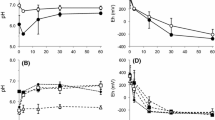
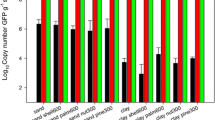
Abstract
Viruses are extremely abundant in soils and have various important ecological implications therein. The quantification and identification of soil viruses are critical for a comprehensive understanding of their abundance, diversity and ecological functions. Herein, we compared three physical dispersion methods (shaking, vortexing and sonicating) and 10 extraction buffers to determine the best protocol for virus extraction from a broad range of soils in the (sub)tropics. A 5-min sonication combined with potassium citrate (AKC) buffer (1% potassium citrate amended with 10% PBS, 5 mM EDTA and 150 mM MgSO4) treatment yielded virus-like particles (VLPs) at a yield one to two orders of magnitude greater than that achieved via other methods, as determined through epifluorescence microscopy (EFM). Using the most successful method, viral abundance in red soils ranged from 2.3 × 107 VLPs g−1 soil to 1.3 × 109 VLPs g−1 soil, with approximately two times more VLPs in paddy soils than in adjacent upland soils. VLP abundance increased with soil water content (R2 = 0.60), soil organic carbon (R2 = 0.70) and bacterial abundance (R2 = 0.69), suggesting that land use strongly affects viral abundance. This comparison clearly showed that extraction methods determine the measured viral abundance to a great extent. Based on our tests of a broad range of physical dispersion methods and chemical solutions, we concluded that a 5-min sonication combined with potassium citrate buffer treatment is the best approach for extracting VLPs from red subtropical soils.







Similar content being viewed by others
References
Baligar VC, Fageria NK, Eswaran H, Wilson MJ, He Z (2004). Nature and properties of red soils of the world. In: Wilson MJ, He Z, Yang X. (eds), The red soils of China. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-2138-1_2
Bi L, Yu DT, Du S, Zhang LM, Zhang LY, Wu CF, Xiong C, Han LL, He JZ (2021) Diversity and potential biogeochemical impacts of viruses in bulk and rhizosphere soils. Environ Microbiol 23:588–599. https://doi.org/10.1111/1462-2920.15010
Bi L, Yu DT, Han LL, Du S, Yuan CY, He JZ, Hu HW (2022) Unravelling the ecological complexity of soil viromes: challenges and opportunities. Sci Total Environ 812:152217. https://doi.org/10.1016/j.scitotenv.2021.152217
Brassard J, Gagné MJ (2018) An efficient recovery method for enteric viral particles from agricultural soils. J Virol Methods 261:1–5. https://doi.org/10.1016/j.jviromet.2018.06.014
Brussaard CPD (2004) Optimization of procedures for counting viruses by flow cytometry. Appl Environ Microbiol 70:1506–1513. https://doi.org/10.1128/AEM.70.3.1506-1513.2004
Chagnon PL, Bradley RL (2011) On the use of sodium hexametaphosphate to extract spores of arbuscular mycorrhizal fungi from soil. Soil Biol Biochem 43:2273–2274. https://doi.org/10.1016/j.soilbio.2011.07.021
Chen ML, An XL, Liao H, Yang K, Su JQ, Zhu YG (2021a) Viral community and virus-associated antibiotic resistance genes in soils amended with organic fertilizers. Environ Sci Technol 55:13881–13890. https://doi.org/10.1021/acs.est.1c03847
Chen X, Weinbauer MG, Jiao N, Zhang R (2021b) Revisiting marine lytic and lysogenic virus-host interactions: Kill-the-Winner and Piggyback-the-Winner. Sci Bull 66:871–874. https://doi.org/10.1016/j.scib.2020.12.014
Chevallereau A, Pons BJ, Van Houte S, Westra ER (2022) Interactions between bacterial and phage communities in natural environments. Nat Rev Microbiol 20:49–62. https://doi.org/10.1038/s41579-021-00602-y
CobiánGüemes AG, Youle M, Cantú VA, Felts B, Nulton J, Rohwer F (2016) Viruses as winners in the game of life. Annu Rev Virol 3:197–214. https://doi.org/10.1146/annurev-virology-100114-054952
Danovaro R, Dell’Anno A, Trucco A, Serresi M, Vanucci S (2001) Determination of virus abundance in marine sediments. Appl Environ Microbiol 67:1384–1387. https://doi.org/10.1128/aem.67.3.1384-1387.2001
Göller PC, Haro-Moreno JM, Rodriguez-Valera F, Loessner MJ, Gómez-Sanz E (2020) Uncovering a hidden diversity: optimised protocols for the extraction of dsDNA bacteriophages from soil. Microbiome 8:17. https://doi.org/10.1186/s40168-020-0795-2
Han LL, Yu DT, Zhang LM, Shen JP, He JZ (2017) Genetic and functional diversity of ubiquitous DNA viruses in selected Chinese agricultural soils. Sci Rep 7:45142. https://doi.org/10.1038/srep45142
Helsley KR, Brown TM, Furlong K, Williamson KE (2014) Applications and limitations of tea extract as a virucidal agent to assess the role of phage predation in soils. Biol Fert Soils 50:263–274. https://doi.org/10.1007/s00374-013-0855-x
Howard-Varona C, Hargreaves KR, Abedon ST, Sullivan MB (2017) Lysogeny in nature: mechanisms, impact and ecology of temperate phages. ISME J 11:1511–1520. https://doi.org/10.1038/ismej.2017.16
Huang D, Yu PF, Ye M, Schwarz C, Jiang X, Alvarez PJJ (2021) Enhanced mutualistic symbiosis between soil phages and bacteria with elevated chromium-induced environmental stress. Microbiome 9:150. https://doi.org/10.1186/s40168-021-01074-1
Jin M, Guo X, Zhang R, Qu W, Gao B, Zeng R (2019) Diversities and potential biogeochemical impacts of mangrove soil viruses. Microbiome 7:58. https://doi.org/10.1186/s40168-019-0675-9
Kimura M, Jia ZJ, Nakayama N, Asakawa S (2008) Ecology of viruses in soils: past, present and future perspectives. Soil Sci Plant Nutr 54:1–32. https://doi.org/10.1111/j.1747-0765.2007.00197.x
Kleber M, Eusterhues K, Keiluweit M, Mikutta C, Mikutta R, Nico PS (2015) Chapter One - Mineral–organic associations: formation, properties, and relevance in soil environments. In: Sparks DL (Ed). Adv Agron. 130:1–140. https://doi.org/10.1016/bs.agron.2014.10.005
Kuzyakov Y, Mason-Jones K (2018) Viruses in soil: nano-scale undead drivers of microbial life, biogeochemical turnover and ecosystem functions. Soil Biol Biochem 127:305–317. https://doi.org/10.1016/j.soilbio.2018.09.032
Lasobras J, Dellunde J, Jofre J, Lucena F (1999) Occurrence and levels of phages proposed as surrogate indicators of enteric viruses in different types of sludges. J Appl Microbiol 86:723–729. https://doi.org/10.1046/j.1365-2672.1999.00722.x
Li Y, Liu H, Pan H, Zhu X, Liu C, Zhang Q, Luo Y, Di H, Xu J (2019) T4-type viruses: Important impacts on shaping bacterial community along a chronosequence of 2000-year old paddy soils. Soil Biol Biochem 128:89–99. https://doi.org/10.1016/j.soilbio.2018.10.007
Liang X, Wagner RE, Zhuang J, DeBruyn JM, Wilhelm SW, Liu F, Yang L, Staton ME, Sherfy AC, Radosevich M (2019) Viral abundance and diversity vary with depth in a southeastern United States agricultural ultisol. Soil Biol Biochem 137:107546. https://doi.org/10.1016/j.soilbio.2019.107546
Liang X, Zhang Y, Wommack KE, Wilhelm SW, DeBruyn JM, Sherfy AC, Zhuang J, Radosevich M (2020) Lysogenic reproductive strategies of viral communities vary with soil depth and are correlated with bacterial diversity. Soil Biol Biochem 144:107767. https://doi.org/10.1016/j.soilbio.2020.107767
Lin D, Cai P, Peacock CL, Wu YC, Gao CH, Peng WX, Huang QY, Liang W (2018) Towards a better understanding of the aggregation mechanisms of iron(hydr)oxide nanoparticles interacting with extracellular polymeric substances: role of pH and electrolyte solution. Sci Total Environ 645:372–379. https://doi.org/10.1016/j.scitotenv.2018.07.136
Lipson SM, Stotzky G (1983) Adsorption of reovirus to clay minerals: effects of cation-exchange capacity, cation saturation, and surface area. Appl Environ Microbiol 46:673–682. https://doi.org/10.1128/aem.46.3.673-682.1983
McKeague JA, Day JH (1966) Dithionite and oxalate extractable Fe and Al as aids in differentiating various classes of soils. Can J Soil Sci 46:13–22. https://doi.org/10.4141/cjss66-003
Middelboe M, Brussaard CPD (2017) Marine viruses: key players in marine ecosystems. Viruses 9:302. https://doi.org/10.3390/v9100302
Middelboe M, Glud RN (2003) Distribution of viruses and bacteria in relation to diagenetic activity in an estuarine sediment. Limnol Oceanogr 48:1447–1456. https://doi.org/10.4319/lo.2003.48.4.1447
Narr A, Nawaz A, Wick LY, Harms H, Chatzinotas A (2017) Soil viral communities vary temporally and along a land use transect as revealed by virus-like particle counting and a modified community fingerprinting approach (fRAPD). Front Microbiol 8:1975. https://doi.org/10.3389/fmicb.2017.01975
Pan D, Watson R, Wang D, Tan ZH, Snow DD, Weber KA (2014) Correlation between viral production and carbon mineralization under nitrate-reducing conditions in aquifer sediment. ISME J 8:1691–1703. https://doi.org/10.1038/ismej.2014.38
Pratama AA, van Elsas JD (2018) The ‘neglected’ soil virome—potential role and impact. Trends Microbiol 26:649–662. https://doi.org/10.1016/j.tim.2017.12.004
Silveira CB, Luque A, Rohwer F (2021) The landscape of lysogeny across microbial community density, diversity and energetics. Environ Microbiol 23:4098–4111. https://doi.org/10.1111/1462-2920.15640
Speir JA, Bothner B, Qu C, Willits DA, Young MJ, Johnson JE (2006) Enhanced local symmetry interactions globally stabilize a mutant virus capsid that maintains infectivity and capsid dynamics. J Virol 80:3582–3591. https://doi.org/10.1128/jvi.80.7.3582-3591.2006
Suttle CA (2005) Viruses in the sea. Nature 437:356–361. https://doi.org/10.1038/nature04160
Srinivasiah S, Bhavsar J, Thapar K, Liles M, Schoenfeld T, Wommack KE (2008). Phages across the biosphere: contrasts of viruses in soil and aquatic environments. Res Microbiol 159:349-357. https://doi.org/10.1016/j.resmic.2008.04.010
Suttle CA (2007) Marine viruses—major players in the global ecosystem. Nat Rev Microbiol 5:801–812. https://doi.org/10.1038/nrmicro1750
Trubl G, Solonenko N, Chittick L, Solonenko SA, Rich VI, Sullivan MB (2016) Optimization of viral resuspension methods for carbon-rich soils along a permafrost thaw gradient. Peer J 4:e1999. https://doi.org/10.7717/peerj.1999
Trubl G, Hyman P, Roux S, Abedon ST (2020) Coming-of-age characterization of soil viruses: a user’s guide to virus isolation, detection within metagenomes, and viromics. Soil Systems 4:23. https://doi.org/10.3390/soilsystems4020023
Wang S, Redmile-Gordon M, Mortimer M, Cai P, Wu Y, Peacock CL, Gao C, Huang Q (2019) Extraction of extracellular polymeric substances (EPS) from red soils (ultisols). Soil Biol Biochem 135:283–285. https://doi.org/10.1016/j.soilbio.2019.05.014
Wang S, Yu S, Zhao X, Zhao X, Mason-Jones K, Zhu Z, Redmile-Gordon M, Li Y, Chen J, Kuzyakov Y, Ge T (2022) Experimental evidence for the impact of phages on mineralization of soil-derived dissolved organic matter under different temperature regimes. Sci Total Environ 846:157517. https://doi.org/10.1016/j.scitotenv.2022.157517
Williamson KE, Wommack KE, Radosevich M (2003) Sampling natural viral communities from soil for culture-independent analyses. Appl Environ Microbiol 69:6628–6633. https://doi.org/10.1128/aem.69.11.6628-6633.2003
Williamson KE, Radosevich M, Wommack KE (2005) Abundance and diversity of viruses in six Delaware soils. Appl Environ Microbiol 71:3119–3125. https://doi.org/10.1128/AEM.71.6.3119-3125.2005
Williamson KE, Corzo KA, Drissi CL, Buckingham JM, Thompson CP, Helton RR (2013) Estimates of viral abundance in soils are strongly influenced by extraction and enumeration methods. Biol Fert Soils 49:857–869. https://doi.org/10.1007/s00374-013-0780-z
Williamson KE, Fuhrmann JJ, Wommack KE, Radosevich M (2017) Viruses in soil ecosystems: an unknown quantity within an unexplored territory. Annu Rev Virol 4:201–219. https://doi.org/10.1146/annurev-virology-101416-041639
Zimmerman AE, Howard-Varona C, Needham DM, John SG, Worden AZ, Sullivan MB, Waldbauer JR, Coleman ML (2020) Metabolic and biogeochemical consequences of viral infection in aquatic ecosystems. Nat Rev Microbiol 18:21–34. https://doi.org/10.1038/s4157
Funding
This study was supported by the National Natural Science Foundation of China (42107312; 42141006), Key Project of Science and Technology Innovation in Ningbo City (2022Z169), “Pioneer” and “Leading Goose” R&D Program of Zhejiang (2023C02016; 2022C02008), Seagull Talent of Yongjiang Talent for Yakov Kuzyakov Fund of Ningbo University and the RUDN University Strategic Academic Leadership Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, S., Wang, S., Zhao, X. et al. Extraction optimisation to measure viral abundance in red soils. Biol Fertil Soils 59, 927–937 (2023). https://doi.org/10.1007/s00374-023-01765-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-023-01765-x