Abstract
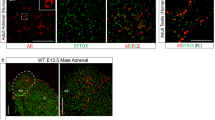
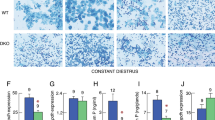
The wild sand rat, Psammomys obesus, displays seasonal variations in adrenocortical activity that parallel those of testicular activity, indicating functional cross-talk between the hypothalamo-pituitary-adrenal and hypothalamo-pituitary–gonadal axes. In the present study, we examined androgen receptor (AR)-mediated actions of testicular steroids in the regulation of adrenocortical function in the sand rat. Specifically, we examined the expression of AR in the adrenal cortex, as well as adrenal apoptosis in male sand rats that had been surgically castrated or castrated and supplemented with testosterone; biochemical indices of adrenocortical function and hormone profiles were also measured. Orchiectomy was followed by an increase in adrenocorticotropic hormone secretion from the anterior pituitary and subsequently, increased adrenocortical activity; the latter was evidenced by orchiectomy-induced increases in the adrenal content of cholesterol and lipids as well as adrenal hypertrophy (seen as an elevation of the RNA/DNA ratio). Further, androgen deprivation respectively up- and downregulated the incidence of apoptosis within the glucocorticoid-producing zona fasciculata and sex steroid-producing zona reticularis. Interestingly, orchiectomy resulted in increased expression of AR in the zona fasciculata. All of the orchiectomy-induced cellular and biochemical responses were reversible after testosterone substitution therapy. Together, these data suggest that adrenocortical activity in the sand rat is seasonally modulated by testicular androgens that act through AR located in the adrenal cortex itself.



Similar content being viewed by others
References
Alario P, Gamallo A, Beato MJ, Trancho G (1987) Body weight gain, food intake and adrenal development in chronic noise stressed rats. Physiol Behav 40:29–32
Almeida OFX, Canoine V, Ali S, Holsboer F, Patchev VK (1997) Activational effects of gonadal steroids on hypothalamo-pituitary-adrenal regulation in the rat disclosed by response to dexamethasone suppression. J Neuroendocrinol l9:129–134
Amirat Z, Brudieux R (1993) Seasonal changes in in vivo cortisol response to ACTH and in plasma and pituitary concentrations of ACTH in a desert rodent, the sand rat (Psammomys obesus). Comp Biochem Physiol 104A:29–34
Amirat Z, Khammar F, Brudieux R (1980) Seasonal changes in plasma and adrenal concentrations of cortisol, corticosterone, aldosterone and electrolytes in the adult male sand rat (Psammomys obesus). Gen Comp Endocrinol 40:36–43
Askew EB, Gampe RT, Stanley TB, Faggart JL, Wilson EM (2007) Modulation of androgen receptor activation function 2 by testosterone and dihydrotestosterone. J Biol Chem 282:25801–25816
Bagamasbad P, Denver RJ (2011) Mechanisms and significance of nuclear receptor auto- and cross-regulation. Gen Comp Endocrinol 170:3–17
Benmouloud A, Zahaf S, Khammar F, Amirat Z (2006) Influence de la castration sur la surrénale du rat des sables Psammomys obesus mâle adulte. Bull Soc Hist Nat Afr Nord 73:197–205
Bentvelsen FM, McPhaul MJ, Wilson CM, Wilson JD, George FW (1996) Regulation of immunoreactive androgen receptor in the adrenal gland of the adult rat. Endocrinology 137:2659–2663
Bingaman EW, Magnuson DJ, Gray TS, Handa RJ (1994) Androgen inhibits the increases in hypothalamic corticotropin-releasing hormone (CRH) and CRH-immunoreactivity following gonadectomy. Neuroendocrinology 59:228–234
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Burke RW, Diamondstone BI, Velapoldi RA, Menis O (1974) Mechanisms of the Liebermann-Burchard and zak color reactions for cholesterol. Clin Chem 20:794–801
Carlson HE (2007) Human adrenal cortex hyperfunction due to LH/hCG. Mol Cell Endocrinol 269:46–50
Chang C, Yeh S, Lee SO, Chang T (2013) Androgen receptor (AR) pathophysiological roles in androgen related diseases in skin, bone/muscle, metabolic syndrome and neuron/immune systems: lessons learned from mice lacking AR in specific cells. Nucl Recep Signaling 11:1–26
Cretzschmar J (1828) In: Atlas zu der Reise im Nördlichen Afrika von Eduard Rüppel, Saügethier, Frankfurt am Main. pp 58–59
Dallman MF, Akana SF, Bhatnagar S, Bell ME, Choi S, Chu A, Horsley C, Levin N, Meijer O, Soriano LR, Strack AM, Viau V (1999) Starvation: early signals, sensors and sequelae. Endocrinology 140:4015–4023
Dalterio SL, Mayfield DL, Michael SD, Macmillan BT, Bartke A (1983) Effects of delta 9-THC and castration on behavior and plasma hormone levels in male mice. Pharmacol Biochem Behav 18:81–86
Daly M, Daly S (1973) On the feeding ecology of Psammomys obesus (Rodentia, Gerbillidae) in the Wadi Saoura, Algeria. Mammalia 37:545–561
Duda T, Waliszewska A, Trzeciak WH, Malendowicz LK (1985) Sex differences in adrenocortical structure and function. XX. The effects of gonadectomy and testosterone or estradiol replacement on cholesterol content and distribution in the gland. J Steroid Biochem 23:577–581
Freedman BD, Kempna PB, Carlone DL, Shah MS, Guagliardo NA, Barrett PQ, Gomez-Sanchez CE, Majzoub JA, Breault DT (2013) Adrenocortical zonation results from lineage conversion of differentiated zona glomerulosa cells. Dev Cell 26:666–673
Gavrieli Y, Sherman Y, Ben-Sasson SA (1992) Identification of programmed cell death in situ: a specific labelling of nuclear DNA fragmentation. J Cell Biol 119:493–501
Gernigon T, Ablaoui R, Boudoucha D, Kandsi F (1992) Cytological and biochemical effects of castration of a Saharan rodent with a seasonal cycle Psammomys obesus. Bull Soc Zoo Fr 117(3):343
Handa RJ, Weiser MJ (2014) Gonadal steroid hormones and the hypothalamo-pituitary-adrenal axis. Front Neuroendocrinol 35:197–220
Handa RJ, Burgess LH, Kerr JE, O’Keefe JA (1994) Gonadal steroid hormone receptors and sex differences in the hypothalamo-pituitary-adrenal axis. Horm Behav 28:464–476
Hirst JJ, West NB, Brenner RM, Novey MJ (1992) Steroid hormone receptors in the adrenal glands of fetal and adult rhesus monkeys. J Clin Endocrinol Metab 75:308–314
Hoeflich A, Bielohuby M (2009) Mechanisms of adrenal gland growth: signal integration by extracellular signal regulated kinases1/2. J Mol Endocrinol 42:191–203
Johnsen IK, Slawik M, Shapiro I, Hartmann MF, Wudy SA, Looyenga BD, Hammer GD, Reincke M, Beuschlein F (2006) Gonadectomy in mice of the inbred strain CE/J induces proliferation of sub-capsular adrenal cells expressing gonadal marker genes. J Endocrinol 190:47–57
Kandsi-Bouhadad F, Hadj-Bekkouche F (2010) Évaluation du contenu surrénalien en androstènedione et effets de la castration chez le lapin domestique (Oryctolagus cuniculus). C R Biologies 333:591–596
Keegan CE, Hammer GD (2002) Recent insights into organogenesis of the adrenal cortex. Trends Endocrinol Metab 13:200–208
Khammar F, Brudieux R (1984) Seasonal changes in testicular contents of testosterone and androstenedione and in metabolic clearance rate of testosterone in the sand rat (Psammomys obesus). J Reprod Fert 71:235–241
Kitay JI (1975) Effects of various hormones on the pituitary-adrenal axis. Adv Exp Med Bio 54:155–167
Kobayashi H, Kambe F, Imai T, Hibi Y, Kikumori T, Ohmori S, Nakao A, Seo H (2006) Differential expression of cyclin-dependent kinase inhibitors, p27Kip1 and p57Kip2, by corticotropin in rat adrenal cortex. J Endocrinol 189:671–679
Lubahn DB, Josef DR, Sullivan PM, Willard HF, French FS, Wilson EM (1988) Cloning of human androgen receptor complementary DNA and localization to the X chromosome. Science 240:327–330
Lund TD, Munson DJ, Haldy ME, Handa RJ (2004) Dihydrotestosterone may inhibit hypothalamo-pituitary-adrenal activity by acting through estrogen receptor in the male mouse. Neurosci Lett 365:43–47
Malendowicz LK, Młynarczyk W (1982) Sex differences in adrenocortical structure and function. X. Lipid and corticosterone in the rat adrenal as affected by gonadectomy and testosterone or estradiol replacement. Endokrinologie 79:292–300
Miller WL (1988) Molecular biology of steroid hormone synthesis. Endocr Rev 9:295–318
Patchev VK, Almeida OFX (1996) Gonadal steroids exert faciliting and “buffering” effects on glucocorticoid-mediated transcriptional regulation of corticotropin-releasing hormone and corticosteroid receptor genes in rat brain. J Neurosci 16:7077–7084
Patchev VK, Almeida OFX (1998) Gender specificity in the neural regulation of the response to stress: new leads from classical paradigms. Mol Neurobiol 16:63–77
Rao CV, Zhou XL, Lei ZM (2004) Functional luteinizing hormone/chorionic gonadotropin receptors in human adrenal cortical H295R cells. Biol Reprod 71:579–587
Raynaud J, Muller K, Schradin C (2012) Experimental increase of testosterone levels in free-ranging juvenile male African striped mice (Rhabdomys pumilio) induces physiological, morphological, and behavioral changes. Gen Comp Endocrinol 178:108–115
Romero LM (2002) Seasonal changes in plasma glucocorticoid concentrations in free-living vertebrates. Gen Comp Endocrinol 128:1–24
Rossi R, Zatelli MC, Valentini A, Cavazzini P, Fallo F, del Senno L, degli Uberti EC (1998) Evidence for androgen receptor gene expression and growth inhibitory effect of dihydrotestosterone on human adrenocortical cells. Endocrinology 159:373–380
Schmidt EE, Schibler U (1995) Cell size regulation, a mechanism that controls cellular RNA accumulation: consequence on regulation of the ubiquitous transcription factor Oct1 and NF-Y and liver enriched transcription factor DBP. J Cell Biol 28:467–483
Schradin C (2008) Seasonal changes in testosterone and corticosterone levels in four social classes of a desert dwelling sociable rodent. Horm Behav 53:573–579
Shibko S, Koivistoven P, Tratnyek CA, Newhall AR, Friedman L (1967) Method for sequential quantitative separation and determination of protein, RNA, DNA, lipid and glycogen from a single liver homogenate or from subcellular fraction. Anal Biochem 19:415–528
Simard J, Ricketts ML, Gingras S, Soucy P, Feltus FA, Melner MH (2005) Molecular biology of the 3beta-hydroxysteroid dehydrogenase/delta5-delta4 isomerase gene family. Endocr Rev 26:525–582
Stalvey JRD (2002) Inhibition of 3β-hydroxysteroid dehydrogenase-isomerase in mouse adrenal cells: a direct effect of testosterone. Steroids 67:721–731
Thomas JL, Bucholtz KM, Kacsoh B (2011) Selective inhibition of human 3β-hydroxysteroid dehydrogenase type 1 as a potential treatment for breast cancer. J Steroid Biochem Mol Biol 125:57–65
Viau V (2002) Functional cross-talk between the hypothalamic-pituitary-gonadal and -adrenal axes. J Neuroendocrinol 14:506–513
Winick M, Noble A (1965) Quantitative change in DNA, RNA and protein during prenatal and postnatal growth in the rat. Dev Biol 12:451–466
Wolkersdörfer GW, Bornstein SR (1998) Tissue remodelling in the adrenal gland. Biochem Pharmacol 56:163–171
Acknowledgments
The authors thank all the personnel of Beni-Abbes Station in the Algerian Sahara for trapping the sand rats used in this study. They also thank M.T. Laurent, N. Mouterfi, and R. Stoffel for technical assistance. The support of the Algerian Ministry of Higher Education and Scientific Research, the Max Planck Institute of Psychiatry (Munich, Germany), the Life and Health Sciences Research Institute (ICVS) of the University of Minho (Braga, Portugal) and the Université Catholique de Lyon (France) is acknowledged. Support from the Algerian–French collaborative Project 09 MDU 756 is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier.
Rights and permissions
About this article
Cite this article
Benmouloud, A., Amirat, Z., Khammar, F. et al. Androgen receptor-mediated regulation of adrenocortical activity in the sand rat, Psammomys obesus . J Comp Physiol B 184, 1055–1063 (2014). https://doi.org/10.1007/s00360-014-0859-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-014-0859-3