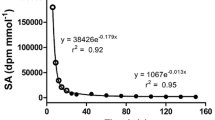
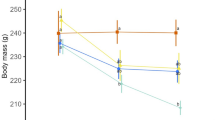
Abstract
The African clawed frog, Xenopus laevis, has been reported to tolerate long-term fasting without dormancy. However, the strategies for energy acquisition during fasting are unclear in this species. We performed 3- and 7-month fasting experiments to investigate how the metabolism of male X. laevis changes during long-term fasting. We found that the levels of several serum biochemical parameters, such as glucose, triglycerides, and free fatty acids, as well as liver glycogen were reduced after 3 months of fasting, whereas after 7 months of fasting, triglyceride levels were reduced, and fat body wet weight was lower than that of fed group indicating the onset of lipid catabolism. In addition, transcript levels of gluconeogenic genes, such as pck1, pck2, g6pc1.1, and g6pc1.2, were increased in the livers of animals fasted for 3 months, suggesting upregulation of gluconeogenesis. Our results raise the possibility that male X. laevis can tolerate much longer fasting than previously reported by utilizing several energy storage molecules. Further investigation of the effects of prolonged fasting on the metabolic switches from carbohydrates to lipids or amino acids in X. laevis is required.




Similar content being viewed by others
Data availability
The data is available from AI on request.
Abbreviations
- BUN:
-
Blood urea nitrogen
- CoA:
-
Coenzyme A
- β-HB:
-
β-Hydroxybutyrate
- HSI:
-
Hepatosomatic index
- NEFA:
-
Non-esterified fatty acid
- RT-qPCR:
-
Reverse transcription-quantitative polymerase chain reaction
- TG:
-
Triglyceride
References
Alberti KG, Hockaday TD (1972) Rapid blood ketone body estimation in the diagnosis of diabetic ketoacidosis. Br Med J 2:565–568. https://doi.org/10.1136/bmj.2.5813.565
Bizjak Mali L, Sepčić K, Bulog B (2013) Long-term starvation in cave salamander effects on liver ultrastructure and energy reserve mobilization. J Morphol 274:887–900. https://doi.org/10.1002/jmor.20145
Browning JD, Baxter J, Satapati S, Burgess SC (2012) The effect of short-term fasting on liver and skeletal muscle lipid, glucose, and energy metabolism in healthy women and men. J Lipid Res 53:577–586. https://doi.org/10.1194/jlr.P020867
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162:156–159. https://doi.org/10.1006/abio.1987.9999
da Silva RSM, Migliorini RH et al (1990) Effects of starvation and refeeding on energy-linked metabolic processes in the turtle (Phrynops hilarii). Comp Biochem Physiol A Comp Physiol 96:415–419
Dias Junior W, Baviera AM, Zanon NM et al (2016) Lipolytic response of adipose tissue and metabolic adaptations to long periods of fasting in red tilapia (Oreochromis sp., Teleostei: Cichlidae). An Acad Bras Cienc 88:1743–1754. https://doi.org/10.1590/0001-3765201620150484
Farrar ES, Dupre RK (1983) The role of diet in glycogen storage by juvenile bullfrogs prior to overwintering. Comp Biochem Physiol A Comp Physiol 75:255–260
Geisler CE, Hepler C, Higgins MR, Renquist BJ (2016) Hepatic adaptations to maintain metabolic homeostasis in response to fasting and refeeding in mice. Nutr Metab 13:62. https://doi.org/10.1186/s12986-016-0122-x
Groom DJE, Kuchel L, Richards JG (2013) Metabolic responses of the South American ornate horned frog (Ceratophrys ornata) to estivation. Comp Biochem Physiol B Biochem Mol Biol 164:2–9. https://doi.org/10.1016/j.cbpb.2012.08.001
Hervant F, Mathieu J, Durand J (2001) Behavioural, physiological and metabolic responses to long-term starvation and refeeding in a blind cave-dwelling (Proteus anguinus) and a surface-dwelling (Euproctus asper) salamander. J Exp Biol 204:269–281. https://doi.org/10.1242/jeb.204.2.269
Ishihara A, Sapon MA, Yamauchi K (2019) Seasonal acclimatization and thermal acclimation induce global histone epigenetic changes in liver of bullfrog (Lithobates catesbeianus) tadpole. Comp Biochem Physiol A Mol Integr Physiol 230:39–48. https://doi.org/10.1016/j.cbpa.2018.12.014
Li C, Wei Q, Gu X et al (2019) Decreased glycogenolysis by miR-338-3p promotes regional glycogen accumulation within the spinal cord of amyotrophic lateral sclerosis mice. Front Mol Nuerosci 12:114. https://doi.org/10.3389/fnmol.2019.00114
Merkle S (1989) Long-term starvation in Xenopus laevis Daudin–III. Effects on enzymes in several tissues. Comp Biochem Physiol B 94:783–788
Merkle S, Hanke W (1988a) Long-term starvation in Xenopus laevis daudin—I. Effects on general metabolism. Comp Biochem Physiol b Comp Biochem 89:719–730. https://doi.org/10.1016/0305-0491(88)90314-8
Merkle S, Hanke W (1988b) Long-term starvation in Xenopus laevis Daudin–II. Effects on several organs. Comp Biochem Physiol A Comp Physiol 90:491–495. https://doi.org/10.1016/0300-9629(88)90225-3
Miwa I, Okudo J, Maeda K, Okuda G (1972) Mutarotase effect on colorimetric determination of blood glucose with -D-glucose oxidase. Clin Chim Acta 37:538–540
Mutel E, Gautier-Stein A, Abdul-Wahed A et al (2011) Control of blood glucose in the absence of hepatic glucose production during prolonged fasting in mice: induction of renal and intestinal gluconeogenesis by glucagon. Diabetes 60:3121–3131. https://doi.org/10.2337/db11-0571
Peres H, Santos S, Oliva-Teles A (2014) Blood chemistry profile as indicator of nutritional status in European seabass (Dicentrarchus labrax). Fish Physiol Biochem 40:1339–1347. https://doi.org/10.1007/s10695-014-9928-5
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45. https://doi.org/10.1093/nar/29.9.e45
Roe JH, Dailey RE (1966) Determination of glycogen with the anthrone reagent. Anal Biochem 15:245–250
Sato T, Yoshida Y, Morita A et al (2016) Glycerol-3-phosphate dehydrogenase 1 deficiency induces compensatory amino acid metabolism during fasting in mice. Metabolism 65:1646–1656. https://doi.org/10.1016/j.metabol.2016.08.005
Secor SM, Carey HV (2016) Integrative physiology of fasting. Compr Physiol 6:773–825. https://doi.org/10.1002/cphy.c150013
Shimizu S, Yasui K, Tani Y, Yamada H (1979) Acyl-CoA oxidase from Candida tropicalis. Biochem Biophys Res Commun 91:108–113. https://doi.org/10.1016/0006-291x(79)90589-8
Soengas JL, Strong EF, Fuentes J et al (1996) Food deprivation and refeeding in Atlantic salmon, Salmo salar: effects on brain and liver carbohydrate and ketone bodies metabolism. Fish Physiol Biochem 15:491–511. https://doi.org/10.1007/BF01874923
Soty M, Chilloux J, Delalande F et al (2016) Post-translational regulation of the glucose-6-phosphatase complex by cyclic adenosine monophosphate is a crucial determinant of endogenous glucose production and is controlled by the glucose-6-phosphate transporter. J Proteome Res 15:1342–1349. https://doi.org/10.1021/acs.jproteome.6b00110
Spayd RW, Bruschi B, Burdick BA et al (1978) Multilayer film elements for clinical analysis: applications to representative chemical determinations. Clin Chem 24:1343–1350
Talke H, Schubert GE (1965) Enzymatic urea determination in the blood and serum in the warburg optical test. Klin Wochenschr 43:174–175. https://doi.org/10.1007/BF01484513
Tamaoki K, Okada R, Ishihara A et al (2016) Morphological, biochemical, transcriptional and epigenetic responses to fasting and refeeding in intestine of Xenopus laevis. Cell Biosci 6:2. https://doi.org/10.1186/s13578-016-0067-9
Tamaoki K, Ishihara A, Yamauchi K (2018) Expression pattern and histone acetylation of energy metabolic genes in Xenopus laevis liver in response to diet statuses. J Exp Zool A Ecol Integr Physiol 331:120–127. https://doi.org/10.1002/jez.2246
Teruya T, Chaleckis R, Takada J et al (2019) Diverse metabolic reactions activated during 58-hr fasting are revealed by non-targeted metabolomic analysis of human blood. Sci Rep 9:854. https://doi.org/10.1038/s41598-018-36674-9
Viegas I, Rito J, González JD et al (2013) Effects of food-deprivation and refeeding on the regulation and sources of blood glucose appearance in European seabass (Dicentrarchus labrax L.). Comp Biochem Physiol A Mol Integr Physiol 166:399–405. https://doi.org/10.1016/j.cbpa.2013.07.013
Williamson DH, Mellanby J, Krebs HA (1962) Enzymic determination of D(-)-beta-hydroxybutyric acid and acetoacetic acid in blood. Biochem J 82:90–96. https://doi.org/10.1042/bj0820090
Woof C, Janssens PA (1978) Effects of fasting and cortisol administration on carbohydrate metabolism in Xenopus laevis Daudin. Gen Comp Endocrinol 36:346–359. https://doi.org/10.1016/0016-6480(78)90116-8
Zammit VA, Newsholme EA (1979) Activities of enzymes of fat and ketone-body metabolism and effects of starvation on blood concentrations of glucose and fat fuels in teleost and elasmobranch fish. Biochem J 184:313–322. https://doi.org/10.1042/bj1840313
Zeng L-Q, Li F-J, Li X-M et al (2012) The effects of starvation on digestive tract function and structure in juvenile southern catfish (Silurus meridionalis Chen). Comp Biochem Physiol A Mol Integr Physiol 162:200–211. https://doi.org/10.1016/j.cbpa.2012.02.022
Acknowledgements
We thank Editage (https://www.editage.jp/) for a thorough and critical reading and revision of the manuscript.
Funding
This work was supported in part by Grant of Fujieda City Open Type Regional Policy Research Project from Fujieda city, Shizuoka, Japan.
Author information
Authors and Affiliations
Contributions
Material preparation, data collection, and analysis were performed by AN, RY, and MS. The first draft of the manuscript was prepared by AI and KY. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
All breeding and experimental procedures were approved by the Shizuoka University Animal Experiment Committee (Permit Number 29F-8) under the International Guidelines on Welfare and Management of Animals (Ministry of the Environment).
Additional information
Communicated by K.H. Dausmann.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nakajima, A., Yamaguchi, R., Sasazaki, M. et al. Adult male Xenopus laevis can tolerate months of fasting by catabolizing carbohydrates and lipids. J Comp Physiol B 193, 227–238 (2023). https://doi.org/10.1007/s00360-023-01478-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-023-01478-5