Abstract
Purpose
Few data are available regarding the nephrotoxicity of immune checkpoint inhibitor (ICI) combination therapy in advanced renal cell carcinoma (RCC). This study aimed to investigate the nephrotoxicity of ICI-based combination therapy versus standard of care sunitinib in patients with advanced RCC.
Methods
We searched Embase/PubMed/Cochrane Library for relevant randomized controlled trials (RCTs). Treatment-related nephrotoxicities including increase of creatinine and proteinuria were analyzed by Review Manager 5.4 software.
Results
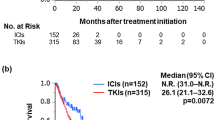
Seven RCTs involving 5239 patients were included. The analysis showed that ICI combination therapy had similar risks of any grade (RR = 1.03, 95% CI: 0.77–1.37, P = 0.87) and grade 3–5 (RR = 1.48, 95% CI: 0.19–11.66, P = 0.71) increased creatinine compared with sunitinib monotherapy. However, ICI combination therapy was associated with significantly higher risks of any grade (RR = 2.33, 95% CI: 1.54–3.51, P < 0.0001) and grade 3–5 proteinuria (RR = 2.25, 95% CI: 1.21–4.17, P = 0.01).
Conclusions
This meta-analysis suggests that ICI combination therapy shows more nephrotoxicity of proteinuria than sunitinib in advanced RCC, which deserves a high attention in the clinic.






Similar content being viewed by others
Data availability
Date related to this article can be obtained from the corresponding author on request.
Abbreviations
- PD-1:
-
Programmed cell death protein-1
- PD-L1:
-
Programmed cell death 1 ligand 1
- ICI:
-
Immune checkpoint inhibitor
- CTLA-4:
-
Cytotoxic T lymphocyte antigen 4
- RR:
-
Relative risk
- RCT:
-
Randomized controlled trial
- AKI:
-
Acute kidney injury
- RCC:
-
Renal cell carcinoma
References
Barata PC, Rini BI (2017) Treatment of renal cell carcinoma: current status and future directions. CA Cancer J Clin 67(6):507–524. https://doi.org/10.3322/caac.21411
Procopio G, Nichetti F, Verzoni E (2020) Pembrolizumab plus axitinib: another step ahead in advanced renal cell carcinoma. Lancet Oncol 21(12):1538–1539. https://doi.org/10.1016/s1470-2045(20)30482-4
Mo DC, Huang JF, Luo PH, Huang SX, Wang HL (2021) Combination therapy with immune checkpoint inhibitors in advanced renal cell carcinoma: a meta-analysis of randomized controlled trials. Clin Immunol 232:108876. https://doi.org/10.1016/j.clim.2021.108876
Rassy E, Flippot R, Albiges L (2020) Tyrosine kinase inhibitors and immunotherapy combinations in renal cell carcinoma. Ther Adv Med Oncol 12:1758835920907504. https://doi.org/10.1177/1758835920907504
Zhang ZF, Wang T, Liu LH, Guo HQ (2014) Risks of proteinuria associated with vascular endothelial growth factor receptor tyrosine kinase inhibitors in cancer patients: a systematic review and meta-analysis. PLoS ONE 9(3):e90135. https://doi.org/10.1371/journal.pone.0090135
Tinawi M, Bastani B (2020) Nephrotoxicity of immune checkpoint inhibitors: acute kidney injury and beyond. Cureus. 12(12):e12204. https://doi.org/10.7759/cureus.12204
Cortazar FB, Kibbelaar ZA, Glezerman IG, Abudayyeh A, Mamlouk O et al (2020) Clinical features and outcomes of immune checkpoint inhibitor-associated AKI: a multicenter study. J Am Soc Nephrol 31(2):435–446. https://doi.org/10.1681/ASN.2019070676
Chen G, Qin Y, Fan QQ, Zhao B, Mei D, Li XM (2020) Renal adverse effects following the use of different immune checkpoint inhibitor regimens: a real-world pharmacoepidemiology study of post-marketing surveillance data. Cancer Med 9(18):6576–6585. https://doi.org/10.1002/cam4.3198
Cortazar FB, Marrone KA, Troxell ML, Ralto KM, Hoenig MP et al (2016) Clinicopathological features of acute kidney injury associated with immune checkpoint inhibitors. Kidney Int 90(3):638–647. https://doi.org/10.1016/j.kint.2016.04.008
Dolladille C, Akroun J, Morice PM, Dompmartin A, Ezine E et al (2021) Cardiovascular immunotoxicities associated with immune checkpoint inhibitors: a safety meta-analysis. Eur Heart J 42(48):4964–4977. https://doi.org/10.1093/eurheartj/ehab618
Jamal S, Hudson M, Fifi-Mah A, Ye C (2020) Immune-related adverse events associated with cancer immunotherapy: a review for the practicing rheumatologist. J Rheumatol 47(2):166–175. https://doi.org/10.3899/jrheum.190084
Perazella MA, Shirali AC (2018) Nephrotoxicity of cancer immunotherapies: past, present and future. J Am Soc Nephrol 29(8):2039–2052. https://doi.org/10.1681/ASN.2018050488
Postow MA, Sidlow R, Hellmann MD (2018) Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med 378(2):158–168. https://doi.org/10.1056/NEJMra1703481
Qu J, Ding Y, Jiang K, Hao J, Li Y et al (2021) Nephrotoxicity of immune checkpoint inhibitors: a disproportionality analysis from 2013 to 2020. Tohoku J Exp Med 254(4):275–282. https://doi.org/10.1620/tjem.254.275
Rizzo A, Mollica V, Santoni M, Rosellini M, Marchetti A, Massari F (2022) Risk of toxicity with immunotherapy-tyrosine kinase inhibitors for metastatic renal cell carcinoma: a meta-analysis of randomized controlled trials. Future Oncol 18(5):625–634. https://doi.org/10.2217/fon-2021-0888
Wanchoo R, Karam S, Uppal NN, Barta VS, Deray G et al (2017) Adverse renal effects of immune checkpoint inhibitors: a narrative review. Am J Nephrol 45(2):160–169. https://doi.org/10.1159/000455014
Wang DY, Salem J-E, Cohen JV, Chandra S, Menzer C et al (2018) Fatal toxic effects associated with immune checkpoint inhibitors. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2018.3923
Wu Z, Chen Q, Qu L, Li M, Wang L et al (2022) Adverse events of immune checkpoint inhibitors therapy for urologic cancer patients in clinical trials: a collaborative systematic review and meta-analysis. Eur Urol 81(4):414–425. https://doi.org/10.1016/j.eururo.2022.01.028
Zhou X, Yao Z, Bai H, Duan J, Wang Z et al (2021) Treatment-related adverse events of PD-1 and PD-L1 inhibitor-based combination therapies in clinical trials: a systematic review and meta-analysis. Lancet Oncol 22(9):1265–1274. https://doi.org/10.1016/s1470-2045(21)00333-8
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. J Clin Epidemiol 134:178–189. https://doi.org/10.1016/j.jclinepi.2021.03.001
Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928. https://doi.org/10.1136/bmj.d5928
Choueiri TK, Motzer RJ, Rini BI, Haanen J, Campbell MT et al (2020) Updated efficacy results from the JAVELIN Renal 101 trial: first-line avelumab plus axitinib versus sunitinib in patients with advanced renal cell carcinoma. Ann Oncol 31(8):1030–1039. https://doi.org/10.1016/j.annonc.2020.04.010
Choueiri TK, Powles T, Burotto M, Escudier B, Bourlon MT et al (2021) Nivolumab plus Cabozantinib versus Sunitinib for advanced renal-cell carcinoma. N Engl J Med 384(9):829–841. https://doi.org/10.1056/NEJMoa2026982
McDermott DF, Huseni MA, Atkins MB, Motzer RJ, Rini BI et al (2018) Clinical activity and molecular correlates of response to atezolizumab alone or in combination with bevacizumab versus sunitinib in renal cell carcinoma. Nat Med 24(6):749–757. https://doi.org/10.1038/s41591-018-0053-3
Motzer R, Alekseev B, Rha SY, Porta C, Eto M et al (2021) Lenvatinib plus Pembrolizumab or Everolimus for advanced renal cell carcinoma. N Engl J Med 384(14):1289–1300. https://doi.org/10.1056/NEJMoa2035716
Motzer RJ, Powles T, Atkins MB, Escudier B, McDermott DF et al (2022) Final Overall survival and molecular analysis in IMmotion151, a phase 3 trial comparing atezolizumab plus Bevacizumab vs Sunitinib in patients with previously untreated metastatic renal cell carcinoma. JAMA Oncol 8(2):275–280. https://doi.org/10.1001/jamaoncol.2021.5981
Motzer RJ, Rini BI, McDermott DF, Arén Frontera O, Hammers HJ et al (2019) Nivolumab plus ipilimumab versus sunitinib in first-line treatment for advanced renal cell carcinoma: extended follow-up of efficacy and safety results from a randomised, controlled, phase 3 trial. Lancet Oncol 20(10):1370–1385. https://doi.org/10.1016/s1470-2045(19)30413-9
Powles T, Plimack ER, Soulières D, Waddell T, Stus V et al (2020) Pembrolizumab plus axitinib versus sunitinib monotherapy as first-line treatment of advanced renal cell carcinoma (KEYNOTE-426): extended follow-up from a randomised, open-label, phase 3 trial. Lancet Oncol 21(12):1563–1573. https://doi.org/10.1016/s1470-2045(20)30436-8
Ning K, Wu Z, Zou X, Liu H, Wu Y, Xiong L, Yu C, Guo S, Han H, Zhou F, Dong P, Zhang Z (2022) Immune checkpoint inhibitors further aggravate proteinuria in patients with metastatic renal cell carcinoma after long-term targeted therapy. Transl Androl Urol 11(3):386–396. https://doi.org/10.21037/tau-21-1015.PMID:35402197;PMCID:PMC898497
Tomita Y, Uemura H, Fujimoto H, Kanayama HO, Shinohara N et al (2011) Key predictive factors of axitinib (AG-013736)-induced proteinuria and efficacy: a phase II study in Japanese patients with cytokine-refractory metastatic renal cell Carcinoma. Eur J Cancer 47(17):2592–2602. https://doi.org/10.1016/j.ejca.2011.07.014
Motzer RJ, McDermott DF, Escudier B, Burotto M, Choueiri TK et al (2022) Conditional survival and long-term efficacy with nivolumab plus ipilimumab versus sunitinib in patients with advanced renal cell carcinoma. Cancer 128(11):2085–2097. https://doi.org/10.1002/cncr.34180
Acknowledgements
The authors would like to thank The Third Affiliated Hospital of Guangxi Medical University.
Funding
There is no funding for this study.
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. Study design, data collection and analysis were performed by A-jT, D-cM, KW and X-xX. The first draft of the manuscript was written by A-jT and D-cM, and all the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tan, Aj., Mo, Dc., Wu, K. et al. Nephrotoxicity of immune checkpoint inhibitor combination therapy in patients with advanced renal cell carcinoma: a meta-analysis. World J Urol 41, 1563–1571 (2023). https://doi.org/10.1007/s00345-023-04407-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-023-04407-x