Abstract
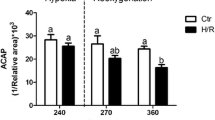
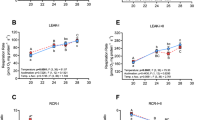
Rapid fluctuations of the oxygen content of both natural and anthropogenic origin are relatively common in freshwater environments. Fish adaptation to these conditions implies tolerance of both low levels of oxygen availability and reoxygenation. Hypoxia tolerance in fish has been widely studied, but the involvement of mitochondria in the response of fish to rapid hypoxia/reoxygenation stress is less known. Zebrafish, a floodplain species, is likely facing significant changes in dissolved oxygen in its natural environment and displays a moderate ability to tolerate hypoxia. In the present study, we report the effects of an acute hypoxia/reoxygenation stress (H/R) protocol on mitochondrial functionality (respiration, complex activities, rate of H2O2 release) and redox state (level of HPs and protein oxidation) of muscle tissue. In parallel, the animal metabolic performance (routine metabolism, nitrogen excretion and swimming performance) was measured. Additionally, the recovery from H/R was tested 20 h after treatment. A significant stimulation by H/R of muscle mitochondrial respiration and H2O2 release was observed, which was only in part counteracted by stimulation of the antioxidant system, resulting in an increased level of lipid peroxides and protein carbonyls. In parallel, H/R increased the animal oxygen consumption and urea excretion rate and reduced routine activity. A significant strong reduction of endurance at 80% Ucrit was also observed. Most of the altered parameter did not recover 20 h after reoxygenation. These data indicate a significant alteration of zebrafish muscle mitochondrial state after acute H/R, associated with changes in tissue redox state and locomotor performance.




Similar content being viewed by others
References
Amérand A, Vettier A, Sébert P, Moisan C (2006) A comparative study of reactive oxygen species in red muscle: pressure effects. Undersea Hyperb Med 33:161–167
Barré H, Bailly L, Rouanet JL (1987) Increased oxidative capacity in skeletal muscles from cold-acclimated ducklings: a comparison with rats. Comp Biochem Physiol Part B Comp Biochem 88:519–522. https://doi.org/10.1016/0305-0491(87)90337-3
Barrionuevo WR, Fernandes MN, Rocha O (2010) Aerobic and anaerobic metabolism for the zebrafish, Danio rerio, reared under normoxic and hypoxic conditions and exposed to acute hypoxia during development. Braz J Biol 70:425–434
Bayley PB (1995) Understanding large river: floodplain ecosystems. Bioscience 45:153–158. https://doi.org/10.2307/1312554
Bickler PE, Buck LT (2007) Hypoxia tolerance in reptiles, amphibians, and fishes: life with variable oxygen availability. Annu Rev Physiol 69:145–170. https://doi.org/10.1146/annurev.physiol.69.031905.162529
Bosworth CA, Chou C-W, Cole RB, Rees BB (2005) Protein expression patterns in zebrafish skeletal muscle: initial characterization and the effects of hypoxic exposure. Proteomics 5:1362–1371. https://doi.org/10.1002/pmic.200401002
Bourdineaud J-P, Rossignol R, Brèthes D (2013) Zebrafish: a model animal for analyzing the impact of environmental pollutants on muscle and brain mitochondrial bioenergetics. Int J Biochem Cell Biol 45:16–22. https://doi.org/10.1016/j.biocel.2012.07.021
Boveris A, Chance B (1973) The mitochondrial generation of hydrogen peroxide. General properties and effect of hyperbaric oxygen. Biochem J 134:707–716
Burgetz IJ, Rojas-Vargas A, Hinch SG, Randall DJ (1998) Initial recruitment of anaerobic metabolism during sub-maximal swimming in rainbow trout (Oncorhynchus mykiss). J Exp Biol 201:2711–2721
Cao C, Leng Y, Huang W, Liu X, Kufe D (2003) Glutathione peroxidase 1 Is regulated by the c-Abl and Arg tyrosine kinases. J Biol Chem 278:39609–39614. https://doi.org/10.1074/jbc.M305770200
Carlberg I, Mannervik B (1975) Purification and characterization of the flavoenzyme glutathione reductase from rat liver. J Biol Chem 250:5475–5480
Chakravarty S, Reddy BR, Sudhakar SR, Saxena S, Das T, Meghah V, Swamy B, Kumar CV, Idris A, M.M (2013) Chronic unpredictable stress (CUS)-induced anxiety and related mood disorders in a zebrafish model: altered brain proteome profile implicates mitochondrial dysfunction. PLoS One 8:e63302. https://doi.org/10.1371/journal.pone.0063302
Chapman LJ, Mckenzie DJ (2009) Chapter 2 behavioral responses and ecological consequences. In: Richards JG, Farrell AP, Brauner CJ (eds) Fish physiology, hypoxia. Academic Press, Cambridge, pp 25–77. https://doi.org/10.1016/S1546-5098(08)00002-2
Cheek A, Landry C, Steele S, Manning S (2009) Diel hypoxia in marsh creeks impairs the reproductive capacity of estuarine fish populations. Mar Ecol Prog Ser 392:211–221. https://doi.org/10.3354/meps08182
Cresci A, De Rosa R, Putman NF, Agnisola C (2017) Earth-strength magnetic field affects the rheotactic threshold of zebrafish swimming in shoals. Comp Biochem Physiol A Mol Integr Physiol 204:169–176. https://doi.org/10.1016/j.cbpa.2016.11.019
Diaz RJ, Breitburg DL (2009) The hypoxic environment. In: Richards JG, Farrell AP, Brauner CJ (eds) Hypoxia, fish physiology. Academic Press, Amsterdam, pp 1–23. https://doi.org/10.1016/S1546-5098(08)00001-0
DiMichele L, Powers DA (1982) Physiological basis for swimming endurance differences between LDH-B genotypes of Fundulus heteroclitus. Science 216:1014–1016. https://doi.org/10.1126/science.7079747
Du SNN, Mahalingam S, Borowiec BG, Scott GR (2016) Mitochondrial physiology and reactive oxygen species production are altered by hypoxia acclimation in killifish (Fundulus heteroclitus). J Exp Biol 219:1130–1138. https://doi.org/10.1242/jeb.132860
Farrell AP, Richards JG (2009) Defining hypoxia: an integrative synthesis of the responses of fish to hypoxia. In: Richards JG, Farrell AP, Brauner CJ (eds) Hypoxia, fish physiology. Academic Press, Amsterdam, pp 487–503. https://doi.org/10.1016/S1546-5098(08)00001-0
Filho DW (2007) Reactive oxygen species, antioxidants and fish mitochondria. Front Biosci 12:1229. https://doi.org/10.2741/2141
Flohé L, Günzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105:114–120
Galli GLJ, Richards JG (2014) Mitochondria from anoxia-tolerant animals reveal common strategies to survive without oxygen. J Comp Physiol B 184:285–302. https://doi.org/10.1007/s00360-014-0806-3
Gornall AG, Bardawill CJ, David MM (1949) Determination of serum proteins by means of the biuret reaction. J Biol Chem 177:751–766
Granger DN, Kvietys PR (2015) Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol 6:524–551. https://doi.org/10.1016/j.redox.2015.08.020
Griffith RW (1980) Chemistry of the body fluids of the coelacanth, latimeria chalumnae. Proc R Soc B Biol Sci 208:329–347. https://doi.org/10.1098/rspb.1980.0054
Griffiths E (2012) Mitochondria and heart disease. In: Scatena R, Bottoni P, Giardina B (eds) Advances in mitochondrial medicine, advances in experimental medicine and biology. Springer, Amsterdam, pp 249–267
Hafner RP, Brown GC, Brand MD (1990) Analysis of the control of respiration rate, phosphorylation rate, proton leak rate and protonmotive force in isolated mitochondria using the ‘top-down’ approach of metabolic control theory. Eur J Biochem 188:313–319. https://doi.org/10.1111/j.1432-1033.1990.tb15405.x
Hagenaars A, Vergauwen L, Benoot D, Laukens K, Knapen D (2013) Mechanistic toxicity study of perfluorooctanoic acid in zebrafish suggests mitochondrial dysfunction to play a key role in PFOA toxicity. Chemosphere 91:844–856. https://doi.org/10.1016/j.chemosphere.2013.01.056
Heath RL, Tappel AL (1976) A new sensitive assay for the measurement of hydroperoxides. Anal Biochem 76:184–191. https://doi.org/10.1016/0003-2697(76)90277-3
Hermes-Lima M, Zenteno-Savın T (2002) Animal response to drastic changes in oxygen availability and physiological oxidative stress. Comp Biochem Physiol Part C Toxicol Pharmacol 133:537–556
Hermes-Lima M, Storey JM, Storey KB (1998) Antioxidant defenses and metabolic depression. The hypothesis of preparation for oxidative stress in land snails. Comp Biochem Physiol B Biochem Mol Biol 120:437–448
Hyslop PA, Sklar LA (1984) A quantitative fluorimetric assay for the determination of oxidant production by polymorphonuclear leukocytes: Its use in the simultaneous fluorimetric assay of cellular activation processes. Anal Biochem 141:280–286. https://doi.org/10.1016/0003-2697(84)90457-3
Ivanov AS, Putvinskiĭ AV, Antonov VF, Vladimirov IA (1977) Magnitude of the protein permeability of liposomes following photoperoxidation of lipids. Biofizika 22:621–624
Jain KE, Hamilton JC, Farrell AP (1997) Use of a ramp velocity test to measure critical swimming speed in rainbow trout (Onchorhynchus mykiss). Comp Biochem Physiol 117A:441–444
Keller ET, Murtha JM (2004) The use of mature zebrafish (Danio rerio) as a model for human aging and disease. Comp Biochem Physiol Part C Toxicol Pharmacol 138:335–341. https://doi.org/10.1016/j.cca.2004.04.001
Landman MJ, Van Den Heuvel MR, Ling N (2005) Relative sensitivities of common freshwater fish and invertebrates to acute hypoxia. N Z J Mar Freshw Res 39:1061–1067. https://doi.org/10.1080/00288330.2005.9517375
Lenaz G (2012) Mitochondria and reactive oxygen species. Which role in physiology and pathology? In: Scatena R, Bottoni P, Giardina B (eds), Advances in mitochondrial medicine, advances in experimental medicine and biology. Springer, Amsterdam, pp 96–136
Lerebours A, Gonzalez P, Adam C, Camilleri V, Bourdineaud J-P, Garnier-Laplace J (2009) Comparative analysis of gene expression in brain, liver, skeletal muscles, and gills of zebrafish (Danio rerio) exposed to environmentally relevant waterborne uranium concentrations. Environ Toxicol Chem 28:1271–1278
Leveelahti L, Rytkönen KT, Renshaw GMC, Nikinmaa M (2014) Revisiting redox-active antioxidant defenses in response to hypoxic challenge in both hypoxia-tolerant and hypoxia-sensitive fish species. Fish Physiol Biochem 40:183–191. https://doi.org/10.1007/s10695-013-9835-1
Li J, Xie X (2018) Inconsistent responses of liver mitochondria metabolism and standard metabolism in Silurus meridionalis when exposed to waterborne cadmium. Comp Biochem Physiol Part C Toxicol Pharmacol 214:17–22. https://doi.org/10.1016/j.cbpc.2018.08.003
Lucas MC, Priede IG (1992) Utilization of metabolic scope in relation to feeding and activity by individual and grouped zebrafish, Brachydanio rerio (Hamilton-Buchanan). J Fish Biol 41:175–190. https://doi.org/10.1111/j.1095-8649.1992.tb02648.x
Lushchak VI (2011) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101:13–30. https://doi.org/10.1016/j.aquatox.2010.10.006
Lushchak VI (2012) Glutathione homeostasis and functions: potential targets for medical interventions. J Amino Acids. https://doi.org/10.1155/2012/736837
Lushchak VI, Bagnyukova TV (2006) Effects of different environmental oxygen levels on free radical processes in fish. Comp Biochem Physiol B Biochem Mol Biol 144:283–289. https://doi.org/10.1016/j.cbpb.2006.02.014
Mandic M, Todgham AE, Richards JG (2009) Mechanisms and evolution of hypoxia tolerance in fish. Proc R Soc B Biol Sci 276:735–744. https://doi.org/10.1098/rspb.2008.1235
Martos-Sitcha JA, Bermejo-Nogales A, Calduch-Giner JA, Pérez-Sánchez J (2017) Gene expression profiling of whole blood cells supports a more efficient mitochondrial respiration in hypoxia-challenged gilthead sea bream (Sparus aurata). Front Zool. https://doi.org/10.1186/s12983-017-0220-2
Mayzaud P, Conover RJ (1988) O:N atomic ratio as a tool to describe zooplankton metabolism. Mar Ecol Prog Ser 45:289–302
McDonald MD, Wood CM (2004) The effect of chronic cortisol elevation on urea metabolism and excretion in the rainbow trout (Oncorhynchus mykiss). J Comp Physiol 174:71–81. https://doi.org/10.1007/s00360-003-0390-4
Onukwufor JO, MacDonald N, Kibenge F, Stevens D, Kamunde C (2014) Effects of hypoxia-cadmium interactions on rainbow trout (Oncorhynchus mykiss) mitochondrial bioenergetics: attenuation of hypoxia-induced proton leak by low doses of cadmium. J Exp Biol 217:831–840. https://doi.org/10.1242/jeb.093344
Onukwufor JO, Kibenge F, Stevens D, Kamunde C (2016) Hypoxia-reoxygenation differentially alters the thermal sensitivity of complex I basal and maximal mitochondrial oxidative capacity. Comp Biochem Physiol A Mol Integr Physiol 201:87–94. https://doi.org/10.1016/j.cbpa.2016.06.033
Paital B (2013) Antioxidant and oxidative stress parameters in brain of Heteropneustes fossilis under air exposure condition; role of mitochondrial electron transport chain. Ecotoxicol Environ Saf 95:69–77. https://doi.org/10.1016/j.ecoenv.2013.05.016
Palmer G, Horgan DJ, Tisdale H, Singer TP, Beinert H (1968) Studies on the respiratory chain-linked reduced nicotinamide adenine dinucleotide dehydrogenase XIV. Location of the sites of inhibition of rotenone, barbiturates, and piericidin by means of electron paramagnetic resonance spectroscopy. J Biol Chem 243:844–847
Putvinskiĭ AV (1977) Decrease in the electrical stability of lipid membranes following UV-irradiation. Biofizika 22:725–727
Ragan CI, Wilson MT, Darley-Usmar VM, Lowe PN (1987) Sub-fractionation of mitochondria and isolation of the proteins of oxidative phosphorylation. In: Darley-Usmar VM, Rickwood D, Wilson MT (eds) Mitochondria: a practical approach. IRL Press, Oxford, pp 79–112
Rees BB, Sudradjat F, Love JW (2001) Acclimation to hypoxia increases survival time of zebrafish, Danio rerio, during lethal hypoxia. J Exp Zool 247
Reznick AZ, Packer L (1994) [38] Oxidative damage to proteins: spectrophotometric method for carbonyl assay. In: methods in enzymology, oxygen radicals in biological systems part C. Academic Press, Cambridge, pp 357–363. https://doi.org/10.1016/S0076-6879(94)33041-7
Richards JG (2009) Metabolic and molecular responses of fish to hypoxia. In: Richards JG, Farrell AP, Brauner CJ (eds) Hypoxia, fish physiology. Academic Press, Amsterdam, pp 443–485. https://doi.org/10.1016/S1546-5098(08)00001-0
Robertson CE, Wright PA, Koblitz L, Bernier NJ (2014) Hypoxia-inducible factor-1 mediates adaptive developmental plasticity of hypoxia tolerance in zebrafish, Danio rerio. Proc R Soc B Biol Sci 281:20140637–20140637. https://doi.org/10.1098/rspb.2014.0637
Simon LM, Robin ED (1971) Relationship of cytochrome oxidase activity to vertebrate total and organ oxygen consumption—ScienceDirect. Int J Biochem 2:569–573
Solórzano L (1969) Determination of ammonia in natural waters by the phenolhypochlorite method. Limnol Oceanogr 14:799–801. https://doi.org/10.4319/lo.1969.14.5.0799
Speers-Roesch B, Sandblom E, Lau GY, Farrell AP, Richards JG (2010) Effects of environmental hypoxia on cardiac energy metabolism and performance in tilapia. Am J Physiol Regul Integr Comp Physiol 298:R104–R119. https://doi.org/10.1152/ajpregu.00418.2009
Spence R, Gerlach G, Lawrence C, Smith C (2008) The behaviour and ecology of the zebrafish, Danio rerio. Biol Rev 83:13–34. https://doi.org/10.1111/j.1469-185X.2007.00030.x
Thengchaisri N, Hein TW, Wang W, Xu X, Li Z, Fossum TW, Kuo L (2006) Upregulation of arginase by H 2 O 2 impairs endothelium-dependent nitric oxide-mediated dilation of coronary arterioles. Arterioscler Thromb Vasc Biol 26:2035–2042. https://doi.org/10.1161/01.ATV.0000233334.24805.62
Thorarensen H (2011) The effect of exercise on respiration. In: Farrell AP, Cech JJ, Richards JG, Stevens ED (eds) Encyclopedia of fish: from genome to environment. Academic Press, Cambridge, pp 812–819
Tierney KB (2011) Swimming performance assessment in fishes. J Vis Exp 1:e2572. https://doi.org/10.3791/2572
Ton C, Stamatiou D, Liew C-C (2003) Gene expression profile of zebrafish exposed to hypoxia during development. Physiol Genom 13:97–106. https://doi.org/10.1152/physiolgenomics.00128.2002
Turrens JF, Alexandre A, Lehninger AL (1985) Ubisemiquinone is the electron donor for superoxide formation by complex III of heart mitochondria. Arch Biochem Biophys 237:408–414. https://doi.org/10.1016/0003-9861(85)90293-0
Uliano E, Cataldi M, Carella F, Migliaccio O, Iaccarino D, Agnisola C (2010) Effects of acute changes in salinity and temperature on routine metabolism and nitrogen excretion in gambusia (Gambusia affinis) and zebrafish (Danio rerio). Comp Biochem Physiol Mol Integr Physiol 157:283–290. https://doi.org/10.1016/j.cbpa.2010.07.019
van der Meer DLM, van den Thillart GEEJM, Witte F, de Bakker MAG, Besser J, Richardson MK, Spaink HP, Leito JTD, Bagowski CP (2005) Gene expression profiling of the long-term adaptive response to hypoxia in the gills of adult zebrafish. Am J Physiol Regul Integr Comp Physiol 289:R1512–R1519. https://doi.org/10.1152/ajpregu.00089.2005
Venditti P, Masullo P, Meo SD (2001) Effects of myocardial ischemia and reperfusion on mitochondrial function and susceptibility to oxidative stress. Cell Mol Life Sci CMLS 58:1528–1537. https://doi.org/10.1007/PL00000793
Venditti P, De Rosa R, Di Meo S (2003) Effect of thyroid state on H2O2 production by rat liver mitochondria. Mol Cell Endocrinol 205:185–192
Venditti P, De Rosa R, Cigliano L, Agnisola C, Di Meo S (2004) Role of nitric oxide in the functional response to ischemia–reperfusion of heart mitochondria from hyperthyroid rats. Cell Mol Life Sci CMLS 61:2244–2252. https://doi.org/10.1007/s00018-004-4125-9
Verkerk AO, Remme CA (2012) Zebrafish: a novel research tool for cardiac (patho)electrophysiology and ion channel disorders. Front Physiol. https://doi.org/10.3389/fphys.2012.00255
Ward AC, Lieschke GJ (2002) The zebrafish as a model system for human disease. Front Biosci 7:d827–d833
Welker AF, Campos ÉG, Cardoso LA, Hermes-Lima M (2012) Role of catalase on the hypoxia/reoxygenation stress in the hypoxia-tolerant Nile tilapia. Am J Physiol Regul Integr Comp Physiol 302:R1111–R1118. https://doi.org/10.1152/ajpregu.00243.2011
Wu G, Morris SM (1998) Arginine metabolism: nitric oxide and beyond. Biochem J 336:1–17. https://doi.org/10.1042/bj3360001
Acknowledgements
AA acknowledges support from the Basic Research Funding of the Biology Department of the Naples University Federico II.
Author information
Authors and Affiliations
Contributions
PV conceived and coordinated the study together with CA and helped draft the manuscript. GN and DB attended to the experiment concerning the evaluation of mitochondrial bioenergetic state, oxidative damage and antioxidant status. DE and EU attended to the experimental set up of in vivo experiments, collected the related experimental data and participated in the data analysis. CA proposed the underlying hypothesis of the study, conceived and coordinated the study together with PV, designed the study, and helped draft the manuscript. All authors gave final approval for publication.
Corresponding author
Additional information
Communicated by G. Heldmaier.
Rights and permissions
About this article
Cite this article
Napolitano, G., Venditti, P., Fasciolo, G. et al. Acute hypoxia/reoxygenation affects muscle mitochondrial respiration and redox state as well as swimming endurance in zebrafish. J Comp Physiol B 189, 97–108 (2019). https://doi.org/10.1007/s00360-018-1198-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-018-1198-6