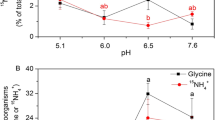
Abstract
Root mucilage modulates soil-plant-water dynamics, but its interactions with microbial community functioning remain poorly understood. The aims of this study were to estimate (I) the impacts of mucilage and soil water content on the microbial community composition and (II) the mucilage consumption by individual microbial groups. C4 root mucilage from maize (at 40 and 200 μg C per gram dry soil, corresponding to 10 and 50% of soil microbial biomass, respectively) was added in single pulses to a C3 soil at two moisture levels: optimum (80% of water-holding capacity (WHC)) and drought (30% of WHC). After 15 days of incubation, the microbial community composition was studied by phospholipid fatty acids (PLFA) analysis and incorporation of mucilage-derived 13C into individual microbial groups was determined by compound-specific isotope analysis. Microbial community composition remained largely unaffected by mucilage addition but was affected by moisture. Whereas an increase in water content reduced mucilage 13C recovery in PLFA for the low-dose mucilage amendment from 19 to 9%, it had no effect under the high-dose amendment (11–12%). This suggests that the role of mucilage for microbial functioning is especially pronounced under drought conditions. The fungal PLFA 18:2ω6,9 was present only under drought conditions, and fungi profited in their mucilage C utilisation from the lower competitiveness of many bacterial groups under drought. In this study, Gram-negatives (G−, characterised by PLFA 18:1ω9c, 18:1ω7c, 16:1ω7c and cy17:0) showed the highest mucilage-derived 13C in PLFA, especially at the high-dose amendment, suggesting them to be the major decomposers of mucilage, especially when the availability of this C source is high. Gram-positives (G+) included different sub-groups with distinct responses to moisture: G+ 1 (a15:0) were only competitive for mucilage C under drought, whereas G+ 3 (i17:0) were only able to utilise mucilage-derived C under optimal moisture conditions. During the 15-day incubation, they built up more than 40% of their membranes from mucilage-derived C, suggesting that in the case of high availability, mucilage can act as an important C source for this microbial group. However, under drought, G− 1 and fungi were incorporating the most mucilage C into their membranes (approx. 20% of PLFA-C). The observation that, for some groups, the high-dose mucilage amendments under drought led to higher 13C incorporation into PLFA than under optimum moisture suggests that mucilage can compensate drought effects for particular microbial groups. Thus, mucilage may not only act as a C source for microorganisms but may also mitigate drought effects for specific rhizosphere microbial groups.





Similar content being viewed by others
References
Ahmed MA, Kroener E, Holz M, Zarebanadkouki M, Carminati A (2014) Mucilage exudation facilitates root water uptake in dry soils. Funct Plant Biol 41:1129–1137. https//doi:10.1071/FP13330
Ahmed MA, Holz M, Woche SK, Bachmann J and Carminati A (2015) Effect of soil drying on mucilage exudation and its water repellency: a new method to collect mucilage. J Plant Nutr Soil Sci 178: 821–824. https//doi:10.1002/jpln.201500177
Ahmed MA, Zarebanadkouki M, Ahmadi K, Kroener E, Kostka S, Kaestner A, Carminati A (2017) Engineering rhizosphere hydraulics: pathways to improve plant adaptation to drought. Vadose Zone J. https://doi.org/10.2136/vzj2016.09.0090
Apostel C, Dippold M, Glaser B, Kuzyakov Y (2013) Biochemical pathways of amino acids in soil: assessment by position-specific labeling and 13C-PLFA analysis. Soil Biol Biochem 67:31–40. https://doi.org/10.1016/j.soilbio.2013.08.005
Bacic A, Moody SF, Clarke AE (1986) Structural analysis of secreted root slime from maize (Zea mays L.) Plant Physiol 80:771–777. https://doi.org/10.1104/pp.80.3.771
Basler A, Dippold M, Helfrich M, Dyckmans J (2015) Microbial carbon recycling—an underestimated process controlling soil carbon dynamics—part 1: a long-term laboratory incubation experiment. Biogeosciences 12:5929–5940. https://doi.org/10.5194/bg-12-5929-2015
Benizri E, Nguyen C, Piutti S, Slezack-Deschaumes S, Philippot L (2007) Additions of maize root mucilage to soil changed the structure of the bacterial community. Soil Biol Biochem 39:1230–1233. https://doi.org/10.1016/j.soilbio.2006.12.026
Berg G, Smalla K (2009) Plant species and soil type cooperatively shape the structure and function of microbial communities in the rhizosphere. FEMS Microbiol Ecol 68:1–13. https://doi.org/10.1111/j.1574-6941.2009.00654.x
Bird JA, Herman DJ, Firestone MK (2011) Rhizosphere priming of soil organic matter by bacterial groups in a grassland soil. Soil Biol Biochem 43:718–725. https://doi.org/10.1016/j.soilbio.2010.08.010
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917. https://doi.org/10.1139/o59-099
Brax M, Buchmann C, Schaumann GE (2017) Biohydrogel induced soil–water interactions: how to untangle the gel effect? A review. J Plant Nutr Soil Scin 180:121–141. https://doi.org/10.1002/jpln.201600453
Carminati A, Benard P, Ahmed MA, Zarebanadkouki M (2017) Liquid bridges at the root-soil interface. Plant Soil 417:1–15. https://doi.org/10.1007/s11104-017-3227-8
Chaboud A (1983) Isolation, purification and chemical composition of maize root cap slime. Plant Soil 73:395–402. https://doi.org/10.1007/BF02184316
Chenu C, Roberson EB (1996) Diffusion of glucose in microbial extracellular polysaccharide as affected by water potential. Soil Biol Biochem 28:877–884
Fierer N, Schimel JP, Holden PA (2003) Influence of drying–rewetting frequency on soil bacterial community structure. Microb Ecol 45:63–71. https://doi.org/10.1007/s00248-002-1007-2
Flemming H-C, Wingender J (2010) The biofilm matrix. Nat Rev Microbiol 8:623–633. https://doi.org/10.1038/nrmicro2415
Frostegård Å, Tunlid A, Bååth E (1991) Microbial biomass measured as total lipid phosphate in soils of different organic content. J Microbiol Methods 14:151–163. https://doi.org/10.1016/0167-7012(91)90018-L
Frostegård Å, Tunlid A, Bååth E (1993) Phospholipid fatty acid composition, biomass, and activity of microbial communities from two soil types experimentally exposed to different heavy metals. Appl Environ Microbiol 59:3605–3617
Ge T, Li B, Zhu Z, Hu Y, Yuan H, Dorodnikov M, Jones DL, Wu J, Kuzyakov Y (2017) Rice rhizodeposition and its utilization by microbial groups depends on N fertilization. Biol Fertil Soils 53:37–48. https://doi.org/10.1007/s00374-016-1155-z
Glaser B, Amelung W (2002) Determination of 13C natural abundance of amino acid enantiomers in soil: methodological considerations and first results. Rapid Commun Mass Spectrom 16:891–898. https://doi.org/10.1002/rcm.650
Griffiths RI, Whiteley AS, O’Donnell AG, Bailey MJ (2003) Physiological and community responses of established grassland bacterial populations to water stress. Appl Environ Microbiol 69:6961–6968. https://doi.org/10.1128/AEM.69.12.6961-6968.2003
Guhr A, Borken W, Spohn M, Matzner E (2015) Redistribution of soil water by a saprotrophic fungus enhances carbon mineralization. Proc Natl Acad Sci 112:14647–14651. https://doi.org/10.1073/pnas.1514435112
Gunina A, Dippold MA, Glaser B, Kuzyakov Y (2014) Fate of low molecular weight organic substances in an arable soil: from microbial uptake to utilisation and stabilisation. Soil Biol Biochem 77:304–313. https://doi.org/10.1016/j.soilbio.2014.06.029
Harris RF (1981) Effect of water potential on microbial growth and activity. SSSA Special Publication, Water Potential Relations in Soil Microbiology 9:23–95. https://doi.org/10.2136/sssaspecpub9.c2
Heuer H, Krsek M, Baker P, Smalla K, Wellington EM (1997) Analysis of actinomycete communities by specific amplification of genes encoding 16S rRNA and gel-electrophoretic separation in denaturing gradients. Appl Environ Microbiol 63:3233–3241
Hueso S, García C, Hernández T (2012) Severe drought conditions modify the microbial community structure, size and activity in amended and unamended soils. Soil Biol Biochem 50:167–173. https://doi.org/10.1016/j.soilbio.2012.03.026
Iijima M, Higuchi T, Barlow PW, Bengough AG (2003) Root cap removal increases root penetration resistance in maize (Zea mays L.) J Exp Bot 54:2105–2109. https://doi.org/10.1093/jxb/erg226
Jones DL, Hodge A, Kuzyakov Y (2004) Plant and mycorrhizal regulation of rhizodeposition. New Phytol 163:459–480. https://doi.org/10.1111/j.1469-8137.2004.01130.x
Jones DL, Nguyen C, Finlay RD (2009) Carbon flow in the rhizosphere: carbon trading at the soil–root interface. Plant Soil 321:5–33. https://doi.org/10.1007/s11104-009-9925-0
Kaur A, Chaudhary A, Kaur A, Choudhary R, Kaushik R (2005) Phospholipid fatty acid: a bioindicator of environment monitoring and assessment in soil ecosystem. Curr Sci 89:1103–1112
Kramer C, Gleixner G (2008) Soil organic matter in soil depth profiles: distinct carbon preferences of microbial groups during carbon transformation. Soil Biol Biochem 40:425–433. https://doi.org/10.1016/j.soilbio.2007.09.016
Kuzyakov Y, Blagodatskaya E (2015) Microbial hotspots and hot moments in soil: concept & review. Soil Biol Biochem 83:184–199. https://doi.org/10.1016/j.soilbio.2015.01.025
López-Gutiérrez JC, Philippot L, Martin-Laurent F (2005) Impact of maize mucilage on atrazine mineralization and atzC abundance. Pest Manag Sci 61:838–844. https://doi.org/10.1002/ps.1078
Lozán JL, Kausch H (1998) Angewandte Statistik für Naturwissenschaftler. Parey Buchverlag, Singhofen
Lynch JP (2007) Turner review no. 14. Roots of the second green revolution. Aust J Bot 55:493–512. https://doi.org/10.1071/BT06118
Marschner P, Marhan S, Kandeler E (2012) Microscale distribution and function of soil microorganisms in the interface between rhizosphere and detritusphere. Soil Biol Biochem 49:174–183. https://doi.org/10.1016/j.soilbio.2012.01.033
McCarthy AJ, Williams ST (1992) Actinomycetes as agents of biodegradation in the environment—a review. Gene 115:189–192. https://doi.org/10.1016/0378-1119(92)90558-7
McCully ME, Boyer JS (1997) The expansion of maize root-cap mucilage during hydration. 3. Changes in water potential and water content. Physiol Plant 99:169–177
Mommer L, Hinsinger P, Prigent-Combaret C, Visser EJW (2016) Advances in the rhizosphere: stretching the interface of life. Plant Soil 407:1–8. https://doi.org/10.1007/s11104-016-3040-9
Mounier E, Hallet S, Chèneby D, Benizri E, Gruet Y, Nguyen C, Piutti S, Robin C, Slezack-Deschaumes S, Martin-Laurent F, Germon JC, Philippot L (2004) Influence of maize mucilage on the diversity and activity of the denitrifying community. Environ Microbiol 6:301–312. https://doi.org/10.1111/j.1462-2920.2004.00571.x
Or D, Phutane S, Dechesne A (2007) Extracellular polymeric substances affecting pore-scale hydrologic conditions for bacterial activity in unsaturated soils. Vadose Zone J 6:298. https://doi.org/10.2136/vzj2006.0080
Paterson E (2003) Importance of rhizodeposition in the coupling of plant and microbial productivity. Eur J Soil Sci 54:741–750. https://doi.org/10.1046/j.1351-0754.2003.0557.x
Paterson E, Gebbing T, Abel C, Sim A, Telfer G (2007) Rhizodeposition shapes rhizosphere microbial community structure in organic soil. New Phytol 173:600–610. https://doi.org/10.1111/j.1469-8137.2006.01931.x
Pii Y, Mimmo T, Tomasi N, Terzano R, Cesco S, Crecchio C (2015) Microbial interactions in the rhizosphere: beneficial influences of plant growth-promoting rhizobacteria on nutrient acquisition process. A review. Biol Fertil Soils 51:403–415. https://doi.org/10.1007/s00374-015-0996-1
Placella SA, Brodie EL, Firestone MK (2012) Rainfall-induced carbon dioxide pulses result from sequential resuscitation of phylogenetically clustered microbial groups. Proc Natl Acad Sci 109:10931–10936. https://doi.org/10.1073/pnas.1204306109
Read DB, Bengough AG, Gregory PJ, Crawford JW, Robinson D, Scrimgeour CM, Young IM, Zhang K, Zhang X (2003) Plant roots release phospholipid surfactants that modify the physical and chemical properties of soil. New Phytol 157:315–326. https://doi.org/10.1046/j.1469-8137.2003.00665.x
Roberson EB, Firestone MK (1992) Relationship between desiccation and exopolysaccharide production in a soil pseudomonas sp. Appl Environ Microbiol 58:1284–1291
Sanaullah M, Razavi BS, Blagodatskaya E, Kuzyakov Y (2016) Spatial distribution and catalytic mechanisms of β-glucosidase activity at the root-soil interface. Biol Fertil Soils 52:505–514. https://doi.org/10.1007/s00374-016-1094-8
Schimel J, Balser TC, Wallenstein M (2007) Microbial stress-response physiology and its implications for ecosystem function. Ecology 88:1386–1394. https://doi.org/10.1890/06-0219
Schimel JP, Scott WJ, Killham K (1989) Changes in cytoplasmic carbon and nitrogen pools in a soil bacterium and a fungus in response to salt stress. Appl Environ Microbiol 55:1635–1637
Schmitt J, Glaser B, Zech W (2003) Amount-dependent isotopic fractionation during compound-specific isotope analysis. Rapid Commun Mass Spectrom 17:970–977. https://doi.org/10.1002/rcm.1009
Tian J, Dippold M, Pausch J, Blagodatskaya E, Fan M, Li X, Kuzyakov Y (2013) Microbial response to rhizodeposition depending on water regimes in paddy soils. Soil Biol Biochem 65:195–203. https://doi.org/10.1016/j.soilbio.2013.05.021
Treonis AM, Ostle NJ, Stott AW, Primrose R, Grayston SJ, Ineson P (2004) Identification of groups of metabolically-active rhizosphere microorganisms by stable isotope probing of PLFAs. Soil Biol Biochem 36:533–537. https://doi.org/10.1016/j.soilbio.2003.10.015
Velasco AG-V, Probanza A, Mañero FJG, Solano BR, Lucas JA (2010) Characterization of the rhizosphere microbial community from different Arabidopsis thaliana genotypes using phospholipid fatty acids (PLFA) analysis. Plant Soil 329:315–325. https://doi.org/10.1007/s11104-009-0160-5
Watt M, McCully ME, Canny MJ (1994) Formation and stabilization of Rhizosheaths of Zea mays L. (effect of soil water content). Plant Physiol 106:179–186. https://doi.org/10.1104/pp.106.1.179
Williams MA (2007) Response of microbial communities to water stress in irrigated and drought-prone tallgrass prairie soils. Soil Biol Biochem 39:2750–2757. https://doi.org/10.1016/j.soilbio.2007.05.025
York LM, Carminati A, Mooney SJ, Ritz K, Bennett MJ (2016) The holistic rhizosphere: integrating zones, processes, and semantics in the soil influenced by roots. J Exp bot 67:3629–3643. https://doi.org/10.1093/jxb/erw108
Yuan H, Zhu Z, Liu S, Ge T, Jing H, Li B, Liu Q, Lynn TM, Wu J, Kuzyakov Y (2016) Microbial utilization of rice root exudates: 13C labeling and PLFA composition. Biol Fertil Soils 52:615–627. https://doi.org/10.1007/s00374-016-1101-0
Zelles L (1999) Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: a review. Biol Fertil Soils 29:111–129. https://doi.org/10.1007/s003740050533
Zelles L (1997) Experimental and theoretical approaches in environmental chemistry phospholipid fatty acid profiles in selected members of soil microbial communities. Chemosphere 35:275–294. https://doi.org/10.1016/S0045-6535(97)00155-0
Zickenrott I-M, Woche SK, Bachmann J, Ahmed MA, Vetterlein D (2016) An efficient method for the collection of root mucilage from different plant species—a case study on the effect of mucilage on soil water repellency. J Plant Nutr Soil Sci 179:294–302. https://doi.org/10.1002/jpln.201500511
Acknowledgments
We highly acknowledge DAAD and Alexander von Humboldt Foundation (AvH) for funding MAA and MS. Research was funded by DFG project “Mucilage: the hydraulic bridge between roots and soil” (CA 921/2-1), DFG KU 1184/29-1, INST 186/1006-1 /P and the Robert-Bosch Foundation in the framework of the Robert Bosch Junior Professorship to MD. We would like to thank the Centre for Stable Isotope Research and Analysis, Goettingen, for IRMS measurements and δ13C determinations. We would also like to thank the editor and two anonymous reviewers for their critical comments which improved the quality of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Ahmed, M.A., Banfield, C.C., Sanaullah, M. et al. Utilisation of mucilage C by microbial communities under drought. Biol Fertil Soils 54, 83–94 (2018). https://doi.org/10.1007/s00374-017-1237-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-017-1237-6