Abstract
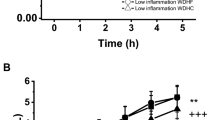
The aim was to study the effect of a standardized oral fat load (OFL) on different inflammatory parameters in a large sample of adult healthy subjects (n = 286) of both sexes. The fat load was given between 08:00 and 09:00 h after a 12-h fast. Blood samples were drawn before and 3, 6, 9, and 12 h after the OFL. All patients underwent a measurement of body mass index (BMI), blood glucose (BG), systolic blood pressure (SBP), diastolic blood pressure (DBP), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), triglycerides (Tg), soluble intercellular adhesion molecule-1 (sICAM-1), interleukin-6 (IL-6), high-sensitivity C-reactive protein (hsCRP), soluble vascular cell adhesion molecule-1 (sVCAM-1), soluble E-selectin (sE-selectin), and tumor necrosis factor-α (TNF-α). Fasting plasma glucose (FPG) increase was +3.26% at 3 h, +4.35% at 6 h, +1.09% at 9 h while FPG decrease was −1.09% at 12 h. High-density lipoprotein cholesterol increase was +2.08% at 3 h, and at 12 h during OFL study; a significant HDL-C decrease was present in subjects after 6 h (−4.17%; P < 0.05 vs 0). A significant Tg change was observed after 6 h (+70.37%; P < 0.01 vs 0) and 9 h (+58.33%; P < 0.05 vs 0) respectively, and the increase was +22.22% at 3 h and +18.52% at 12 h. Total cholesterol increase was +0.52% after 3 h, +1.04% after 6 h, while after 12 h the decrease was −0.52%. Low-density lipoprotein cholesterol increase was +1.64% after 6 h with a decrease of −0.82% at 9 and 12 h. A significant sICAM-1, hsCRP, and sE-selectin variation was observed after 6 and 9 h, while a significant sVCAM-1 change occurred after 3, 6, and 9 h. Soluble ICAM-1 increase was +20.58% at 3 h, +34.10% at 6 h (P < 0.05 vs 0) +25.94% at 9 h (P < 0.01 vs 0), and +19.14% at 12 h; sVCAM-1 increase was +13.97% (P < 0.05 vs 0) at 3 h, +18.55% at 6 h (P < 0.01 vs 0), +12.02% at 9 h (P < 0.05 vs 0), and +8.70% at 12 h. High-sensitivity CRP increase was +36.36% at 3 h, +90.91% at 6 h (P < 0.01 vs 0), +63.64% at 9 h (P < 0.05 vs 0), and +36.36% at 12 h. Soluble E-selectin increase was +27.11% at 3 h, +51.90% at 6 h (P < 0.05 vs 0), +45.19% at 9 h (P < 0.01 vs 0), and +20.12% at 12 h. Interleukin-6 increase was +61.11% at 3 h (P < 0.05 vs 0), +83.33% at 6 h (P < 0.001 vs 0), +55.56% at 9 h (P < 0.01 vs 0), and +22.22% at 12 h. Tumor necrosis factor-α increase was +42.86% at 3 h (P < 0.05 vs 0), +71.43% at 6 h (P < 0.01 vs 0), (+50.00% at 9 h (P < 0.05 vs 0), and +28.57% at 12 h. We observed that the OFL induces a complex and massive systemic inflammatory response that includes IL-6, TNF-α, hsCRP, and cell adhesion molecules, even before Tg significantly rises.
Similar content being viewed by others
References
McBride PE (2007) Triglycerides and risk for coronary heart disease. JAMA 298(3):336–338
Nordestgaard BG, Benn M, Schnohr P, Tybjaerg-Hansen A (2007) Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA 298(3): 299–308
Groot PH, van Stiphout WA, Krauss XH, Jansen H, van Tol A, van Ramshorst E, Chin-On S, Hofman A, Cresswell SR, Havekes L (1991) Postprandial lipoprotein metabolism in normolipidemic men with and without coronary artery disease. Arterioscler Thromb 11(3):653–662
Maggi FM, Raselli S, Grigore L, Redaelli L, Fantappiè S, Catapano AL (2004) Lipoprotein remnants and endothelial dysfunction in the postprandial phase. J Clin Endocrinol Metab 89(6):2946–2950
Karpe F, Steiner G, Uffelman K, Olivecrona T, Hamsten A (1994) Postprandial lipoproteins and progression of coronary atherosclerosis. Atherosclerosis 106(1):83–97
Karpe F, Olivecrona T, Walldius G, Hamsten A (1992) Lipoprotein lipase in plasma after an oral fat load: relation to free fatty acids. J Lipid Res 33(7):975–984
De Bruin TW, Brouwer CB, Gimpel JA, Erkelens DW (1991) Postprandial decrease in HDL cholesterol and HDL apo A-I in normal subjects in relation to triglyceride metabolism. Am J Physiol 260(3 Pt 1):E492–E498
Miller M, Kwiterovich PO Jr, Bachorik PS, Georgopoulos A (1993) Decreased postprandial response to a fat meal in normotriglyceridemic men with hypoalphalipoproteinemia. Arterioscler Thromb 13(3):385–392
Karpe F, Steiner G, Olivecrona T, Carlson LA, Hamsten A (1993) Metabolism of triglyceride-rich lipoproteins during alimentary lipemia. J Clin Invest 91(3):748–758
Cassader M, Gambino R, Ruiu G, Marena S, Bodoni P, Pagano G (1996) Postprandial triglyceride-rich lipoprotein changes in elderly and young subjects. Aging 8(6):421–428
Parks EJ (2001) Recent findings in the study of postprandial lipemia. Curr Atheroscler Rep 3(6):462–470
Van Oostrom AJ, Alipour A, Plokker TW, Sniderman AD, Cabezas MC (2007) The metabolic syndrome in relation to complement component 3 and postprandial lipemia in patients from an outpatient lipid clinic and healthy volunteers. Atherosclerosis 190(1):167–173
Peake PW, Kriketos AD, Campbell LV, Charlesworth JA (2005) Response of the alternative complement pathway to an oral fat load in first-degree relatives of subjects with type II diabetes. Int J Obes 29(4):429–435
European Diabetes Policy Group (1999) A desktop guide to type 2 diabetes mellitus. Diabet Med 16:716–730
Klose S, Borner K (1978) Enzymatische Bestimmung des Gesamtcholesterins mit dem Greiner Selective Analyzer (GSA II). J Clin Chem Clin Biochem 15:121–130
Wahlefeld AW (1974) Triglycerides determination after enzymatic hydrolysis. In: Bermeyer HU (ed) Methods of enzymatic analysis, 2nd English edn. Academic Press, New York, pp 18–31
Havel RJ, Edr HA, Bragdon JH (1955) The distribution and chemical composition of ultracentrifugally separated lipoproteins in human serum. J Clin Invest 34:1345–1353
Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low density lipoprotein in plasma, without use of the preparative ultracentrifuge. Clin Chem 18:499–502
Song M, Kellum JA (2005) Interleukin-6. Crit Care Med 33(12): S463–S465
Zhang M, Tracey K (1988) The cytokine handbook, 3rd edn. Academic Press, San Diego
Rifai N, Tracy RP, Ridker PM (1999) Clinical efficacy of an automated high-sensitivity C-reactive protein assay. Clin Chem 45(12): 2136–2141
Witkowska AM, Borawska MH (2004) Soluble intercellular adhesion molecule-1 (sICAM-1): an overview. Eur Cytokine Netw 15(2):91–98
Peter K, Weirich U, Nordt TK, Ruef J, Bode C (1999) Soluble vascular cell adhesion molecule-1 (VCAM-1) as potential marker of atherosclerosis. Thromb Haemost 82(1):38–43
Constans J, Conri C (2006) Circulating markers of endothelial function in cardiovascular disease. Clin Chim Acta 368(1–2): 33–47
Winer BJ (1971) Statistical principles in experimental design, 2nd edn. McGraw-Hill, New York
Matthews JNS, Altman DG, Campbell MJ, Royton P (1990) Analysis of serial measurements in medical research. BMJ 300: 230–235
Haffner SM, Foster DM, Kushwaha RS, Hazzard WR (1984) Retarded chylomicron apolipoprotein-B catabolism in type 2 (non-insulin-dependent) diabetic subjects with lipaemia. Diabetologia 26(5):349–354
Barbagallo CM, Averna MR, Amato S, Davì G, Pagano D, Noto D, Notarbartolo A (1991) Lipid and apoprotein behaviour after oral fat load in hypertriglyceridaemia. Diabetes Metab 17(6): 512–519
Genest J, Sniderman A, Cianflone K, Teng B, Wacholder S, Marcel Y, Kwiterovich P Jr (1986) Hyperapobetalipoproteinemia. Plasma lipoprotein responses to oral fat load. Arteriosclerosis 6(3): 297–304
Kay RM, Rao S, Arnott C, Miller NE, Lewis B (1980) Acute effects of the pattern of fat ingestion on plasma high density lipoprotein components in man. Atherosclerosis 36(4):567–573
Karpe F, Bard JM, Steiner G, Carlson LA, Fruchart JC, Hamsten A (1993) HDLs and alimentary lipemia. Studies in men with previous myocardial infarction at a young age. Arterioscler Thromb 13(1):11–22
Alipour A, Elte JW, van Zaanen HC, Rietveld AP, Cabezas MC (2007) Postprandial inflammation and endothelial dysfunction. Biochem Soc Trans 35(Pt 3):466–469
Constans J, Conri C (2006) Circulating markers of endothelial function in cardiovascular disease. Clin Chim Acta 368(1–2): 33–47
Twickler TB, Dallinga-Thie GM, Visseren FL, de Vries WR, Erkelens DW, Koppeschaar HP (2003) Induction of postprandial inflammatory response in adult onset growth hormone deficiency is related to plasma remnant-like particle-cholesterol concentration. J Clin Endocrinol Metab 88(3):1228–1233
Norata GD, Grigore L, Raselli S, Redaelli L, Hamsten A, Maggi F, Eriksson P, Catapano AL (2007) Postprandial endothelial dysfunction in hypertriglyceridemic subjects: molecular mechanisms and gene expression studies. Atherosclerosis 193(2):321–327
Lundman P, Boquist S, Samnegård A, Bennermo M, Held C, Ericsson CG, Silveira A, Hamsten A, Tornvall P (2007) A high-fat meal is accompanied by increased plasma interleukin-6 concentrations. Nutr Metab Cardiovasc Dis 17(3):195–202
Tsai WC, Li YH, Lin CC, Chao TH, Chen JH (2004) Effects of oxidative stress on endothelial function after a high-fat meal. Clin Sci 106(3):315–319
Mattes RD (1996) Oral fat exposure alters postprandial lipid metabolism in humans. Am J Clin Nutr 63(6):911–917
Derosa G, D’Angelo A, Scalise F, Avanzini MA, Tinelli C, Peros E, Fogari E, Cicero AF (2007) Comparison between metalloproteinases-2 and -9 in healthy subjects, diabetics, and subjects with acute coronary syndrome. Heart Vessels 22(6):361–370
Murasaki S, Murasaki K, Tanoue K, Kawana M, Hagiwara N, Kasanuki H (2007) Circulating platelet and neutrophil activation correlates with the clinical course of unstable angina. Heart Vessels 22(6):376–382
Shimabukuro M, Chinen I, Higa N, Takasu N, Yamakawa K, Ueda S (2007) Effects of dietary composition on postprandial endothelial function and adiponectin concentrations in healthy humans: a crossover controlled study. Am J Clin Nutr 86(4):923–928
Coutinho ER, Macedo GM, Campos FS, Bandeira FA (2008) Changes in HDL cholesterol and in the inflammatory markers of atherogenesis after an oral fat load in type-2 diabetic patients and normal individuals. Metab Syndr Relat Disord 6(2):153–157
Dickinson S, Hancock DP, Petocz P, Ceriello A, Brand-Miller J (2008) High-glycemic index carbohydrate increases nuclear factor-kappaB activation in mononuclear cells of young, lean healthy subjects. Am J Clin Nutr 87(5):1188–1193
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Derosa, G., Ferrari, I., D’Angelo, A. et al. Oral fat load effects on inflammation and endothelial stress markers in healthy subjects. Heart Vessels 24, 204–210 (2009). https://doi.org/10.1007/s00380-008-1109-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-008-1109-y