Abstract
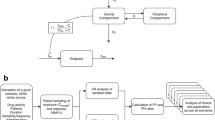
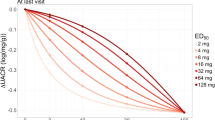
Dose-finding studies in rare diseases are faced with unique challenges including low patient numbers, limited understanding of the dose-exposure-response relationship, variability around the endpoints. In addition, patient exposure to placebo is often not feasible. To describe the disease progression for different dose groups, we introduce a longitudinal model for the change from baseline for a clinical endpoint. We build a nonlinear mixed effects model using the techniques which have become popular over the past two decades in the design and analysis of population pharmacokinetic/pharmacodynamics studies. To evaluate operating characteristics of the proposed design, we derive the Fisher information matrix and validate analytical results via simulations. Alternative considerations, such as trend analysis, are discussed as well.





Similar content being viewed by others
References
Atkinson AC, Donev A (1992) Optimum experimental design. Clarendon Press, Oxford
Bates D, Machler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Soft 67(1):1–48
Bretz F, Dette H, Pinheiro J (2010) Practical considerations for optimal designs in clinical dose finding studies. Stat Med 29(7–8):731–742
Dette H, Kiss M, Bevanda M, Bretz F (2010) Optimal designs for the Emax, log-linear and exponential models. Biometrika 97(2):513–518
Fedorov VV, Leonov SL (2013) Optimal design for nonlinear response models. Chapman & Hall, Boca Raton
Hedayat AS, Yan B, Pezutto JM (1997) Modeling and identifying optimum designs for fitting dose response curves based on raw optical data. J Am Stat Assoc 92:1132–1140
International Conference on Harmonization Working Group (1994) ICH Harmonized Tripartite Guideline E4: Dose-Response Information to Support Drug Registration. https://database.ich.org/sites/default/files/E4_Guideline.pdf
Jonckheere AR (1954) A distribution-free k-sample test against ordered alternatives. Biometrika 41(1/2):133–145
Karpinski KF (1990) Optimality assessment in the enzyme-linked immunosorbent assay (ELISA). Biometrics 46:381–390
Leonov SL, Aliev A (2013) Approximation of the Fisher information matrix for nonlinear mixed effects models in population PK/PD studies. In: Ucinski D, Atkinson AC, Patan M (eds) mODa 10—advances in model-oriented design and analysis. Springer, Heidelberg, pp 145–152
Nyberg J, Bazzoli C, Ogungbenro K, Aliev A, Leonov S, Duffull S, Hooker A, Mentré F (2015) Methods and software tools for design evaluation in population pharmacokinetics-pharmacodynamics. Br J Clin Pharm 79(1):6–17
Pinheiro JC, Bates DM (2000) Mixed-effects models in S and S-PLUS. Springer, New York
Pinheiro J, Bates D, DebRoy S, Sarkar D (2020) nlme: linear and nonlinear mixed effects models. R Core Team. R package version 3.1-148
Pozuelo-Campos S, Casero-Alonso V, Amo-Salas V (2021) Effect of probability distribution of the response variable in optimal experimental design with applications in medicine. Mathematics 9(9):1010
Pukelsheim F (1993) Optimal design of experiments. Wiley, New York
Pukelsheim F, Rieder S (1992) Efficient rounding of approximate designs. Biometrika 79(4):763–770
Ratkowsky DA, Reedy TJ (1986) Choosing non-linear parameters in the four-parameter logistic model for radioligand and related assays. Biometrics 42(1):575–582
Acknowledgements
The authors are grateful to two anonymous referees for their constructive comments on an earlier version of the paper which helped to improve the presentation of the results.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Y., Fries, M. & Leonov, S. Longitudinal model for a dose-finding study for a rare disease treatment. Stat Papers 64, 1343–1360 (2023). https://doi.org/10.1007/s00362-023-01424-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00362-023-01424-1
Keywords
- Dose-finding
- Nonlinear mixed effects model
- Longitudinal model
- Optimal design
- D-optimality
- \(ED_p\)-optimality