Abstract
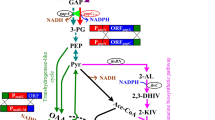
To investigate whether leucine biosynthesis plays a role in survival and growth under acidic conditions, leucine auxotrophs were produced from acid-tolerant Rhizobium tropici CIAT899. An internal fragment of the R. tropici leuA gene was PCR amplified using degenerate primers and cloned into an integrative plasmid for mutagenesis of the locus. Sequence and homology analysis confirmed the identity of the 550-bp gene fragment, with highest similarity (86% identity) to leuA from Sinorhizobium meliloti. The resultant mutants were strict leucine auxotrophs when tested on solid and liquid media. The mutants could be divided into two groups dependent on their ability to grow in the presence of leucine at both pH 6.8 and pH 5.5, or only at pH 5.5. One mutant, HS20, which could only grow properly in the presence of leucine at pH 5.5, was tested for its acid tolerance. When inoculated into medium at pH 3.5, the mutant was able to survive and to alter the pH from 3.5 to 3.8 whereas the wildtype could not. The wildtype was able to grow at pH 4.0, 4.5 and 5.0 without raising the extracellular pH, indicating that alteration of extracellular pH is not one of the strategies used by the wildtype for growth under acidic conditions. The ability of the mutant to raise the extracellular pH from 3.5 to 3.8 may enable it to survive at the extreme of its pH range of growth.


Similar content being viewed by others
References
Bearson S, Bearson B, Foster JW (1997) Acid stress responses in enterobacteria. FEMS Microbiol Lett 147:173–180
Booth IR (1999) The regulation of intracellular pH in bacteria. In: Chadwick DJ, Cardew G (eds) Bacterial responses to pH. Wiley, Chichester, pp 19–37
Castro SM, Glenn AR, Dilworth MJ (2000) Substrate selection as a possible strategy for amelioration of acid pH by Rhizobium leguminosarum biovar viciae. Soil Biol Biochem 32:1039–1041
Clarke LM, Dilworth MJ, Glenn AR (1993) Survival of Rhizobium meliloti WSM419 in laboratory culture: effect of combined pH shock and carbon substrate stress. Soil Biol Biochem 25:1289–1291
Dilworth MJ, Glenn AR (1999) Problems of adverse pH and bacterial strategies to combat it. In: Chadwick DJ, Cardew G (eds) Bacterial responses to pH. Wiley, Chichester, pp 4--14
Glenn AR, Dilworth MJ (1994) The life of root nodule bacteria in the acidic underground. FEMS Microbiol Lett 123:1--10
Glenn AR, Reeve WG, Tiwari RP, Dilworth MJ (1999) Acid tolerance in root nodule bacteria. In: Chadwick DJ, Cardew G (eds) Bacterial responses to pH. Wiley, Chichester, pp 112–130
Graham PH, Draeger KJ, Ferrey ML, Conroy MJ, Hammer BE, Martinez E, Aarons SR, Quinto C (1994) Acid pH tolerance in strains of Rhizobium and Bradyrhizobium, and initial studies on the basis for acid tolerance of Rhizobium tropici UMR1899. Can J Microbiol 40:198–207
Kuykendall LD (1981) Mutants of Rhizobium that are altered in legume interaction and nitrogen fixation. In: Giles KL, Atherly AG (eds) Biology of the Rhizobiaceae. Academic Press, London, pp 299–309
O'Hara GW, Glenn AR (1994) The adaptive acid tolerance response in root nodule bacteria and Escherichia coli. Arch Microbiol 161:286--292
Poole PS, Franklin M, Glenn AR, Dilworth MJ (1985) The transport of L-glutamate by Rhizobium leguminosarum involves a common amino acid carrier. J Gen Microbiol 131:1441–1448
Schäfer A, Tauch A, Jäger W, Kalinowski G, Thierbach G, Pühler A (1994) Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmids pK18 and pK19: selection of defined deletions in the chromosome of Corynebacterium glutamicum. Gene 145:69–73
Steele HL, Völker B, Fuhrmann GF, Werner D (1999) Strain specificities in Rhizobium tropici and R. etli using glucose transport inhibition by acidity and daidzein. Soil Biol Biochem 31:1059–1061
Tiwari RP, Reeve WG, Dilworth MJ, Glenn AR (1999) Genetic circuits involved in the response of root nodule bacteria to low pH. In: Pedrosa FO, Hungria M, Yates MG, Newton WE (eds) Nitrogen fixation: from molecules to crop productivity. Kluwer, Dortrecht, pp 475--476
Vinuesa P, Rademaker JLW, de Bruijn FJ, Werner D (1998) Genotypic characterization of Bradyrhizobium strains nodulating endemic woody legumes of the Canary Islands by PCR-restriction fragment length polymorphism analysis of genes encoding 16S rRNA (16S rDNA) and 16S-23S rDNA intergenic spacers, repetitive extragenic palindromic PCR genomic fingerprinting, and partial 16S rDNA sequencing. Appl Environ Microbiol 64:2096–2104
Walshaw DL, Poole PS (1996) The general L-amino acid permease of Rhizobium leguminosarum is an ABC uptake system that also influences efflux of solutes. Mol Microbiol 21:1239--1252
Werner D, Stripf R (1978) Differentiation of Rhizobium japonicum. I. Enzymatic comparison of nitrogenase repressed and derepressed free living cells and of bacteroids. Z Naturforsch 33:245—252
Acknowledgements
We thank the European Union for the "Training and Mobility of Researchers" network "Symbiosis and Defence of Plants and Microorganisms", and the SFB395 for funding this research. We also thank H. Thierfelder and M. Lange for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steele, H.L., Vinuesa, P. & Werner, D. A leucine biosynthesis mutant of Rhizobium tropici CIAT899 which survives at pH 3.5.. Biol Fertil Soils 38, 84–88 (2003). https://doi.org/10.1007/s00374-003-0616-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-003-0616-3