic pressure and peak left atrial wall strain during left ventricular systole. J Am Soc Echocardiogr 22(7):847–851
Cameli M, Lisi M, Mondillo S, Padeletti M, Ballo P, Tsioulpas C, Bernazzali S, Maccherini M (2010) Left atrial longitudinal strain by speckle tracking echocardiography correlates well with left ventricular filling pressures in patients with heart failure. Cardiovasc Ultrasound 8:14
Hirose T, Kawasaki M, Tanaka R, Ono K, Watanabe T, Iwama M, Noda T, Watanabe S, Takemura G, Minatoguchi S (2012) Left atrial function assessed by speckle tracking echocardiography as a predictor of new-onset non-valvular atrial fibrillation: results from a prospective study in 580 adults. Eur Heart J Cardiovasc Imaging 13(3):243–250
Yasuda R, Murata M, Roberts R, Tokuda H, Minakata Y, Suzuki K, Tsuruta H, Kimura T, Nishiyama N, Fukumoto K, Aizawa Y, Tanimoto K, Takatsuki S, Abe T, Fukuda K (2015) Left atrial strain is a powerful predictor of atrial fibrillation recurrence after catheter ablation: study of a heterogeneous population with sinus rhythm or atrial fibrillation. Eur Heart J Cardiovasc Imaging 16(9):1008–1014
Inoue K, Khan FH, Remme EW, Ohte N, García-Izquierdo E, Chetrit M, Moñivas-Palomero V, Mingo-Santos S, Andersen ØS, Gude E, Andreassen AK, Wang TKM, Kikuchi S, Stugaard M, Ha JW, Klein AL, Nagueh SF, Smiseth OA (2021) Determinants of left atrial reservoir and pump strain and use of atrial strain for evaluation of left ventricular filling pressure. Eur Heart J Cardiovasc Imaging 23(1):61–70
Smiseth OA, Morris DA, Cardim N, Cikes M, Delgado V, Donal E, Flachskampf FA, Galderisi M, Gerber BL, Gimelli A, Klein AL, Knuuti J, Lancellotti P, Mascherbauer J, Milicic D, Seferovic P, Solomon S, Edvardsen T, Popescu BA, Committee RTdwrbmotESD (2021) Multimodality imaging in patients with heart failure and preserved ejection fraction: an expert consensus document of the European association of cardiovascular imaging. Eur Heart J Cardiovasc Imaging 23(2):e34–e61
Morris DA, Belyavskiy E, Aravind-Kumar R, Kropf M, Frydas A, Braunauer K, Marquez E, Krisper M, Lindhorst R, Osmanoglou E, Boldt LH, Blaschke F, Haverkamp W, Tschöpe C, Edelmann F, Pieske B, Pieske-Kraigher E (2018) Potential usefulness and clinical relevance of adding left atrial strain to left atrial volume index in the detection of left ventricular diastolic dysfunction. JACC Cardiovasc Imaging 11(10):1405–1415
Morris DA, Takeuchi M, Krisper M, Köhncke C, Bekfani T, Carstensen T, Hassfeld S, Dorenkamp M, Otani K, Takigiku K, Izumi C, Yuda S, Sakata K, Ohte N, Tanabe K, Osmanoglou E, Kühnle Y, Düngen HD, Nakatani S, Otsuji Y, Haverkamp W, Boldt LH (2015) Normal values and clinical relevance of left atrial myocardial function analysed by speckle-tracking echocardiography: multicentre study. Eur Heart J Cardiovasc Imaging 16(4):364–372
Santos AB, Kraigher-Krainer E, Gupta DK, Claggett B, Zile MR, Pieske B, Voors AA, Lefkowitz M, Bransford T, Shi V, Packer M, McMurray JJ, Shah AM, Solomon SD (2014) Impaired left atrial function in heart failure with preserved ejection fraction. Eur J Heart Fail 16(10):1096–1103
Sakaguchi E, Yamada A, Naruse H, Hattori H, Nishimura H, Kawai H, Muramatsu T, Ishii J, Hata T, Saito K, Izawa H (2022) Long-term prognostic value of changes in left ventricular global longitudinal strain in patients with heart failure with preserved ejection fraction. Heart Vessels. https://doi.org/10.1007/s00380-022-02211-y
van Woerden G, van Veldhuisen DJ, Gorter TM, Willems TP, van Empel VPM, Peters A, Pundziute G, den Akker op JW, Rienstra M, Westenbrink BD (2022) The clinical and prognostic value of late gadolinium enhancement imaging in heart failure with mid-range and preserved ejection fraction. Heart Vessels 37(2):273–281
Santos AB, Roca GQ, Claggett B, Sweitzer NK, Shah SJ, Anand IS, Fang JC, Zile MR, Pitt B, Solomon SD, Shah AM (2016) Prognostic relevance of left atrial dysfunction in heart failure with preserved ejection fraction. Circ Heart Fail 9(4):e002763
Acknowledgements
We thank Editage (www.editage.jp) for editing a draft of this manuscript.
Funding
The authors have no funding concerning this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Numata, R., Takigiku, K., Obinata, H. et al. Assessment of left atrial deformation in patients with total anomalous pulmonary venous connection by two-dimensional speckle-tracking echocardiography. Heart Vessels 38, 825–838 (2023). https://doi.org/10.1007/s00380-023-02232-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-023-02232-1