Abstract
Purpose
The goal of this systematic review was to examine the current literature on the urinary microbiome and its associations with noninfectious, nonmalignant, urologic diseases. Secondarily, we aimed to describe the most common bioinformatics used to analyze the urinary microbiome.
Methods
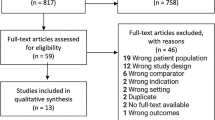
A comprehensive literature search of Ovid MEDLINE using the keywords “microbiota” AND “prostatic hyperplasia,” “microbiota” AND “urinary bladder, overactive,” “microbiota” AND “pelvic pain,” and “microbiota” AND “urolithiasis” OR “nephrolithiasis” OR “urinary calculi” AND “calcium oxalate” was performed to identify relevant clinical microbiome studies associated with noninfectious benign urological conditions published from 2010 to 2022. We included human studies that evaluated the urinary, stone, or semen microbiota, or any combination of the above-mentioned locations.
Results
A total of 25 human studies met the inclusion criteria: 4 on benign prostatic hyperplasia (BPH), 9 on overactive bladder (OAB), 8 on calcium oxalate stones, and 4 on chronic pelvic pain syndrome (CPPS). Specific taxonomic profiles in the urine microbiome were associated with each pathology, and evaluation of alpha- and beta-diversity and relative abundance was accounted for most of the studies. Symptom prevalence and severity were also analyzed and showed associations with specific microbes.
Conclusion
The study of the urogenital microbiome is rapidly expanding in urology. Noninfectious benign urogenital diseases, such as BPH, calcium oxalate stones, CPPS, and OAB were found to be associated with specific microbial taxonomies. Further research with larger study populations is necessary to solidify the knowledge of the urine microbiome in these conditions and to facilitate the creation of microbiome-based diagnostic and therapeutic approaches.

Similar content being viewed by others
References
Wolfe AJ et al (2012) Evidence of uncultivated bacteria in the adult female bladder. J Clin Microbiol 50(4):1376–1383. https://doi.org/10.1128/JCM.05852-11
Wang Y, Qian P-Y (2009) Conservative fragments in bacterial 16S rRNA genes and primer design for 16S ribosomal DNA amplicons in metagenomic studies. PLoS One 4(10):e7401. https://doi.org/10.1371/journal.pone.0007401
Liu Z, Lozupone C, Hamady M, Bushman FD, Knight R (2007) Short pyrosequencing reads suffice for accurate microbial community analysis. Nucleic Acids Res 35(18):e120. https://doi.org/10.1093/nar/gkm541
Kim M, Morrison M, Yu Z (2011) Evaluation of different partial 16S rRNA gene sequence regions for phylogenetic analysis of microbiomes. J Microbiol Methods 84(1):81–87. https://doi.org/10.1016/j.mimet.2010.10.020
Schloss PD, Westcott SL (2011) Assessing and improving methods used in operational taxonomic unit-based approaches for 16S rRNA gene sequence analysis. Appl Environ Microbiol 77(10):3219–3226. https://doi.org/10.1128/AEM.02810-10
Bolyen E et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37(8):852–857. https://doi.org/10.1038/s41587-019-0209-9
Schloss PD (2021) Amplicon Sequence Variants Artificially Split Bacterial Genomes into Separate Clusters. mSphere 6(4):e0019121. https://doi.org/10.1128/mSphere.00191-21
Van Dooren TJM (2019) Assessing species richness trends: Declines of bees and bumblebees in the Netherlands since 1945. Ecol Evol 9(23):13056–13068. https://doi.org/10.1002/ece3.5717
Lin H, Peddada SD (2020) Analysis of compositions of microbiomes with bias correction. Nat Commun 11(1):3514. https://doi.org/10.1038/s41467-020-17041-7
Segata N et al (2011) Metagenomic biomarker discovery and explanation. Genome Biol 12(6):R60. https://doi.org/10.1186/gb-2011-12-6-r60
Liberati A et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700. https://doi.org/10.1136/bmj.b2700
Nickel JC et al (2016) Assessment of the Lower Urinary Tract Microbiota during Symptom Flare in Women with Urologic Chronic Pelvic Pain Syndrome: A MAPP Network Study. J Urol 195(2):356–362. https://doi.org/10.1016/j.juro.2015.09.075
Berry SH et al (2011) Prevalence of symptoms of bladder pain syndrome/interstitial cystitis among adult females in the United States. J Urol 186(2):540–544. https://doi.org/10.1016/j.juro.2011.03.132
Nickel JC (2011) Prostatitis. Can Urol Assoc J 5(5):306–315. https://doi.org/10.5489/cuaj.11211
Shoskes DA, Altemus J, Polackwich AS, Tucky B, Wang H, Eng C (2016) The Urinary Microbiome Differs Significantly Between Patients With Chronic Prostatitis/Chronic Pelvic Pain Syndrome and Controls as Well as Between Patients With Different Clinical Phenotypes. Urology 92:26–32. https://doi.org/10.1016/j.urology.2016.02.043
Holvoet T et al (2021) Fecal Microbiota Transplantation Reduces Symptoms in Some Patients With Irritable Bowel Syndrome With Predominant Abdominal Bloating: Short- and Long-term Results From a Placebo-Controlled Randomized Trial. Gastroenterology 160(1):145-157.e8. https://doi.org/10.1053/j.gastro.2020.07.013
Kartha GK, Kerr H, Shoskes DA (2013) Clinical phenotyping of urologic pain patients. Curr Opin Urol 23(6):560–564. https://doi.org/10.1097/MOU.0b013e3283652a9d
Joshi N et al (2010) Association between periodontal disease and prostate-specific antigen levels in chronic prostatitis patients. J Periodontol 81(6):864–869. https://doi.org/10.1902/jop.2010.090646
Shoskes DA, Wang H, Polackwich AS, Tucky B, Altemus J, Eng C (2016) Analysis of Gut Microbiome Reveals Significant Differences between Men with Chronic Prostatitis/Chronic Pelvic Pain Syndrome and Controls. J Urol 196(2):435–441. https://doi.org/10.1016/j.juro.2016.02.2959
Alkanani AK et al (2015) Alterations in Intestinal Microbiota Correlate With Susceptibility to Type 1 Diabetes. Diabetes 64(10):3510–3520. https://doi.org/10.2337/db14-1847
Lundy SD et al (2021) Functional and Taxonomic Dysbiosis of the Gut, Urine, and Semen Microbiomes in Male Infertility. Eur Urol 79(6):826–836. https://doi.org/10.1016/j.eururo.2021.01.014
Mändar R et al (2017) Seminal microbiome in men with and without prostatitis. Int J Urol 24(3):211–216. https://doi.org/10.1111/iju.13286
Weng S-L et al (2014) Bacterial communities in semen from men of infertile couples: metagenomic sequencing reveals relationships of seminal microbiota to semen quality. PLoS One 9(10):e110152. https://doi.org/10.1371/journal.pone.0110152
Barbonetti A et al (2011) Effect of vaginal probiotic lactobacilli on in vitro-induced sperm lipid peroxidation and its impact on sperm motility and viability. Fertil Steril 95(8):2485–2488. https://doi.org/10.1016/j.fertnstert.2011.03.066
Abernethy MG, Rosenfeld A, White JR, Mueller MG, Lewicky-Gaupp C, Kenton K (2017) Urinary Microbiome and Cytokine Levels in Women With Interstitial Cystitis. Obstet Gynecol 129(3):500–506. https://doi.org/10.1097/AOG.0000000000001892
Siddiqui H, Lagesen K, Nederbragt AJ, Jeansson SL, Jakobsen KS (2012) Alterations of microbiota in urine from women with interstitial cystitis. BMC Microbiol 12:205. https://doi.org/10.1186/1471-2180-12-205
Darbro BW, Petroelje BK, Doern GV (2009) Lactobacillus delbrueckii as the cause of urinary tract infection. J Clin Microbiol 47(1):275–277. https://doi.org/10.1128/JCM.01630-08
Nickel JC et al (2022) The bacterial microbiota of Hunner lesion interstitial cystitis/bladder pain syndrome. BJU Int 129(1):104–112. https://doi.org/10.1111/bju.15519
Kachroo N et al (2021) Meta-analysis of Clinical Microbiome Studies in Urolithiasis Reveal Age, Stone Composition, and Study Location as the Predominant Factors in Urolithiasis-Associated Microbiome Composition. mBio 12(4):e02007-21. https://doi.org/10.1128/mBio.02007-21
Deng Q, Wang Z, Wang J, Zhang J, Zhang Y, Liang H (2022) 16S rRNA gene sequencing reveals altered composition of gut microbiota in postoperative individuals with renal stones. Lett Appl Microbiol 75(2):271–280. https://doi.org/10.1111/lam.13701
Ziemba JB, Matlaga BR (2017) Epidemiology and economics of nephrolithiasis. Investig Clin Urol 58(5):299–306. https://doi.org/10.4111/icu.2017.58.5.299
Saigal CS, Joyce G, Timilsina AR, Urologic Diseases in America Project (2005) Direct and indirect costs of nephrolithiasis in an employed population: opportunity for disease management? Kidney Int 68(4):1808–1814. https://doi.org/10.1111/j.1523-1755.2005.00599.x
Xiang L et al (2022) Prediction of the occurrence of calcium oxalate kidney stones based on clinical and gut microbiota characteristics. World J Urol 40(1):221–227. https://doi.org/10.1007/s00345-021-03801-7
Shen C et al (2021) Identifying Two Novel Clusters in Calcium Oxalate Stones With Urinary Tract Infection Using 16S rDNA Sequencing. Front Cell Infect Microbiol 11:723781. https://doi.org/10.3389/fcimb.2021.723781
Kachroo N, Monga M, Miller AW (2022) Comparative functional analysis of the urinary tract microbiome for individuals with or without calcium oxalate calculi. Urolithiasis 50(3):303–317. https://doi.org/10.1007/s00240-022-01314-5
Dornbier RA et al (2020) The microbiome of calcium-based urinary stones. Urolithiasis 48(3):191–199. https://doi.org/10.1007/s00240-019-01146-w
Zhao Z, Xia Y, Xue J, Wu Q (2014) Role of E. coli-Secretion and Melamine in Selective Formation of CaC2O4·H2O and CaC2O4·2H2O Crystals. Cryst Growth Des 14(2):450–458. https://doi.org/10.1021/cg400960c
Chutipongtanate S, Sutthimethakorn S, Chiangjong W, Thongboonkerd V (2013) Bacteria can promote calcium oxalate crystal growth and aggregation. J Biol Inorg Chem 18(3):299–308. https://doi.org/10.1007/s00775-012-0974-0
Izatulina AR, Nikolaev AM, Kuz’mina MA, Frank-Kamenetskaya OV, Malyshev VV (2019) Bacterial Effect on the Crystallization of Mineral Phases in a Solution Simulating Human Urine. Crystals. https://doi.org/10.3390/cryst9050259
Saw JJ et al (2021) In Vivo Entombment of Bacteria and Fungi during Calcium Oxalate, Brushite, and Struvite Urolithiasis. Kidney360 2(2):298–311. https://doi.org/10.34067/KID.0006942020
Borghi L, Nouvenne A, Meschi T (2012) Nephrolithiasis and urinary tract infections: ‘the chicken or the egg’ dilemma? Nephrol Dial Transplant 27(11):3982–3984. https://doi.org/10.1093/ndt/gfs395
Kim H-N et al (2022) Gut microbiota and the prevalence and incidence of renal stones. Sci Rep 12(1):3732. https://doi.org/10.1038/s41598-022-07796-y
Liu M et al (2017) Oxalobacter formigenes-associated host features and microbial community structures examined using the American Gut Project. Microbiome 5:108. https://doi.org/10.1186/s40168-017-0316-0
Suryavanshi MV, Bhute SS, Jadhav SD, Bhatia MS, Gune RP, Shouche YS (2016) Hyperoxaluria leads to dysbiosis and drives selective enrichment of oxalate metabolizing bacterial species in recurrent kidney stone endures. Sci Rep 6:34712. https://doi.org/10.1038/srep34712
Chen F et al (2021) Gut microbiota affect the formation of calcium oxalate renal calculi caused by high daily tea consumption. Appl Microbiol Biotechnol 105(2):789–802. https://doi.org/10.1007/s00253-020-11086-w
Zampini A, Nguyen AH, Rose E, Monga M, Miller AW (2019) Defining Dysbiosis in Patients with Urolithiasis. Sci Rep 9:5425. https://doi.org/10.1038/s41598-019-41977-6
Denburg MR, Koepsell K, Lee J-J, Gerber J, Bittinger K, Tasian GE (2020) Perturbations of the Gut Microbiome and Metabolome in Children with Calcium Oxalate Kidney Stone Disease. J Am Soc Nephrol 31(6):1358–1369. https://doi.org/10.1681/ASN.2019101131
Rusakov AV, Vlasov AD, Zelenskaya MS, Frank-Kamenetskaya OV, Vlasov DYu (2016) The Crystallization of Calcium Oxalate Hydrates Formed by Interaction Between Microorganisms and Minerals”, in Biogenic—Abiogenic Interactions in Natural and Anthropogenic Systems. In: Frank-Kamenetskaya OV, Panova EG, Vlasov DYu (eds) Lecture Notes in Earth System Sciences. Springer International Publishing, Cham, pp 357–377. https://doi.org/10.1007/978-3-319-24987-2_28
Izatulina AR, Yelnikov VYu (2008) Structure, Chemistry and Crystallization Conditions of Calcium Oxalates—The Main Components of Kidney Stones. In: Krivovichev SV (ed) Minerals as Advanced Materials I. Berlin, Heidelberg, Springer, Berlin Heidelberg, pp 231–239. https://doi.org/10.1007/978-3-540-77123-4_29
Ruijter GJG, Van De Vondervoort PJI, Visser J (1999) Oxalic acid production by Aspergillus niger: an oxalate-non-producing mutant produces citric acid at pH 5 and in the presence of manganese. Microbiology 145(9):2569–2576. https://doi.org/10.1099/00221287-145-9-2569
Payne CL, Dark MJ, Conway JA, Farina LL (2017) A retrospective study of the prevalence of calcium oxalate crystals in veterinary Aspergillus cases. J VET Diagn Invest 29(1):51–58. https://doi.org/10.1177/1040638716672254
Zhao E, Zhang W, Geng B, You B, Wang W, Li X (2021) Intestinal dysbacteriosis leads to kidney stone disease. Mol Med Rep 23(3):180. https://doi.org/10.3892/mmr.2020.11819
Miller AW, Choy D, Penniston KL, Lange D (2019) Inhibition of urinary stone disease by a multi-species bacterial network ensures healthy oxalate homeostasis. Kidney Int 96(1):180–188. https://doi.org/10.1016/j.kint.2019.02.012
Liu F et al (2020) Characteristics of the urinary microbiome in kidney stone patients with hypertension. J Transl Med 18:130. https://doi.org/10.1186/s12967-020-02282-3
Parsons JK (2010) Benign Prostatic Hyperplasia and Male Lower Urinary Tract Symptoms: Epidemiology and Risk Factors. Curr Bladder Dysfunct Rep 5(4):212–218. https://doi.org/10.1007/s11884-010-0067-2
Yu H, Meng H, Zhou F, Ni X, Shen S, Das UN (2015) Urinary microbiota in patients with prostate cancer and benign prostatic hyperplasia. Arch Med Sci 11(2):385–394. https://doi.org/10.5114/aoms.2015.50970
Bajic P et al (2020) Male Bladder Microbiome Relates to Lower Urinary Tract Symptoms. Eur Urol Focus 6(2):376–382. https://doi.org/10.1016/j.euf.2018.08.001
Holland B et al (2020) The effect of the urinary and faecal microbiota on lower urinary tract symptoms measured by the International Prostate Symptom Score: analysis utilising next-generation sequencing. BJU Int 125(6):905–910. https://doi.org/10.1111/bju.14972
Pearce MM et al (2014) The female urinary microbiome: a comparison of women with and without urgency urinary incontinence. mBio 5(4):e01283-01214. https://doi.org/10.1128/mBio.01283-14
Lee H-Y et al (2021) The impact of urine microbiota in patients with lower urinary tract symptoms. Ann Clin Microbiol Antimicrob 20(1):23. https://doi.org/10.1186/s12941-021-00428-9
Radej S, Szewc M, Maciejewski R (2022) Prostate Infiltration by Treg and Th17 Cells as an Immune Response to Propionibacterium acnes Infection in the Course of Benign Prostatic Hyperplasia and Prostate Cancer. Int J Mol Sci 23(16):8849. https://doi.org/10.3390/ijms23168849
Lloyd GL, Marks JM, Ricke WA (2019) Benign Prostatic Hyperplasia and Lower Urinary Tract Symptoms: What Is the Role and Significance of Inflammation? Curr Urol Rep 20(9):54. https://doi.org/10.1007/s11934-019-0917-1
Scarneciu I et al (2021) Overactive bladder: a review and update. Exp Ther Med 22(6):1444. https://doi.org/10.3892/etm.2021.10879
Hilt EE et al (2014) Urine is not sterile: use of enhanced urine culture techniques to detect resident bacterial flora in the adult female bladder. J Clin Microbiol 52(3):871–876. https://doi.org/10.1128/JCM.02876-13
Curtiss N, Balachandran A, Krska L, Peppiatt-Wildman C, Wildman S, Duckett J (2017) A case controlled study examining the bladder microbiome in women with Overactive Bladder (OAB) and healthy controls. Eur J Obstet Gynecol Reprod Biol 214:31–35. https://doi.org/10.1016/j.ejogrb.2017.04.040
Li K et al (2022) Interplay between bladder microbiota and overactive bladder symptom severity: a cross-sectional study. BMC Urol 22(1):39. https://doi.org/10.1186/s12894-022-00990-0
Price TK, Hilt EE, Thomas-White K, Mueller ER, Wolfe AJ, Brubaker L (2020) The urobiome of continent adult women: a cross-sectional study. BJOG 127(2):193–201. https://doi.org/10.1111/1471-0528.15920
Karstens L et al (2016) Does the Urinary Microbiome Play a Role in Urgency Urinary Incontinence and Its Severity? Front Cell Infect Microbiol 6:78. https://doi.org/10.3389/fcimb.2016.00078
Nardos R et al (2022) Network-Based Differences in the Vaginal and Bladder Microbial Communities Between Women With and Without Urgency Urinary Incontinence. Front Cell Infect Microbiol 12:759156. https://doi.org/10.3389/fcimb.2022.759156
Newman ME (2003) A Measure of Betweenness Centrality based on Random Walks. Soc Netw 27:39–54. https://doi.org/10.1016/j.socnet.2004.11.009
Stavropoulou E et al (2021) Focus on the Gut-Kidney Axis in Health and Disease. Front Med 7:620102. https://doi.org/10.3389/fmed.2020.620102
Author information
Authors and Affiliations
Contributions
Dr HNS had full access to all the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis. Protocol/project development: SA, JM, KS, RM, TRWH, KA. Data collection or management: SA, JM, JGP, PKS, KS, SR. Data analysis: PKS, SR. Manuscript writing/editing: SA, JM, JGP, PKS, SR, KS, RM, TRWH, KA, HNS.
Corresponding author
Ethics declarations
Conflict of interest
Dr Hemendra Shah received $1000 from Lumenis for mentoring urologists for HoLEP in 2019, and he has been sponsored by Boston Scientific and Applaud. No funding from these companies was received for the study. All other authors have nothing to disclose.
Ethical approval
This systematic review was conducted retrospectively from deidentified data obtained from previously published articles only performed in humans. We consulted extensively with the IRB of the University of Miami who determined that our study did not need ethical approval in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Suarez Arbelaez, M.C., Monshine, J., Porto, J.G. et al. The emerging role of the urinary microbiome in benign noninfectious urological conditions: an up-to-date systematic review. World J Urol 41, 2933–2948 (2023). https://doi.org/10.1007/s00345-023-04588-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-023-04588-5